Revolutionizing Allyl Thiomethyl Ether Production: A Scalable AgOTf-Catalyzed Route for Global Supply Chains
Revolutionizing Allyl Thiomethyl Ether Production: A Scalable AgOTf-Catalyzed Route for Global Supply Chains
The landscape of organic synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN114656380B, which discloses a novel and simplified method for synthesizing allyl thiomethyl ether derivatives. This technology represents a paradigm shift from conventional thioether synthesis, utilizing a silver-catalyzed cross-dehydrogenative coupling (CDC) strategy that leverages dimethyl sulfoxide (DMSO) not merely as a solvent, but as a critical sulfur source. For R&D directors and procurement specialists in the pharmaceutical and fine chemical sectors, this innovation offers a compelling alternative to traditional routes that often rely on hazardous reagents and complex multi-step sequences. The ability to directly functionalize readily available olefins under relatively mild conditions opens new avenues for the rapid construction of valuable sulfur-containing scaffolds used extensively in drug discovery and material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl sulfides and thioethers has been fraught with significant chemical and operational challenges that hinder large-scale manufacturing efficiency. Traditional protocols frequently depend on the nucleophilic substitution of allylic halides with thiols or thiolates, a process that necessitates the use of stoichiometric strong bases and generates substantial amounts of salt waste. Furthermore, the reliance on alkyl halides introduces toxicity concerns and supply chain volatility, as these precursors are often regulated and expensive. Other methods involving the addition of thiols to alkenes under metal catalysis or strong acid conditions often suffer from poor regioselectivity, leading to complex mixtures of Markovnikov and anti-Markovnikov adducts that are difficult and costly to separate. Additionally, many existing cross-dehydrogenative coupling systems require harsh oxidants, high temperatures, or precious metal catalysts with limited turnover numbers, resulting in low atom economy and significant environmental burdens. These cumulative inefficiencies create bottlenecks in production timelines and inflate the cost of goods sold (COGS) for downstream API manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN114656380B introduces a streamlined catalytic cycle that dramatically simplifies the synthetic workflow. By employing silver trifluoromethanesulfonate (AgOTf) as a potent Lewis acid catalyst in conjunction with trifluoroacetic acid (TFA) as an additive, this method activates simple olefins for direct coupling with DMSO. This approach eliminates the need for pre-functionalized halide substrates and avoids the use of hazardous thiol reagents, thereby enhancing overall process safety. The reaction proceeds efficiently in DMSO itself, which serves a dual role as both the reaction medium and the methylthio source, maximizing atom economy. As illustrated in the general reaction scheme below, this transformation tolerates a wide array of functional groups, allowing for the direct conversion of various substituted styrenes into high-value allyl thiomethyl ethers with exceptional selectivity.
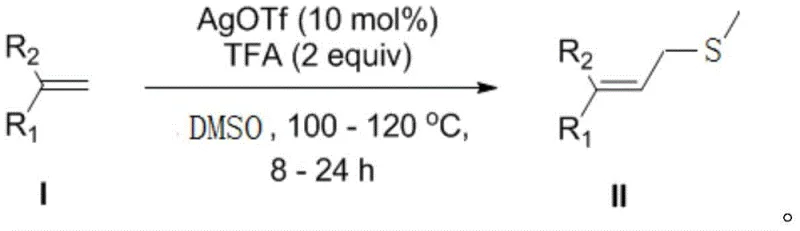
The operational simplicity of this novel route cannot be overstated; it typically requires heating the reaction mixture to moderate temperatures between 100°C and 120°C for 8 to 24 hours, followed by a straightforward aqueous workup and column chromatography. This robustness makes it highly attractive for industrial scale-up, where reproducibility and ease of handling are paramount. By bypassing the need for protecting group strategies often required in traditional thiol chemistry, this method reduces the total number of synthetic steps, directly translating to reduced labor costs and shorter lead times for pilot plant operations.
Mechanistic Insights into AgOTf-Catalyzed Cross-Dehydrogenative Coupling
From a mechanistic perspective, the efficacy of this transformation relies on the unique ability of the silver catalyst to coordinate with the olefinic double bond, thereby increasing its electrophilicity and facilitating nucleophilic attack or radical interception. The presence of TFA likely plays a crucial role in protonating the DMSO oxygen or stabilizing intermediate cationic species, lowering the activation energy for the C-S bond formation. Unlike traditional radical processes that might require peroxides or high-energy initiators, this silver-mediated pathway appears to operate through a controlled mechanism that preserves the stereochemistry of the double bond, predominantly yielding the trans-alkene products as observed in the experimental data. The catalytic cycle is remarkably efficient, utilizing only 10 mol% of the silver catalyst to drive the reaction to completion, which suggests a high turnover frequency and effective regeneration of the active catalytic species throughout the reaction duration.
Furthermore, the impurity profile of reactions conducted under these conditions is notably clean, which is a critical factor for pharmaceutical intermediate manufacturing. The high selectivity observed across diverse substrates, including those with electron-withdrawing groups like fluorine and chlorine, indicates that the mechanism is robust against electronic perturbations on the aromatic ring. This stability ensures that side reactions such as polymerization of the styrene substrate or over-oxidation of the sulfur moiety are minimized. For quality control teams, this means that the crude reaction mixtures contain fewer closely related impurities, simplifying the purification burden and ensuring that the final product meets stringent purity specifications (often exceeding 99% as reported in the patent examples) with minimal effort. The ability to achieve such high purity without recrystallization or complex distillation setups underscores the practical value of this chemistry for GMP environments.
How to Synthesize Allyl Thiomethyl Ether Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to ensure optimal yield and safety. The protocol is designed to be user-friendly, utilizing common laboratory equipment and reagents that are readily accessible in most chemical inventories. The following guide outlines the standardized procedure derived from the patent data, providing a clear roadmap for chemists looking to replicate this high-efficiency transformation. It is essential to maintain the specified molar ratios and temperature ranges to avoid decomposition of the sensitive allylic products or deactivation of the silver catalyst.
- Dissolve the olefin substrate (0.5 mmol scale) in DMSO (3-4 mL) within a reaction vessel to serve as both solvent and sulfur source.
- Add Silver Trifluoromethanesulfonate (AgOTf) at 10 mol% loading and Trifluoroacetic Acid (TFA) at 2.0 equivalents relative to the olefin.
- Heat the mixture in an oil bath at 100-120°C for 8-24 hours, then cool, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this AgOTf-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and regulated allylic halides and volatile thiols with commodity chemicals like styrenes and DMSO, companies can significantly mitigate supply chain risks associated with specialized reagent shortages. DMSO is a bulk solvent produced on a massive global scale, ensuring consistent availability and price stability, while substituted styrenes are widely manufactured feedstocks for the polymer industry, making them inexpensive and easy to source in multi-ton quantities. This shift to commoditized inputs directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering the baseline material costs and reducing the complexity of vendor management.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of stoichiometric waste and the reduction of unit operations. Traditional thioether synthesis often generates equimolar amounts of salt waste from base neutralization, which incurs disposal costs and environmental fees. In contrast, this catalytic method produces minimal inorganic waste, primarily consisting of the silver catalyst which can potentially be recovered, and benign organic byproducts. Furthermore, the omission of protection and deprotection steps shortens the overall production cycle, reducing energy consumption and labor hours per kilogram of product. While specific percentage savings depend on the specific substrate, the qualitative reduction in processing time and waste treatment overhead translates to substantial margin improvements for high-volume production runs.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions and the stability of the reagents. The process operates at atmospheric pressure without the need for specialized high-pressure reactors or inert gas manifolds beyond standard nitrogen blanketing, making it adaptable to existing multipurpose reactor trains. The tolerance for various functional groups means that a single platform technology can be used to manufacture a diverse library of analogues, allowing for flexible production scheduling in response to market demand. This versatility reduces the need for dedicated production lines for specific sulfur-containing intermediates, thereby optimizing asset utilization and ensuring continuous supply even during fluctuations in demand for specific API precursors.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is facilitated by the homogeneous nature of the reaction mixture and the absence of exothermic hazards typically associated with thiol additions. The use of DMSO, a high-boiling polar aprotic solvent, allows for safe heating without significant vapor pressure buildup, enhancing operational safety. From an environmental compliance standpoint, the process aligns with green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product). The simplified workup involving aqueous extraction and standard chromatography minimizes the volume of organic solvents required for purification, further reducing the facility's solvent recovery load and carbon footprint, which is increasingly critical for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the scope and limitations of the method. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this AgOTf-catalyzed method over traditional thiol alkylation?
A: Unlike traditional methods requiring hazardous alkyl halides and strong bases, this protocol utilizes DMSO as a safe methylthio source and simple olefins, significantly reducing waste and operational hazards while maintaining high yields.
Q: Does this synthesis tolerate electron-withdrawing groups on the styrene substrate?
A: Yes, the method demonstrates excellent functional group tolerance, successfully converting both electron-donating and electron-withdrawing substituted styrenes, including fluoro, chloro, and bromo derivatives, into the corresponding thioethers.
Q: Is the purification process complex for these allyl thiomethyl ether derivatives?
A: No, the process is designed for simplicity. The crude products are easily purified using standard silica gel column chromatography with ethyl acetate and petroleum ether, yielding high-purity materials suitable for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Thiomethyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the AgOTf-mediated synthesis of allyl thiomethyl ethers. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative academic and patent literature into robust, commercial-scale processes. Our team is equipped to handle the nuances of silver catalysis and sulfur chemistry, ensuring that the transition from lab-scale optimization to pilot and commercial production is seamless. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Whether you require custom synthesis of specific allyl thiomethyl ether derivatives or process development services to optimize an existing workflow, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can enhance your supply chain resilience and reduce your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
