Advanced Manufacturing of Lesinurad Intermediates via Safe Bromination and One-Pot Substitution
Introduction to Next-Generation Lesinurad Synthesis
The global demand for effective gout treatments continues to rise, driving the need for robust and scalable supply chains for key active pharmaceutical ingredients (APIs) like Lesinurad. Patent CN108947919B introduces a groundbreaking preparation method for Lesinurad and its critical intermediates, addressing significant bottlenecks found in legacy manufacturing processes. This technology represents a paradigm shift in how uric acid transporter inhibitors are produced, moving away from hazardous reagents toward a more sustainable and efficient chemical architecture. For R&D directors and procurement specialists, understanding this novel pathway is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent quality and safety standards. The invention not only streamlines the synthetic route but also fundamentally alters the impurity profile, ensuring higher purity specifications are met with greater consistency.
At the core of this innovation is the development of a new intermediate structure that facilitates a smoother transition to the final drug substance. By re-engineering the halogenation and substitution steps, the patent holders have created a process that is inherently safer and more economically viable. This report analyzes the technical merits of this approach, highlighting how it resolves the toxicity issues associated with thiophosgene and carbon disulfide while simultaneously boosting overall reaction yields. For stakeholders in the fine chemical industry, adopting such advanced methodologies is key to maintaining competitiveness in the generic and branded pharmaceutical markets.
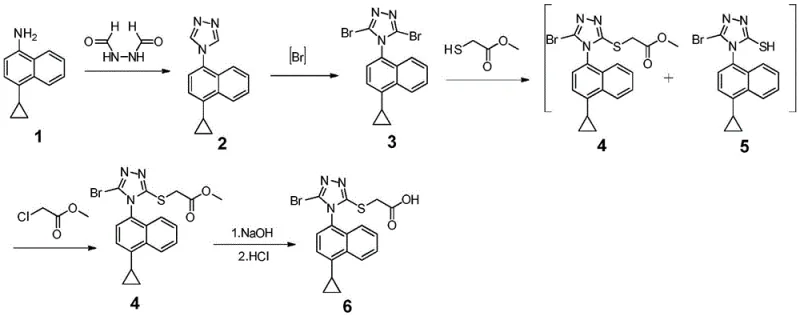
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lesinurad has been plagued by reliance on extremely hazardous reagents that complicate both laboratory handling and industrial scale-up. Prior art, such as the routes disclosed in WO2006026356 and WO2014008295, heavily depends on thiophosgene, a potent lachrymator and pulmonary toxin that requires specialized containment infrastructure. Furthermore, alternative routes reported in Chinese patents like CN103524440 utilize carbon disulfide, a neurotoxin with significant environmental persistence. These legacy methods not only endanger operator health but also impose heavy costs related to waste disposal and regulatory compliance. Additionally, many of these conventional pathways involve lengthy reaction sequences with multiple isolation steps, leading to cumulative yield losses that drive up the cost of goods sold (COGS).
Beyond safety concerns, the purification challenges in traditional synthesis are substantial. Older methods often generate complex mixtures of isomers that are difficult to separate without resorting to column chromatography, a technique that is notoriously inefficient and costly at a commercial scale. The reliance on expensive starting materials and the generation of toxic byproducts create a fragile supply chain vulnerable to regulatory shutdowns and raw material shortages. For a procurement manager, these factors translate into higher prices and increased risk of supply disruption, making the search for alternative manufacturing technologies a strategic priority.
The Novel Approach
The methodology outlined in CN108947919B offers a decisive break from these constraints by introducing a synthetic route that completely bypasses the use of thiophosgene and carbon disulfide. Instead, it employs safer brominating agents such as N-bromosuccinimide (NBS) and utilizes a clever one-pot substitution strategy to maximize yield. This novel approach allows for the direct conversion of reaction mixtures containing byproducts into the desired intermediate, effectively turning what was once waste into value. By eliminating the need for high-toxicity reagents, the process significantly lowers the barrier to entry for manufacturing facilities, allowing for production in standard chemical plants without the need for exotic containment systems.
Moreover, the new route simplifies the purification workflow by leveraging crystallization and slurry techniques rather than chromatography. This shift is critical for cost reduction in pharmaceutical manufacturing, as it reduces solvent consumption and processing time. The ability to perform consecutive reactions in a single vessel (telescoping) minimizes material transfer losses and exposure to the environment. For supply chain heads, this translates to a more robust and continuous production flow, reducing lead times and ensuring a steady availability of high-purity pharmaceutical intermediates. The strategic advantage lies in the process's inherent simplicity and safety, which aligns perfectly with modern green chemistry principles.
Mechanistic Insights into Bromination and Substitution Dynamics
The chemical elegance of this new process lies in its precise control over halogenation and nucleophilic substitution. The synthesis begins with the formation of a triazole ring on the naphthalene scaffold, followed by a critical dibromination step. Unlike indiscriminate halogenation methods that can lead to poly-brominated impurities, the use of NBS in tetrahydrofuran (THF) allows for selective introduction of bromine atoms at the 3 and 5 positions of the triazole ring. This selectivity is paramount for ensuring the downstream reactivity of the intermediate. The resulting dibromo-compound serves as a highly reactive electrophile, primed for subsequent functionalization with sulfur-containing nucleophiles.
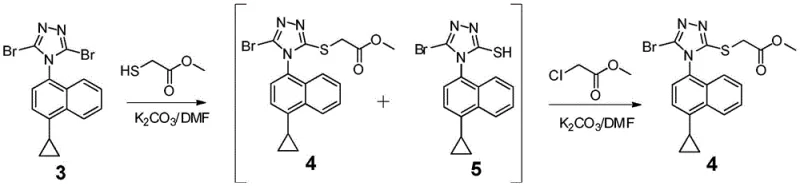
The most innovative aspect of the mechanism involves the handling of the substitution byproducts. When the dibromo-intermediate reacts with methyl thioglycolate, a mixture of isomers is inevitably formed: the desired 3-substituted product (Compound III) and the undesired 5-substituted isomer (Compound IV). In conventional chemistry, separating these isomers is arduous due to their similar polarities. However, this patent reveals a mechanistic workaround where the addition of an alkylating agent (R3X) in the presence of base converts the thiol byproduct (IV) back into the desired thioether structure (III). This dynamic equilibrium manipulation ensures that nearly all starting material is funneled into the target molecule, drastically improving atom economy.
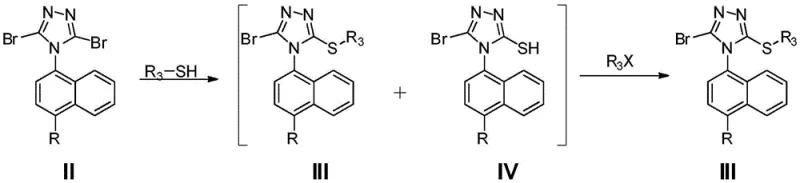
Furthermore, the final hydrolysis step is optimized to proceed under mild alkaline conditions, preserving the integrity of the sensitive triazole ring while efficiently cleaving the ester moiety to reveal the carboxylic acid functionality of Lesinurad. The use of sodium hydroxide in THF/water mixtures provides a homogeneous reaction environment that facilitates rapid conversion. Understanding these mechanistic nuances is vital for R&D teams aiming to replicate or license this technology, as slight deviations in pH or temperature could disrupt the delicate balance of the one-pot substitution. The robustness of this chemistry underpins the high purity levels achievable, meeting the rigorous specifications required for API production.
How to Synthesize 4-(4-cyclopropylnaphthalene)-3-thioacetic acid-5-bromo-1,2,4-triazole Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and order of addition, particularly during the telescoped substitution phase. The process is designed to be operationally simple, minimizing the need for intermediate isolation which often acts as a bottleneck in batch processing. By following the standardized protocol derived from the patent examples, manufacturers can achieve consistent results with minimal batch-to-batch variation. The detailed标准化 synthesis steps见下方的指南 provide a clear roadmap for translating this laboratory-scale success into pilot and commercial production environments.
- Cyclize 4-cyclopropyl-1-naphthylamine with N,N-diformylhydrazide using TMSCl and pyridine to form the triazole ring.
- Perform selective dibromination of the triazole intermediate using N-bromosuccinimide (NBS) in THF.
- Execute a one-pot substitution reaction with methyl thioglycolate followed by alkylation with methyl chloroacetate to convert byproducts.
- Hydrolyze the ester group using sodium hydroxide to yield the final Lesinurad acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage is the drastic simplification of the supply chain for raw materials. By removing thiophosgene and carbon disulfide from the bill of materials, companies eliminate the need for specialized hazardous material logistics and storage, which are often subject to strict regulatory scrutiny and high insurance premiums. This shift not only reduces direct material costs but also mitigates the risk of supply interruptions caused by regulatory bans on toxic precursors. The reliance on commodity chemicals like NBS and methyl chloroacetate ensures a stable and competitive pricing structure for the long term.
Enhanced supply chain reliability is another critical outcome of this technology. The one-pot nature of the key substitution step significantly shortens the manufacturing cycle time. In traditional multi-step syntheses, each isolation and drying stage adds days to the production schedule. By telescoping these reactions, the overall throughput of the manufacturing facility is increased, allowing for faster response to market demand fluctuations. This agility is invaluable in the pharmaceutical sector, where speed to market can determine the commercial success of a generic launch. Furthermore, the high conversion rates mean less raw material is wasted, contributing to substantial cost savings in manufacturing without compromising on quality.
Scalability and environmental compliance are also markedly improved. The avoidance of column chromatography is a major win for industrial scale-up, as chromatographic separation is often the limiting factor in producing multi-kilogram quantities of intermediates. The new method relies on crystallization and filtration, unit operations that are easily scaled from grams to tons using standard stainless steel reactors. From an environmental perspective, the reduction in solvent usage and the elimination of toxic waste streams align with increasingly stringent global environmental regulations. This makes the process not only economically attractive but also socially responsible, enhancing the corporate sustainability profile of any organization adopting this reliable pharmaceutical intermediates supplier strategy.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Lesinurad synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios.
Q: Why is the new synthesis route for Lesinurad considered safer than conventional methods?
A: Conventional routes often utilize highly toxic reagents like thiophosgene and carbon disulfide, which pose severe neurotoxic and vascular risks. The novel method described in CN108947919B eliminates these hazardous chemicals entirely, replacing them with safer brominating agents like NBS and standard alkylating reagents, significantly improving operational safety and environmental compliance.
Q: How does the one-pot reaction strategy improve overall yield?
A: In traditional substitution reactions, side products (isomer IV) are often difficult to separate and are discarded, lowering yield. This patent describes a method where the reaction mixture containing both the desired product (III) and the byproduct (IV) is treated directly with an alkylating agent (R3X). This converts the byproduct IV into the desired product III without isolation, thereby maximizing atom economy and total recovery.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. It avoids complex purification steps like column chromatography, which are impractical on a multi-ton scale. By utilizing crystallization and slurry techniques for purification and enabling one-pot conversions, the method reduces processing time and equipment requirements, making it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lesinurad Intermediate Supplier
As the pharmaceutical landscape evolves, the need for partners who can navigate complex synthetic challenges becomes ever more critical. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing reactions like the bromination and one-pot substitution described in CN108947919B, ensuring that the transition from lab to plant is seamless. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Lesinurad intermediate meets the highest international standards for safety and efficacy.
We invite you to explore how our advanced manufacturing capabilities can drive value for your organization. By leveraging our expertise in cost reduction in pharmaceutical manufacturing, we can help you optimize your supply chain for better margins and reliability. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-purity pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
