Advanced Metal-Free Synthesis of Polycyclic 2-Hydropyrazoles for Commercial Scale-Up
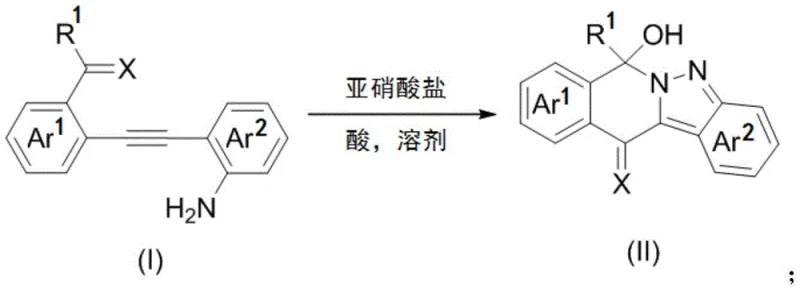
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds, particularly those containing nitrogen-rich motifs like pyrazoles. A significant breakthrough in this domain is detailed in Chinese Patent CN108276406B, which discloses a novel, environmentally benign synthesis method for polycyclic 2-hydropyrazole compounds. This technology represents a paradigm shift from traditional transition-metal-catalyzed processes to a more sustainable, nitrite-mediated cyclization strategy. By leveraging simple diarylalkyne precursors and inexpensive inorganic nitrites under mild acidic conditions, this invention offers a direct pathway to valuable bioactive intermediates. The process operates effectively within a temperature range of -20°C to 50°C, utilizing solvents that can include water, thereby aligning with green chemistry principles. For R&D directors and process chemists, this patent provides a critical alternative to legacy methods that often suffer from regioselectivity issues and harsh reaction requirements.
Historically, the construction of 2-hydropyrazole frameworks has relied heavily on methodologies such as Cadogan cyclization, C-H bond activation, or cycloaddition reactions involving diazo compounds and benzyne intermediates. While effective in specific contexts, these conventional approaches present substantial limitations for large-scale manufacturing. For instance, cycloaddition reactions often struggle with regioselectivity, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, Cadogan cyclizations and C-H activation protocols typically necessitate the use of expensive transition metal catalysts, which not only inflate raw material costs but also introduce the risk of heavy metal contamination in the final API. The removal of trace metals to meet stringent regulatory standards often requires additional purification steps, such as scavenging resins or repeated recrystallizations, which drastically reduce overall process efficiency and yield.
In stark contrast, the novel approach outlined in the patent data circumvents these bottlenecks by designing the dipole and alkyne functionalities into a single molecular precursor, facilitating an intramolecular cyclization that does not require external metal catalysis. This metal-free strategy utilizes aromatic amines and nitrites as the nitrogen source, triggering a cascade reaction that efficiently closes the pyrazole ring. The absence of transition metals simplifies the downstream processing significantly, as there is no need for specialized metal removal protocols. Moreover, the reaction conditions are remarkably mild and operationally simple, allowing for the direct precipitation of the product from the reaction mixture. This eliminates the need for labor-intensive and solvent-heavy column chromatography, a common requirement in laboratory-scale syntheses that is notoriously difficult to translate to industrial production. The result is a streamlined process that delivers high-purity polycyclic 2-hydropyrazoles with excellent yields, making it highly attractive for cost-sensitive commercial applications.
Mechanistic Insights into Nitrite-Mediated Intramolecular Cyclization
To fully appreciate the elegance of this synthetic route, one must delve into the mechanistic details of the nitrite-mediated transformation. The reaction initiates with the generation of a diazonium species in situ. Under acidic conditions, the primary amine moiety on the diarylalkyne substrate reacts with the nitrite ion to form a reactive diazonium intermediate. This electrophilic species is perfectly positioned to undergo an intramolecular attack on the proximal alkyne triple bond. The electron-rich pi-system of the alkyne acts as a nucleophile, attacking the terminal nitrogen of the diazonium group, which triggers a cyclization event. This step is crucial as it forms the new carbon-nitrogen bonds required to establish the pyrazole core. The resulting cyclic intermediate then undergoes further rearrangement and hydrolysis steps, ultimately leading to the stable polycyclic 2-hydropyrazole structure observed in the final product.
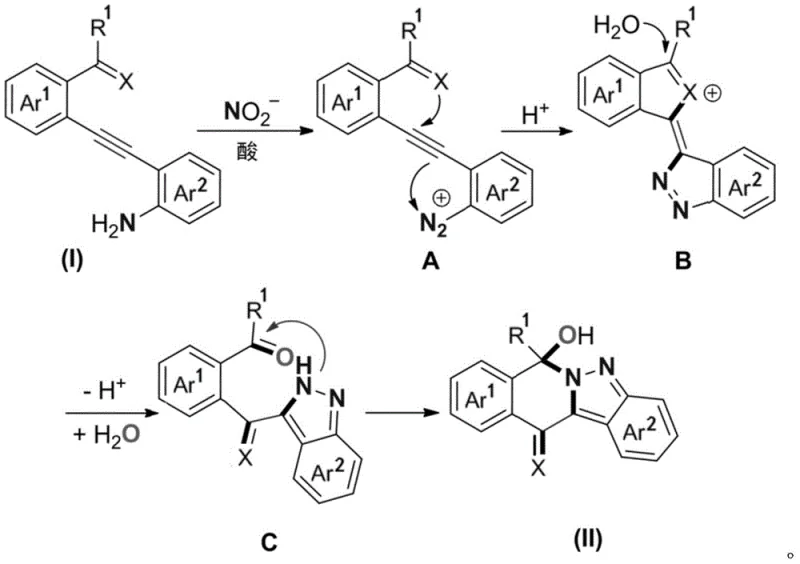
The control of impurities in this mechanism is inherently superior to metal-catalyzed alternatives. Since the reaction relies on well-defined acid-base chemistry and electrophilic aromatic substitution principles rather than complex organometallic cycles, the formation of side products associated with metal-ligand dissociation or beta-hydride elimination is virtually non-existent. The primary byproducts are typically inorganic salts derived from the acid and nitrite reagents, which are easily removed during the aqueous workup. Furthermore, the intramolecular nature of the cyclization ensures high regioselectivity, as the reacting centers are tethered within the same molecule, preventing intermolecular polymerization or oligomerization that often plagues intermolecular cycloadditions. This mechanistic clarity allows process chemists to predict and control the impurity profile with high confidence, ensuring that the final material meets the rigorous purity specifications required for pharmaceutical intermediates without extensive purification efforts.
How to Synthesize Polycyclic 2-Hydropyrazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting involves a straightforward sequence of operations that prioritizes safety and efficiency. The general protocol involves dissolving the diarylalkyne starting material in a solvent system that may comprise organic solvents like acetonitrile, DMF, or methanol, often mixed with water to facilitate the solubility of the inorganic nitrite salt. An acid, such as sulfuric acid, hydrochloric acid, or acetic acid, is added to generate the necessary acidic environment for diazotization. The key to success lies in the controlled addition of the nitrite salt, typically sodium nitrite or potassium nitrite, at temperatures maintained between -20°C and 50°C depending on the specific substrate reactivity. Once the addition is complete, the reaction mixture is stirred until conversion is confirmed, after which the product is induced to precipitate by the addition of water, filtered, and dried.
- Dissolve the diarylalkyne starting material in a suitable organic solvent or aqueous mixture and add the chosen acid catalyst.
- Slowly introduce the nitrite salt (such as sodium nitrite) at controlled temperatures ranging from -20°C to 50°C to initiate diazotization.
- Stir the reaction mixture until completion, then precipitate the product by adding water, followed by filtration and washing to obtain high-purity solids.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free synthesis route offers transformative advantages that directly impact the bottom line and operational resilience. The most significant benefit is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts. Traditional methods often rely on palladium, rhodium, or copper complexes, which are subject to volatile market pricing and supply constraints. By replacing these with commodity chemicals like sodium nitrite and common mineral acids, manufacturers can decouple their production costs from the fluctuations of the precious metals market. Additionally, the simplification of the purification process removes the need for expensive chromatography media and large volumes of organic solvents, leading to substantial savings in consumables and waste disposal costs. This leaner material profile enhances the overall economic viability of producing these complex intermediates at scale.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond simple reagent costs. By eliminating the need for transition metal catalysts, the process avoids the expensive and time-consuming steps required to scavenge residual metals from the final product, which is a critical quality attribute for pharmaceutical ingredients. Furthermore, the ability to isolate the product via simple filtration rather than column chromatography significantly reduces solvent consumption and labor hours. This streamlined workflow translates to a lower cost of goods sold (COGS) and improved margin potential for high-volume production runs. The use of water as a co-solvent or anti-solvent further reduces the reliance on costly anhydrous organic solvents, contributing to a more sustainable and cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount for continuous manufacturing operations, and this methodology strengthens reliability by utilizing widely available, commodity-grade reagents. Sodium nitrite, sulfuric acid, and common organic solvents are produced globally in massive quantities, ensuring a secure and consistent supply chain that is less prone to disruption compared to specialized ligands or custom-synthesized catalysts. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, also provides flexibility in sourcing. If a specific solvent becomes unavailable or prohibitively expensive due to logistics issues, the process can often be adapted to use alternative solvents without compromising yield or quality, thereby mitigating supply chain risks and ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden challenges, but this nitrite-mediated cyclization is inherently scalable due to its simplicity and safety profile. The reaction does not involve hazardous reagents like azides or highly unstable diazo compounds that require specialized handling equipment; instead, the diazonium species is generated and consumed in situ under controlled conditions. This reduces the safety risks associated with batch processing and facilitates easier technology transfer to larger reactors. From an environmental standpoint, the reduction in organic solvent usage and the absence of heavy metal waste streams simplify wastewater treatment and regulatory compliance. This aligns with increasingly strict environmental regulations and corporate sustainability goals, making the process not only commercially attractive but also environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method explicitly eliminates the need for transition metal catalysts such as palladium or rhodium, utilizing inexpensive nitrite salts instead, which significantly reduces raw material costs and simplifies metal removal processes.
Q: Is column chromatography necessary for purifying the final product?
A: Column chromatography is not required. The process is designed to allow the product to precipitate directly from the reaction mixture upon the addition of water, enabling purification through simple filtration and washing steps.
Q: What is the substrate scope for the aryl groups in this reaction?
A: The method demonstrates broad substrate applicability, accommodating various aryl and substituted aryl groups including phenyl, pyridyl, naphthyl, and benzofuranyl moieties, as well as substituents like alkyl, alkoxy, and halogens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polycyclic 2-Hydropyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the one described in CN108276406B for advancing drug discovery and development programs. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such complex, metal-free pathways from literature concepts into robust, commercial-scale realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of polycyclic 2-hydropyrazole intermediate meets the highest industry standards for identity, potency, and impurity profiles.
We invite you to collaborate with us to leverage this cost-effective and green synthesis technology for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to this metal-free route can optimize your budget. Please contact us today to request specific COA data for similar structures or to discuss route feasibility assessments for your target molecules. Let us help you secure a reliable, high-quality supply of these critical pharmaceutical intermediates while driving down your overall manufacturing costs.
