Scalable Synthesis of Histidine-Based Acrylic Acid Derivatives for Advanced Peptide Manufacturing
Introduction to Advanced Histidine Peptide Synthesis
The field of peptide therapeutics has witnessed a paradigm shift with the discovery of histidine-rich polypeptides exhibiting potent biological activities, ranging from anti-arrhythmic properties to significant antitumor effects against ovarian and lung cancer cell lines. Patent CN108707115B introduces a groundbreaking synthetic methodology for producing (S)-2-(2-(tert-butyloxycarbonylamino)-3-(1-trityl-1H-imidazole-4-yl)propionamide)acrylic acid, a novel dipeptide compound containing essential histidine fragments. This specific acrylic acid derivative serves as a critical building block for constructing new polypeptides through subsequent reactions with other amino acids, thereby unlocking potential applications in drug screening and targeted therapy development. The invention addresses a significant gap in the prior art, where no literature had previously reported the synthesis of this specific histidine-containing acrylate, providing the pharmaceutical industry with a reliable pathway to access these high-value intermediates.
For R&D directors and process chemists, the availability of such specialized intermediates is paramount for accelerating the discovery of next-generation biologic drugs. The synthetic route detailed in this patent not only ensures the structural integrity of the chiral histidine center but also introduces a reactive acrylate moiety that allows for further functionalization. This dual functionality makes the compound an invaluable asset for medicinal chemists aiming to explore structure-activity relationships (SAR) in histidine-based therapeutic candidates. By establishing a robust and reproducible synthesis, this technology paves the way for the commercialization of complex peptide drugs that were previously difficult to manufacture due to the lack of efficient precursor availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of modified amino acid derivatives, particularly those containing sensitive functional groups like acrylates alongside protected amines and imidazoles, has been fraught with challenges. Conventional methods often suffer from low atom economy, requiring harsh reaction conditions that can lead to racemization of the chiral centers, thereby compromising the biological efficacy of the final peptide drug. Furthermore, traditional routes frequently involve multiple protection and deprotection steps that drastically increase the overall processing time and waste generation. The absence of a direct and efficient method to couple histidine derivatives with acrylic acid moieties meant that researchers had to rely on cumbersome multi-step sequences or inefficient coupling agents that resulted in poor yields and difficult purification profiles. These limitations significantly hindered the rapid scale-up required for clinical trials and commercial supply.
The Novel Approach
In stark contrast to these traditional hurdles, the novel approach outlined in the patent utilizes a streamlined four-step sequence that maximizes efficiency and minimizes operational complexity. The strategy employs a logical progression of condensation, activation, elimination, and hydrolysis to construct the target molecule with high fidelity. By starting with readily available (S)-2-(tert-butoxycarbonylamino)-3-(1-trityl-1H-imidazole-4-yl)propionic acid and serine methyl ester hydrochloride, the process leverages cost-effective raw materials. The use of EDCI for the initial amide bond formation ensures mild conditions that preserve stereochemistry, while the subsequent mesylation and DBU-mediated elimination provide a clean route to the acrylate functionality. This cohesive strategy eliminates the need for exotic catalysts or extreme temperatures, making it highly attractive for industrial adoption.
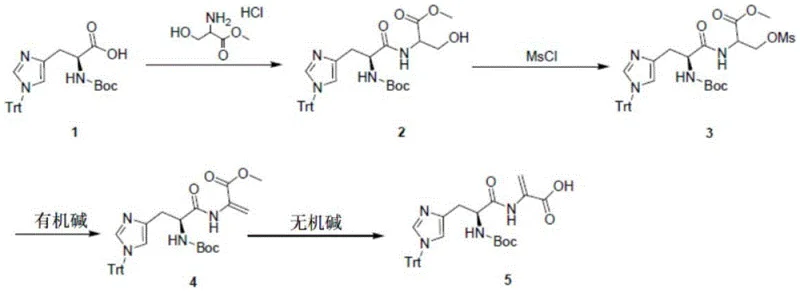
The visual representation of the reaction pathway highlights the elegance of this synthetic design, where each step flows logically into the next without the need for intermediate isolation in some variations, although the patent describes isolation for purity control. This modular approach allows for precise control over impurities, a critical factor for regulatory compliance in pharmaceutical manufacturing. The ability to generate the acrylic double bond late in the synthesis prevents premature polymerization or side reactions that could occur if the acrylate group were present during the initial coupling stages. Consequently, this novel approach represents a significant technological leap forward, offering a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of process robustness and product quality.
Mechanistic Insights into EDCI Coupling and Base-Mediated Elimination
The core of this synthesis lies in the precise orchestration of two key mechanistic events: the carbodiimide-mediated amide coupling and the base-promoted elimination reaction. In the first step, the carboxylic acid of the protected histidine is activated by EDCI (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide) to form an O-acylisourea intermediate. This highly reactive species is then attacked by the amine group of serine methyl ester hydrochloride, facilitated by an organic base such as pyridine or triethylamine which neutralizes the generated HCl. This mechanism is crucial because it proceeds under mild conditions (20-30 °C), effectively preventing the epimerization of the alpha-carbon of the histidine residue, which is a common pitfall in peptide synthesis. The resulting amide bond is stable, yet the presence of the free hydroxyl group on the serine side chain sets the stage for the subsequent transformation.
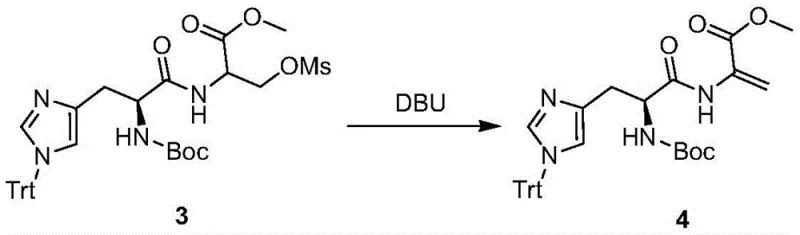
Following the activation of the hydroxyl group via mesylation with methanesulfonyl chloride (MsCl), the synthesis enters its most critical phase: the formation of the carbon-carbon double bond. This is achieved through an E2 elimination mechanism driven by a strong, non-nucleophilic organic base like DBU (1,8-diazabicycloundec-7-ene). The base abstracts the acidic proton alpha to the ester carbonyl, while the mesylate group acts as an excellent leaving group, departing simultaneously to form the acrylate double bond. The choice of DBU is mechanistically significant; its steric bulk prevents nucleophilic attack on the ester or the imidazole ring, ensuring that elimination is the exclusive pathway. This specificity is vital for maintaining the purity of the intermediate, as nucleophilic side products would be extremely difficult to separate from the desired acrylate.
Furthermore, the final hydrolysis step utilizing lithium hydroxide (LiOH) demonstrates a keen understanding of chemoselectivity. LiOH is strong enough to hydrolyze the methyl ester to the free carboxylic acid but is employed under controlled conditions (20-30 °C) that do not compromise the Boc protecting group or the trityl group on the imidazole nitrogen. This orthogonal stability is essential for the utility of the final product in downstream peptide synthesis, where these protecting groups are needed to direct subsequent coupling reactions. The mechanistic clarity of this route ensures that impurities are minimized at every stage, resulting in a final product that meets the stringent purity specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize (S)-2-(2-(tert-butyloxycarbonylamino)-3-(1-trityl-1H-imidazole-4-yl)propionamide)acrylic acid Efficiently
To successfully implement this synthesis in a laboratory or pilot plant setting, strict adherence to the reaction parameters defined in the patent is essential. The process begins with the dissolution of the protected histidine acid in a polar aprotic solvent like DMF, followed by the controlled addition of reagents to manage exotherms. The subsequent steps require careful temperature monitoring, particularly during the mesylation and elimination phases, to prevent degradation of the sensitive acrylate moiety. While the general workflow is straightforward, the devil is in the details regarding stoichiometry and workup procedures, which are critical for achieving the high yields reported in the examples. For a comprehensive guide on the exact operational parameters, please refer to the standardized synthesis steps provided below.
- Perform condensation reaction between (S)-2-(tert-butoxycarbonylamino)-3-(1-trityl-1H-imidazole-4-yl)propionic acid and serine methyl ester hydrochloride using EDCI.
- Conduct acylation reaction on the resulting hydroxypropionate using methanesulfonyl chloride (MsCl) to activate the hydroxyl group.
- Execute elimination reaction using an organic base like DBU to form the acrylic double bond.
- Complete the synthesis via inorganic base hydrolysis (e.g., LiOH) followed by acidification to obtain the final acrylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers transformative benefits that directly impact the bottom line and operational reliability. The primary advantage lies in the utilization of commodity chemicals and widely available reagents. Starting materials such as serine methyl ester hydrochloride and protected histidine derivatives are produced on a massive global scale, ensuring a stable supply chain that is resistant to market fluctuations. Unlike processes that rely on scarce transition metal catalysts or proprietary ligands, this method depends on standard organic reagents like EDCI, MsCl, and DBU, which can be sourced from multiple vendors. This diversification of the supply base significantly reduces the risk of production stoppages due to raw material shortages, a critical consideration for supply chain heads managing long-term contracts.
- Cost Reduction in Manufacturing: The economic viability of this process is underscored by its high atom economy and the avoidance of expensive catalytic systems. By eliminating the need for precious metals such as palladium or rhodium, the process removes the substantial costs associated with catalyst procurement and, more importantly, the rigorous downstream processing required to remove trace metal residues to meet regulatory limits. Additionally, the high yields reported in the patent examples, such as the 95% yield in the initial condensation step, mean that less raw material is wasted per kilogram of product produced. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the marketplace without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain resilience. The reactions are performed at near-ambient temperatures (20-30 °C) and do not require specialized high-pressure or cryogenic equipment. This simplicity means that the process can be easily transferred between different manufacturing sites or scaled up from pilot to commercial scale with minimal re-engineering. For procurement managers, this flexibility ensures that production can be ramped up quickly to meet sudden increases in demand, such as those driven by successful clinical trial results. The use of common solvents like dichloromethane, THF, and ethyl acetate further simplifies logistics, as these are standard items in any chemical warehouse.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of the method through examples conducted on a multi-kilogram scale, starting with over 2 kg of raw material. This proof of concept at a meaningful scale de-risks the technology transfer to commercial manufacturing units. From an environmental standpoint, the process generates waste streams that are relatively straightforward to treat compared to heavy metal-laden effluents. The absence of toxic metals simplifies wastewater treatment protocols and reduces the environmental compliance burden. This aligns with the growing industry demand for greener chemistry practices, potentially qualifying the manufacturing site for various sustainability incentives and improving the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for their specific applications. The following questions address common inquiries regarding the practical implementation and capabilities of this technology. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this synthetic method for histidine derivatives?
A: The method described in patent CN108707115B offers a simple operation process with high yields suitable for large-scale production. It utilizes readily available starting materials and avoids complex purification steps often associated with peptide modifications.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the patent explicitly demonstrates scalability through examples conducted on a multi-kilogram scale (e.g., starting with 2200g of raw material). The use of standard organic solvents and common reagents facilitates easy technology transfer to commercial plants.
Q: What specific reagents are critical for the elimination step?
A: The elimination step critically relies on strong organic bases such as 1,8-diazabicycloundec-7-ene (DBU), triethylamine, or DIPEA. DBU is particularly effective in promoting the elimination of the mesylate group to form the desired acrylate double bond without side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-(2-(tert-butyloxycarbonylamino)-3-(1-trityl-1H-imidazole-4-yl)propionamide)acrylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of life-saving peptide therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the safe handling of reagents like MsCl and DBU, and we maintain stringent purity specifications through our rigorous QC labs. We are committed to delivering (S)-2-(2-(tert-butyloxycarbonylamino)-3-(1-trityl-1H-imidazole-4-yl)propionamide)acrylic acid with the consistency and reliability that global pharmaceutical leaders demand.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in turning complex chemical challenges into commercial successes, ensuring a secure and cost-effective supply of this critical histidine-based building block for your future drug candidates.
