Scalable Visible Light Promoted Synthesis of Biphenyl Benzothiazole Compounds for Pharmaceutical Applications
Scalable Visible Light Promoted Synthesis of Biphenyl Benzothiazole Compounds for Pharmaceutical Applications
The landscape of organic synthesis for heterocyclic compounds is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective methodologies that do not compromise on yield or purity. A pivotal development in this arena is detailed in Chinese Patent CN112939891A, which discloses a novel preparation method for biphenyl benzothiazole compounds. This technology represents a paradigm shift from traditional transition metal-catalyzed processes to a visible light-promoted, one-pot synthetic route. By utilizing N-(2-bromophenyl) alkyl thioamide derivatives as raw materials and sodium phosphate as a mild base, this invention achieves high reaction yields without the necessity for expensive photosensitizers or toxic heavy metal ions during the initial cyclization step. For R&D directors and procurement managers in the pharmaceutical sector, this innovation offers a compelling value proposition: the ability to produce high-purity pharmaceutical intermediates with a drastically simplified workflow and reduced environmental footprint.
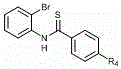
The significance of this patent lies not just in the chemical transformation itself, but in its potential to redefine the supply chain dynamics for complex heterocyclic building blocks. Benzothiazole derivatives are ubiquitous in medicinal chemistry, serving as core scaffolds for a wide array of bioactive molecules including anticancer agents, antifungal drugs, and kinase inhibitors. However, their conventional synthesis often involves harsh conditions, multiple purification steps, and the use of scarce precious metals. The methodology described in CN112939891A circumvents these bottlenecks by employing a simple 45W household compact fluorescent lamp as the energy source. This accessibility translates directly into operational flexibility for manufacturing facilities, allowing for the commercial scale-up of complex pharmaceutical intermediates without the need for specialized high-energy UV equipment or stringent handling protocols associated with air-sensitive organometallic catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzothiazole compounds and their biphenyl derivatives has relied heavily on the presence of visible or UV light in conjunction with sophisticated transition metal complexes. Prior art frequently cites the use of photocatalysts such as [Ru(bpy)3]2+ or [Ir(ppy)3], which, while effective, introduce substantial economic and logistical burdens. These transition metal complexes are not only exorbitantly expensive but also pose significant challenges regarding residual metal removal, a critical quality attribute for any active pharmaceutical ingredient (API) or advanced intermediate. Furthermore, the reliance on organic photosensitizers can introduce toxic organic pollutants into the reaction matrix, complicating waste stream management and increasing the cost of downstream processing. The need for additional equipment to handle sensitive catalysts and the multi-step nature of traditional routes often result in lower overall atom economy and extended production lead times, creating friction in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a brand-new visible light-promoted one-pot method that elegantly bypasses the limitations of prior art. The process initiates with an intramolecular cross-coupling reaction of N-(2-bromophenyl) thioamide under visible light irradiation in the presence of a simple inorganic base, specifically sodium phosphate. This step efficiently constructs the benzothiazole core without any external photosensitizer or transition metal catalyst. Following this cyclization, the reaction proceeds directly to a palladium-catalyzed coupling with phenylboronic acid in the same vessel. This telescoping of reactions eliminates the need for isolating the intermediate benzothiazole compound, thereby saving time, solvents, and labor. The entire process is green, efficient, and remarkably easy to operate, achieving unexpected technical effects through the use of common household lighting sources rather than specialized photochemical reactors.
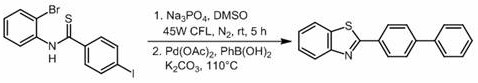
Mechanistic Insights into Visible Light-Promoted Cyclization and Cross-Coupling
The mechanistic pathway of this transformation is a fascinating interplay of radical chemistry and classical cross-coupling, optimized for industrial robustness. The initial phase involves the activation of the N-(2-bromophenyl) thioamide substrate under visible light irradiation. Although the patent notes the absence of an added photosensitizer, the reaction likely proceeds through a substrate-mediated electron transfer process or a radical mechanism facilitated by the interaction between the thioamide moiety and the base in DMSO solvent. The bromine site on the aromatic ring undergoes an intramolecular nucleophilic attack or radical substitution by the sulfur atom, leading to the formation of the benzothiazole ring system. This step is conducted at room temperature for 2 to 5 hours under an inert atmosphere, typically nitrogen, ensuring that oxygen-sensitive radical intermediates are protected while maintaining mild thermal conditions that preserve functional group integrity.
Following the successful formation of the benzothiazole intermediate, the reaction mixture is seamlessly transitioned into a Suzuki-Miyaura type cross-coupling regime. Without purification, phenylboronic acid, a palladium catalyst such as Pd(OAc)2, and potassium carbonate are introduced directly into the reaction vessel. The temperature is then elevated to between 100°C and 120°C to facilitate the oxidative addition, transmetallation, and reductive elimination cycles characteristic of palladium catalysis. This second stage installs the biphenyl motif onto the benzothiazole core. The choice of DMSO as the solvent is critical here, as it effectively dissolves both the organic substrates and the inorganic bases, creating a homogeneous medium that supports both the photochemical and thermal steps. The clarity of the reaction profile, as evidenced by the high HPLC yields reported in the examples, suggests minimal side reactions and a clean impurity profile, which is paramount for reducing lead time for high-purity pharmaceutical intermediates.
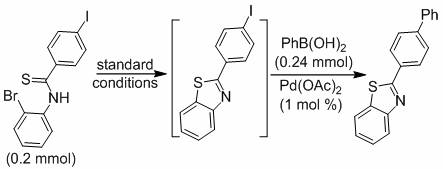
How to Synthesize Biphenyl Benzothiazole Efficiently
The operational simplicity of this synthesis makes it an ideal candidate for technology transfer and rapid adoption in contract manufacturing organizations. The protocol begins by charging a dry reaction tube equipped with a magnetic stirrer with N-(2-bromophenyl) thioamide and sodium phosphate in a molar ratio of approximately 1:0.5. Dimethyl sulfoxide (DMSO) is added as the solvent, and the system is purged with nitrogen to establish an inert environment. The mixture is then subjected to irradiation from a standard 45W compact fluorescent lamp for 5 hours at room temperature. Upon completion of the cyclization, the reaction does not require workup; instead, the palladium catalyst, phenylboronic acid, and potassium carbonate are added directly to the crude mixture. The temperature is raised to 110°C, and the reaction is stirred for an additional 5 hours. This streamlined procedure minimizes unit operations and maximizes throughput.
- Mix N-(2-bromophenyl) thioamide with sodium phosphate in DMSO under nitrogen and irradiate with a 45W CFL lamp for 5 hours to form the benzothiazole intermediate.
- Without purification, add phenylboronic acid, Pd(OAc)2 catalyst, and potassium carbonate directly to the reaction mixture.
- Heat the mixture to 110°C for 5 hours under nitrogen atmosphere to complete the cross-coupling and isolate the final biphenyl benzothiazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light-promoted methodology offers tangible strategic advantages that extend beyond mere chemical curiosity. The primary driver for cost optimization in this process is the elimination of expensive transition metal photocatalysts. By removing the dependency on Ruthenium or Iridium complexes, the raw material cost per kilogram of the final product is significantly reduced. Furthermore, the absence of these heavy metals in the initial cyclization step simplifies the purification process, potentially reducing the load on downstream metal scavenging resins or chromatography columns. This reduction in processing complexity translates directly into lower manufacturing overheads and a more competitive pricing structure for the reliable pharmaceutical intermediate supplier offering this technology.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the reagent profile and energy consumption. By utilizing a simple inorganic base like sodium phosphate instead of costly organic bases or metal catalysts for the cyclization step, the direct material costs are drastically lowered. Additionally, the use of a standard 45W household compact fluorescent lamp as the light source implies negligible energy costs compared to high-power UV arrays or cryogenic cooling systems often required for sensitive photochemistry. The one-pot nature of the synthesis further contributes to cost reduction in API manufacturing by consolidating two distinct chemical transformations into a single reactor run, thereby saving on solvent usage, labor hours, and equipment occupancy time.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable starting materials. N-(2-bromophenyl) thioamides and phenylboronic acids are commodity chemicals with robust global supply networks, reducing the risk of shortages associated with specialized catalysts. The mild reaction conditions, particularly the room temperature photochemical step, reduce the stress on reactor infrastructure and minimize the risk of thermal runaway incidents, ensuring consistent batch-to-batch reliability. Moreover, the tolerance of the method to various halogen substituents allows for the flexible sourcing of different thioamide precursors, providing procurement teams with the agility to switch suppliers based on availability and price without compromising the synthetic route.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with the principles of green chemistry, facilitating easier regulatory approval and waste management. The elimination of toxic heavy metal photocatalysts reduces the burden of hazardous waste disposal and lowers the environmental compliance costs associated with heavy metal discharge limits. The high atom economy and the ability to perform the reaction in a polar aprotic solvent like DMSO, which can be recovered and recycled, enhance the overall sustainability profile. This makes the commercial scale-up of complex pharmaceutical intermediates not only technically feasible but also environmentally responsible, appealing to multinational corporations with strict ESG (Environmental, Social, and Governance) mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is the visible light method superior to traditional transition metal catalysis for benzothiazoles?
A: Traditional methods often require expensive and toxic transition metal complexes like Ruthenium or Iridium photosensitizers. The patented visible light method eliminates the need for these auxiliary catalysts, using simple household compact fluorescent lamps instead, which drastically reduces reagent costs and removes heavy metal contamination risks from the final product.
Q: What are the specific reaction conditions for the one-pot synthesis?
A: The process utilizes N-(2-bromophenyl) thioamide as the starting material with sodium phosphate as the base in DMSO solvent. The initial cyclization occurs at room temperature under 45W CFL irradiation for 5 hours, followed immediately by the addition of palladium catalyst and boronic acid for heating at 110°C.
Q: Can this method tolerate various substituents on the aromatic ring?
A: Yes, the methodology demonstrates excellent functional group tolerance. The patent data confirms high yields for substrates containing electron-withdrawing groups such as chlorine, bromine, iodine, and trifluoromethyl groups, making it highly versatile for synthesizing diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Benzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible light-promoted synthesis described in CN112939891A and are fully equipped to leverage this technology for our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory benchtop to industrial reactor is seamless and efficient. Our state-of-the-art facilities are capable of accommodating the specific requirements of photochemical reactions, including the installation of scalable LED or CFL irradiation arrays, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains using this innovative methodology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our benzothiazole portfolio and to discuss route feasibility assessments for your next project. Let us help you harness the power of green chemistry to drive down costs and accelerate your time to market.
