Advanced Microwave Synthesis of Alkoxy Porphyrins for High-Purity Pharmaceutical Intermediates
Advanced Microwave Synthesis of Alkoxy Porphyrins for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the need for rapid, efficient, and high-yield synthetic routes, particularly for complex macrocycles like porphyrins. Patent CN101235037A introduces a groundbreaking methodology for the synthesis of alkoxy porphyrins, leveraging microwave-assisted organic synthesis (MAOS) to overcome the inherent limitations of conventional thermal heating. This technology represents a pivotal shift for a reliable alkoxy porphyrin supplier, enabling the production of high-purity intermediates essential for pharmaceutical and advanced material applications. By utilizing microwave irradiation, the process achieves reaction completion in merely minutes rather than the dozens of hours required by traditional oil or water baths, fundamentally altering the economic and operational feasibility of producing these valuable compounds. The structural versatility of the resulting alkoxy porphyrins, as depicted in the general formula below, allows for extensive customization to meet specific client requirements for solubility and electronic properties.
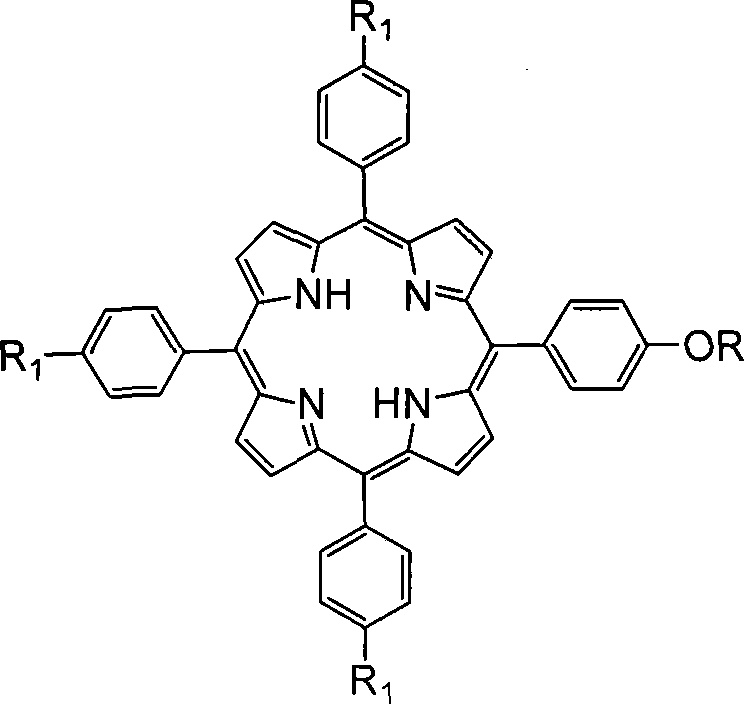
This innovation addresses the critical bottleneck of reaction time and thermal degradation that has historically plagued the commercial scale-up of complex porphyrins. The patent details a robust protocol where phenolic hydroxyl porphyrins react with halogenated alkanes in the presence of anhydrous potassium carbonate under controlled microwave power. This approach not only accelerates the kinetics of the Williamson ether synthesis but also ensures uniform heating throughout the reaction medium, thereby preventing the localized hot spots that often lead to substrate decomposition. For R&D directors and procurement managers alike, this translates to a more predictable supply chain and a product with a superior impurity profile, making it an ideal candidate for cost reduction in fine chemical intermediates manufacturing where efficiency is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of alkoxy porphyrins has long been hindered by the inefficiencies of conductive heat transfer methods such as oil baths and heating mantles. These conventional techniques rely on the slow propagation of heat from the vessel wall to the reaction center, often resulting in significant thermal gradients and uneven reaction rates. Such conditions frequently necessitate prolonged heating periods ranging from several hours to tens of hours to drive the nucleophilic substitution to completion, which exponentially increases energy consumption and operational costs. Furthermore, the extended exposure to high temperatures often induces thermal degradation of the sensitive porphyrin macrocycle, leading to the formation of complex impurity profiles that are difficult to separate. This thermal stress can compromise the integrity of the phenolic starting materials and the final ether product, resulting in lower overall yields and necessitating rigorous, multi-step purification processes that erode profit margins. For a supply chain head, these factors introduce substantial variability in lead times and batch-to-batch consistency, creating risks for downstream manufacturing schedules.
The Novel Approach
In stark contrast, the novel microwave-assisted approach described in the patent utilizes dielectric heating to energize molecules directly, bypassing the thermal inertia associated with conventional heating. This method allows for the precise control of reaction parameters, specifically power output between 100 to 500W and reaction times typically under 10 minutes, to achieve rapid conversion. The ability to complete the synthesis in such a short timeframe effectively eliminates the window for thermal decomposition, preserving the structural integrity of the porphyrin ring and its substituents. Consequently, the yield consistently reaches levels above 70%, significantly outperforming the yields obtained through traditional thermochemical reactions which often struggle to exceed lower benchmarks due to side reactions. This dramatic reduction in processing time not only enhances throughput capacity but also simplifies the workup procedure, as the crude product contains fewer degradation byproducts. For manufacturers seeking reducing lead time for high-purity porphyrins, this technology offers a direct pathway to faster turnaround and improved resource utilization.
Mechanistic Insights into Microwave-Promoted Williamson Ether Synthesis
The core chemical transformation driving this process is the Williamson ether synthesis, a nucleophilic substitution reaction where a phenoxide ion attacks a halogenated alkane. In the context of porphyrin chemistry, the phenolic hydroxyl groups on the meso-phenyl rings are deprotonated by the base, anhydrous potassium carbonate, to form the reactive phenoxide species. Under microwave irradiation, the polar solvent DMF (N,N-dimethylformamide) and the ionic intermediates absorb electromagnetic energy efficiently, leading to rapid molecular rotation and collision. This superheating effect lowers the activation energy barrier for the SN2 substitution, allowing the alkyl group to displace the halide leaving group with exceptional speed and selectivity. The specific interaction between the microwave field and the dipole moments of the reactants ensures that the energy is delivered uniformly, preventing the formation of localized hot spots that could otherwise trigger unwanted side reactions or polymerization of the sensitive macrocycle.
Furthermore, the choice of anhydrous conditions and calcined potassium carbonate is critical for maintaining the efficacy of the nucleophile and preventing hydrolysis of the halogenated alkane. The absence of water ensures that the phenoxide ion remains highly nucleophilic, driving the equilibrium towards the formation of the alkoxy porphyrin product. The patent specifies that the halogen atom can be either chlorine or bromine, with bromoalkanes generally exhibiting higher reactivity due to the better leaving group ability of the bromide ion. This mechanistic understanding allows for the fine-tuning of reaction conditions, such as adjusting the microwave power or reaction time, to accommodate different alkyl chain lengths ranging from ethyl to hexadecyl. By controlling these variables, manufacturers can optimize the process for specific derivatives, ensuring consistent quality and minimizing the formation of mono-alkylated or partially substituted impurities that could complicate downstream applications.
How to Synthesize Alkoxy Porphyrin Efficiently
The implementation of this microwave synthesis protocol requires careful attention to reagent preparation and equipment setup to ensure safety and reproducibility. The process begins with the dissolution of the phenolic porphyrin precursor and the chosen halogenated alkane in anhydrous DMF, followed by the addition of the base. Detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and microwave settings required to achieve optimal results across various substrates. Adhering to these parameters is essential for maximizing yield and minimizing the generation of waste, aligning with modern green chemistry principles.
- Dissolve phenolic hydroxyl porphyrin and halogenated alkane in anhydrous DMF within a glass reaction vessel equipped with reflux.
- Add calcined anhydrous potassium carbonate as the base and place the mixture into a microwave reactor.
- Irradiate at 100-500W for 3-90 minutes, monitor via TLC, then purify via column chromatography and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this microwave technology offers profound benefits that extend beyond mere technical superiority, directly impacting the bottom line and supply chain resilience. The drastic reduction in reaction time from hours to minutes translates into a significant increase in equipment utilization rates, allowing existing infrastructure to produce substantially more material without capital expansion. This efficiency gain is a key driver for cost reduction in fine chemical intermediates manufacturing, as it lowers the fixed cost allocation per kilogram of product. Moreover, the simplified purification profile resulting from reduced side reactions means less solvent consumption and shorter chromatography times, further driving down variable costs. For procurement managers, this implies a more stable pricing structure and the ability to secure high-quality materials at competitive rates.
- Cost Reduction in Manufacturing: The elimination of prolonged heating cycles drastically reduces energy consumption, which is a major operational expense in batch chemical processing. By completing reactions in under 10 minutes, the facility saves substantial amounts of electricity and cooling water compared to running oil baths for half a day. Additionally, the higher yield (>70%) means less raw material is wasted on failed batches or lost to degradation, optimizing the atom economy of the process. The reduction in solvent usage for purification, due to cleaner crude products, further contributes to overall cost savings and waste disposal reduction. These cumulative efficiencies create a leaner manufacturing model that is highly resistant to fluctuations in utility prices.
- Enhanced Supply Chain Reliability: The speed of the microwave process inherently reduces the manufacturing lead time, enabling faster response to market demands and urgent orders. Shorter cycle times mean that inventory turnover is accelerated, reducing the capital tied up in work-in-progress goods and finished stock. The robustness of the method, evidenced by consistent yields across a wide range of alkylating agents, ensures high batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts. This reliability mitigates the risk of production delays that can disrupt the customer's own manufacturing schedules, fostering stronger partnerships and trust. Consequently, the supply chain becomes more agile and capable of adapting to sudden changes in volume requirements.
- Scalability and Environmental Compliance: Microwave technology is increasingly scalable, with industrial continuous-flow reactors available to translate batch success to commercial tonnage. The process generates fewer byproducts and uses less solvent, aligning with stringent environmental regulations and sustainability goals. The use of DMF, while requiring careful handling, is a standard industrial solvent with established recovery protocols, minimizing environmental impact. The reduction in thermal degradation products simplifies waste treatment, lowering the burden on effluent processing facilities. This environmental compatibility ensures long-term operational viability and reduces the regulatory compliance burden on the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the microwave synthesis of alkoxy porphyrins, based on the detailed data provided in the patent literature. These insights are designed to clarify the operational benefits and technical feasibility for potential partners and stakeholders. Understanding these nuances is crucial for evaluating the integration of this technology into existing supply chains.
Q: What are the primary advantages of microwave synthesis over traditional thermal heating for porphyrins?
A: Microwave synthesis drastically reduces reaction time from several hours to under 10 minutes while minimizing local overheating and decomposition, resulting in yields exceeding 70%.
Q: Which halogenated alkanes are compatible with this alkoxy porphyrin synthesis method?
A: The process supports a wide range of halogenated alkanes including bromoethane, bromobutane, chlorocyclohexane, and long-chain variants like bromohexadecane, allowing for diverse functionalization.
Q: How does this method impact the purity profile of the final porphyrin product?
A: By avoiding prolonged thermal stress, the microwave method significantly reduces side reactions and degradation products, facilitating easier purification and higher final purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkoxy Porphyrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of microwave-assisted synthesis in delivering high-value intermediates for the global pharmaceutical and materials sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in the lab are fully realized at an industrial level. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of alkoxy porphyrin meets the exacting standards required for sensitive applications. Our capability to adapt advanced technologies like the one described in CN101235037A allows us to offer superior products that drive innovation for our clients.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your strategic partner in sourcing high-performance chemical intermediates.
