Advanced Nickel Catalysis for Efficient Isoquinoline-1,3-Dione Production and Commercial Scale-Up
Advanced Nickel Catalysis for Efficient Isoquinoline-1,3-Dione Production and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies to introduce fluorine-containing motifs into heterocyclic scaffolds, driven by the profound impact of fluorination on metabolic stability and bioavailability. Patent CN114832862B discloses a groundbreaking catalytic composition utilizing a nickel-based system for the preparation of isoquinoline-1,3-dione compounds, addressing critical limitations in current synthetic routes. This technology leverages a specific molar ratio of nickel catalyst, nitrogen/phosphine ligands, and halide cocatalysts to achieve efficient difluoroalkylation under remarkably mild conditions. By shifting away from precious metal dependency, this innovation offers a sustainable pathway for generating high-value nitrogen-containing heterocycles essential for modern drug discovery pipelines. The strategic integration of inexpensive transition metals not only lowers the barrier to entry for complex molecule synthesis but also aligns with green chemistry principles by minimizing toxic waste generation.
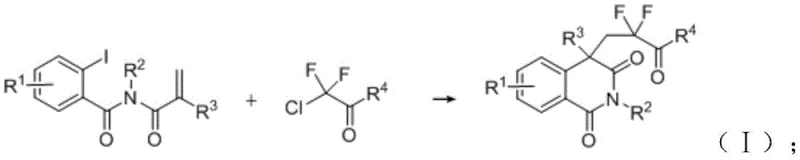
The introduction of fluorinated groups into the isoquinoline-1,3-dione core is pivotal for enhancing the lipophilicity and membrane permeability of potential therapeutic agents. Traditional approaches often struggle with harsh reaction parameters or limited substrate scope, creating bottlenecks in the rapid iteration of lead compounds. This patented methodology overcomes these hurdles by employing a dual-ligand system comprising 2,2'-bipyridyl derivatives and diphosphines, which stabilizes the active nickel species throughout the catalytic cycle. The result is a highly selective transformation that tolerates diverse functional groups, enabling medicinal chemists to explore broader chemical space without compromising yield. For organizations aiming to secure a reliable isoquinoline-1,3-dione supplier, adopting such advanced catalytic protocols ensures access to high-purity intermediates necessary for preclinical and clinical development stages.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in the synthesis of fluorinated isoquinoline-1,3-diones has predominantly relied on visible-light photocatalysis or palladium-catalyzed systems, both of which present significant operational challenges for large-scale manufacturing. Photocatalytic methods, while innovative, often require specialized equipment and suffer from scalability issues due to light penetration limits in bulk reactors, leading to inconsistent reaction rates and prolonged processing times. Similarly, palladium-catalyzed difluoroalkylation, though effective, incurs substantial costs associated with the precious metal catalyst and necessitates rigorous purification steps to remove trace metal residues that could compromise drug safety profiles. Furthermore, these conventional techniques frequently demand strict anhydrous conditions or elevated temperatures that can degrade sensitive substrates, resulting in lower overall yields and increased formation of difficult-to-separate impurities. Such inefficiencies directly impact the cost reduction in pharmaceutical intermediate manufacturing, making the final API prohibitively expensive for widespread therapeutic application.
The Novel Approach
In stark contrast, the novel nickel-catalyzed approach described in the patent utilizes earth-abundant metals to drive the coupling reaction with exceptional efficiency and economic viability. By operating at moderate temperatures around 60°C in polar aprotic solvents like DMSO, this method eliminates the need for energy-intensive heating or complex photochemical setups. The inclusion of magnesium chloride as a cocatalyst plays a synergistic role, activating the nickel center to facilitate oxidative addition and migratory insertion steps more effectively than previous systems. This optimization allows for the use of simple, commercially available reagents such as manganese powder as the terminal reductant, further simplifying the supply chain logistics. Consequently, this streamlined process not only accelerates the timeline for producing complex fluorinated scaffolds but also significantly reduces the environmental footprint associated with heavy metal waste disposal, offering a compelling advantage for sustainable chemical production.
Mechanistic Insights into Nickel-Catalyzed Difluoroalkylation
The catalytic cycle initiates with the formation of an active low-valent nickel species through the reduction of the nickel precursor by manganese in the presence of the bidentate ligand system. This active complex undergoes oxidative addition with the o-iodoacryloyl benzamide substrate, cleaving the carbon-iodine bond to generate a key organonickel intermediate. Subsequently, the difluoroalkyl radical or anion, generated from the difluoro compound, engages in a migratory insertion or radical capture event, constructing the new carbon-carbon bond with high regioselectivity. The unique electronic properties of the nickel center, modulated by the phosphine and nitrogen ligands, prevent premature beta-hydride elimination or other decomposition pathways that typically plague base-metal catalysis. This precise control over the reaction trajectory ensures that the fluorinated side chain is installed cleanly, preserving the integrity of the sensitive amide and olefin functionalities within the molecule.
Following the bond-forming event, the intermediate undergoes intramolecular cyclization to close the isoquinoline-1,3-dione ring, driven by the nucleophilic attack of the amide nitrogen onto the activated carbonyl or alkene moiety. The final step involves reductive elimination or protonolysis to release the desired product and regenerate the nickel catalyst, completing the turnover cycle. The presence of the halide cocatalyst is critical in maintaining the solubility and stability of the nickel species, preventing the formation of inactive nickel black precipitates that would halt the reaction. Understanding these mechanistic nuances is vital for process chemists aiming to optimize reaction parameters for commercial scale-up of complex pharmaceutical intermediates. By fine-tuning the ligand-to-metal ratio and solvent polarity, manufacturers can maximize turnover numbers and minimize catalyst loading, thereby achieving superior process economics without sacrificing product quality.
How to Synthesize Fluorinated Isoquinoline-1,3-Dione Efficiently
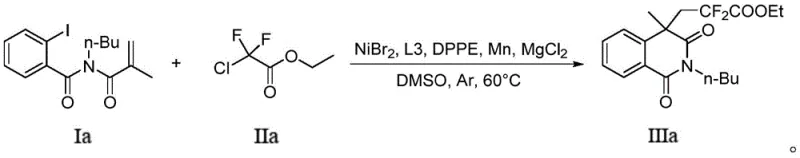
The practical implementation of this synthesis involves a straightforward one-pot procedure that combines all reagents in a single vessel under an inert atmosphere, minimizing handling errors and exposure to moisture. As demonstrated in the specific examples, the reaction proceeds smoothly in dimethyl sulfoxide, a solvent chosen for its ability to dissolve both organic substrates and inorganic salts effectively. The detailed standardized synthesis steps below outline the precise stoichiometry and workup procedures required to isolate the target compound with high purity, serving as a robust template for laboratory and pilot plant operations. Adhering to these protocols ensures reproducibility and safety, particularly when handling reactive metal powders and halogenated reagents on larger scales.
- Charge a Schlenk flask with nickel catalyst (NiBr2), ligands (L3 and DPPE), reducing agent (Mn), cocatalyst (MgCl2), o-iodoacryloyl benzamide substrate, and difluoro compound in DMSO solvent.
- Stir the reaction mixture under an inert argon atmosphere at 60°C for approximately 24 hours until TLC indicates complete consumption of the starting material.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, dry over sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this nickel-catalyzed protocol represents a strategic opportunity to optimize raw material costs and enhance supply security. The substitution of expensive palladium catalysts with inexpensive nickel bromide drastically reduces the direct material cost per kilogram of product, allowing for more competitive pricing in the global marketplace. Moreover, the reliance on commodity chemicals like manganese powder and magnesium chloride mitigates the risk of supply disruptions often associated with specialty catalysts sourced from limited geographic regions. This resilience is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients who require just-in-time inventory management.
- Cost Reduction in Manufacturing: The elimination of precious metals removes the necessity for costly scavenging resins or complex purification trains designed to meet stringent residual metal specifications. This simplification of the downstream processing workflow translates into significant operational savings, as fewer unit operations are required to bring the crude reaction mixture to specification. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further contributing to a lower overall cost of goods sold. By adopting this economically favorable route, manufacturers can offer substantial cost savings to their partners while maintaining healthy profit margins in a competitive industry landscape.
- Enhanced Supply Chain Reliability: The broad substrate scope of this methodology means that a single catalytic platform can be utilized to produce a diverse library of analogues, reducing the need for multiple specialized production lines. This flexibility allows suppliers to respond rapidly to changing market demands or clinical trial requirements without extensive retooling or process revalidation. Furthermore, the use of stable, shelf-stable reagents ensures that inventory can be held safely for extended periods, buffering against short-term market volatility. Such reliability is paramount for building long-term partnerships with multinational corporations that prioritize consistent quality and uninterrupted supply of critical building blocks.
- Scalability and Environmental Compliance: Scaling this reaction from gram to multi-ton quantities is facilitated by the absence of hazardous reagents and the use of standard stainless steel reactor equipment compatible with polar solvents. The simplified workup procedure, involving basic aqueous quenching and extraction, generates less hazardous waste compared to processes requiring strong acids or bases, easing the burden on wastewater treatment facilities. This alignment with environmental regulations reduces compliance costs and enhances the corporate sustainability profile, a factor increasingly weighted in vendor selection criteria by major pharmaceutical companies. The ability to execute this chemistry safely and cleanly at scale positions manufacturers as preferred partners for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this nickel-catalyzed technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational advantages and chemical versatility of the method for technical decision-makers evaluating potential process changes. Understanding these details helps in assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is nickel preferred over palladium for this difluoroalkylation reaction?
A: Nickel catalysts are significantly more cost-effective than palladium and exhibit higher oxidative addition activity due to lower electronegativity, allowing for milder reaction conditions and reduced side reactions.
Q: What is the specific role of magnesium chloride in this catalytic system?
A: Magnesium chloride acts as a crucial cocatalyst that enhances the activity of the nickel catalyst, facilitating the coupling process and improving overall yield without requiring expensive additives.
Q: Does this method support a broad range of substrate substituents?
A: Yes, the protocol demonstrates excellent tolerance for various substituents on the benzene ring, including halogens and alkyl groups, making it versatile for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline-1,3-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology in accelerating the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory bench to full-scale manufacturing. We are committed to delivering high-purity isoquinoline-1,3-dione derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging our deep expertise in transition metal catalysis and continuous flow processing, we can further optimize this patented route to maximize yield and minimize impurity profiles, providing you with a distinct competitive edge in the marketplace.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate specific COA data for a new analogue, our specialists are ready to provide comprehensive route feasibility assessments. Partnering with us ensures not only access to cutting-edge chemical technology but also a dedicated support system focused on your long-term success. Contact us today to request a quote and discover how we can drive efficiency and value into your pharmaceutical intermediate sourcing strategy.
