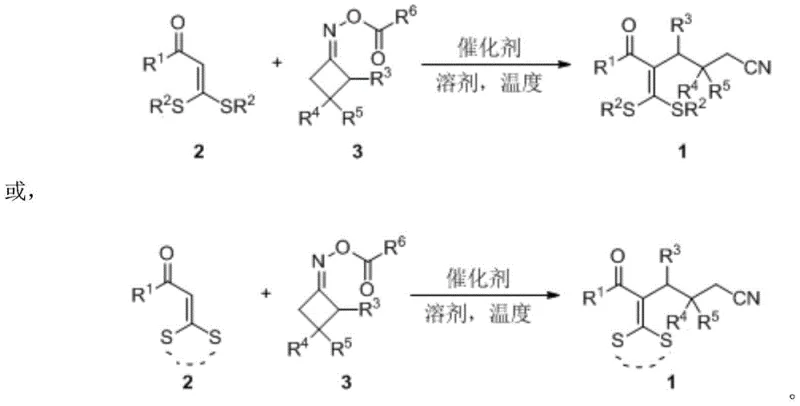
Advanced Iron-Catalyzed Synthesis of Cyanoalkyl Tetra-Substituted Olefins for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular scaffolds efficiently. Patent CN111253293B introduces a groundbreaking approach for synthesizing cyanoalkyl-substituted tetra-substituted olefin derivatives, which serve as critical building blocks in drug discovery and material science. This technology leverages an intermolecular free radical addition reaction catalyzed by inexpensive iron salts, marking a significant departure from traditional precious metal-catalyzed processes. By utilizing 3,3-dialkylthio-2-propen-1-one and cyclobutanone oxime esters as key synthons, the method achieves high atom economy and operational simplicity. For R&D directors and procurement specialists, this patent represents a viable pathway to access high-purity pharmaceutical intermediates with reduced environmental impact and lower raw material costs. The ability to generate structural diversity through simple substituent regulation makes this technology particularly attractive for developing new active pharmaceutical ingredients (APIs) and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted olefins has relied heavily on transition metal catalysis involving palladium, rhodium, ruthenium, or iridium complexes. These conventional methods typically necessitate rigorous pre-functionalization of starting materials, such as halogenation, silicidation, or boronation, to activate carbon-carbon bond formation. Such requirements not only increase the number of synthetic steps but also generate substantial chemical waste, thereby inflating the overall production cost and environmental footprint. Furthermore, the reliance on precious metals introduces significant supply chain vulnerabilities, as the prices of these catalysts are subject to volatile market fluctuations and geopolitical constraints. From a safety perspective, some traditional protocols require harsh reaction conditions or toxic reagents that complicate industrial scale-up and necessitate expensive containment and waste treatment infrastructure. These factors collectively hinder the economic feasibility of producing complex olefin derivatives on a commercial scale, creating a pressing need for more sustainable and cost-effective alternatives.
The Novel Approach
The methodology disclosed in patent CN111253293B overcomes these historical bottlenecks by employing a direct radical addition strategy mediated by abundant iron salts. This novel approach eliminates the need for extensive pre-functionalization, allowing for the direct coupling of readily available ketones and oxime esters in a single step. The use of ferric chloride as a catalyst not only drastically reduces raw material costs but also aligns with green chemistry principles by minimizing heavy metal contamination in the final product. The reaction proceeds under mild thermal conditions, typically between 25°C and 120°C, which enhances energy efficiency and operational safety within the manufacturing plant. By streamlining the synthetic route, this technology significantly shortens the production timeline and reduces the consumption of solvents and reagents. This efficiency translates directly into improved margins for manufacturers and more reliable supply chains for downstream clients seeking reliable pharmaceutical intermediate suppliers.
Mechanistic Insights into FeCl3-Catalyzed Radical Addition
The core innovation of this technology lies in the generation of iminyl radicals from cyclobutanone oxime esters under the influence of the iron catalyst. Upon heating in a polar aprotic solvent such as trifluorotoluene, the iron species facilitates the homolytic cleavage of the N-O bond in the oxime ester, releasing a cyanoalkyl radical species. This highly reactive intermediate then undergoes regioselective addition to the electron-deficient double bond of the 3,3-dialkylthio-2-propen-1-one substrate. The resulting carbon-centered radical is subsequently stabilized and quenched to form the final tetra-substituted olefin skeleton with high stereoselectivity. This mechanism avoids the high-energy barriers associated with oxidative addition and reductive elimination steps common in palladium cycles, thereby allowing the reaction to proceed smoothly at moderate temperatures. Understanding this mechanistic pathway is crucial for process chemists aiming to optimize reaction parameters for specific substrates and ensure consistent batch-to-batch quality.
Impurity control is inherently superior in this radical-mediated process due to the high chemoselectivity of the iron catalyst towards the specific N-O bond activation. Unlike broad-spectrum transition metal catalysts that might promote side reactions such as homocoupling or over-reduction, the iron system demonstrates remarkable tolerance for various functional groups including carbonyls, sulfides, and nitriles. This selectivity minimizes the formation of difficult-to-remove byproducts, simplifying the downstream purification process significantly. The resulting products exhibit excellent purity profiles, often requiring only standard silica gel chromatography to achieve pharmaceutical-grade specifications. For quality assurance teams, this means reduced analytical burden and higher confidence in the consistency of the supplied intermediates. The robust nature of the reaction mechanism ensures that even with slight variations in raw material quality, the process remains stable and reproducible, which is a key requirement for GMP-compliant manufacturing environments.
How to Synthesize Cyanoalkyl Substituted Tetra-Substituted Olefin Efficiently
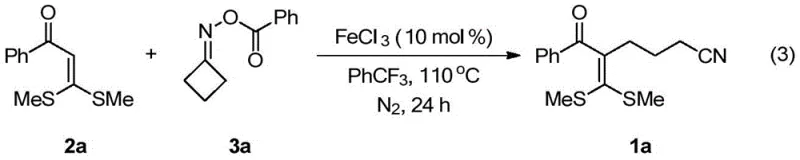
To implement this synthesis effectively, operators must adhere to strict anhydrous and anaerobic conditions to prevent catalyst deactivation and radical quenching by oxygen or moisture. The standard protocol involves charging a reactor with the ketone synthon and oxime ester in a molar ratio that favors complete conversion of the limiting reagent, typically using a slight excess of the oxime component. Following the addition of the iron catalyst, the mixture is heated to the optimal temperature range of 110°C for a duration of 10 to 48 hours, depending on the specific steric hindrance of the substrates. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 3,3-dialkylthio-2-propen-1-one and cyclobutanone oxime ester in trifluorotoluene solvent under argon atmosphere.
- Add ferric chloride (FeCl3) catalyst at 10 mol% concentration and heat the mixture to 110°C for approximately 24 hours to facilitate radical addition.
- Purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate to isolate the target cyanoalkyl-substituted olefin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed technology offers transformative benefits regarding cost stability and operational reliability. The shift from precious metal catalysts to commodity iron salts removes a major variable cost driver, insulating the production budget from the volatility of the platinum group metals market. Furthermore, the simplified workflow reduces the demand for specialized labor and complex equipment, allowing for more flexible manufacturing scheduling and faster turnaround times. The mild reaction conditions also lower energy consumption rates, contributing to a smaller carbon footprint and aligning with corporate sustainability goals. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts results in substantial cost savings on raw materials, while the one-step nature of the reaction reduces labor and utility expenses associated with multi-step sequences. The high atom economy ensures that a greater proportion of input materials are converted into valuable product, minimizing waste disposal costs. Additionally, the use of common solvents like trifluorotoluene allows for efficient recovery and recycling, further driving down the cost of goods sold. These economic efficiencies enable competitive pricing strategies for high-purity pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Sourcing iron salts and basic organic building blocks is significantly more stable than relying on specialized organometallic complexes that may face supply shortages. The robustness of the reaction against minor variations in feedstock quality ensures consistent output, reducing the risk of batch failures and production delays. This reliability is critical for maintaining continuous supply to downstream API manufacturers who operate on tight just-in-time schedules. By diversifying the supplier base for catalysts and reagents, companies can mitigate risks associated with single-source dependencies and geopolitical trade disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable from gram-scale laboratory experiments to multi-ton industrial production without requiring fundamental changes to the reaction engineering. The absence of toxic heavy metals simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste management. This ease of compliance accelerates the approval process for new manufacturing sites and facilitates faster market entry for new products. Moreover, the energy-efficient operating temperatures contribute to lower greenhouse gas emissions, supporting global initiatives for greener chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production portfolios.
Q: What are the primary advantages of using iron catalysis over traditional transition metals for this synthesis?
A: Iron catalysis offers substantial cost advantages due to the abundance and low price of iron salts compared to precious metals like palladium or rhodium. Additionally, iron is less toxic and environmentally hazardous, simplifying waste treatment and regulatory compliance for large-scale manufacturing.
Q: How does this method improve the functional group tolerance for complex molecule synthesis?
A: The mild reaction conditions (25-120°C) and the specific radical mechanism allow for excellent compatibility with diverse functional groups such as carbonyls, alkylthio, and cyano groups. This versatility enables chemists to synthesize complex structures without extensive protecting group strategies.
Q: Is this synthetic route suitable for industrial scale-up and continuous production?
A: Yes, the process utilizes readily available raw materials and operates under relatively mild thermal conditions without requiring high-pressure equipment. The simple workup procedure involving standard column chromatography or crystallization supports scalable manufacturing from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoalkyl Substituted Tetra-Substituted Olefin Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this innovative patent into commercial reality for our global clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of cyanoalkyl substituted olefin meets the highest industry standards for pharmaceutical applications. Our commitment to quality and reliability makes us the preferred partner for companies seeking to secure their supply chain for critical fine chemical intermediates.
We invite you to collaborate with us to explore how this technology can optimize your specific manufacturing processes. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of partnering with us for your next project. Let us help you achieve your production goals with efficiency and confidence.
