Advanced Asymmetric Synthesis of Difluoroalkyl Cyclopentenones for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies to incorporate fluorine atoms into organic scaffolds, driven by the unique ability of fluorine to enhance metabolic stability and oral bioavailability of drug candidates. Patent CN113501766B introduces a groundbreaking asymmetric synthesis method for generating polyfunctionalized cyclopentenone derivatives containing difluoroalkyl groups, addressing a significant gap in current organic synthesis literature. This innovation leverages a sophisticated dual-catalytic system combining visible light photocatalysis with chiral Bronsted acid catalysis to achieve precise stereocontrol. Unlike traditional methods that often struggle with regioselectivity or require extreme conditions, this approach operates under mild room temperature environments, utilizing readily available starting materials such as furan olefins, aromatic amines, and difluoroalkyl halides. The strategic integration of these components allows for the construction of complex molecular architectures with high efficiency, making it a highly valuable asset for the development of next-generation therapeutic agents.

Historically, the introduction of difluoroalkyl groups into the side chains of cyclopentenone frameworks has been a formidable challenge for synthetic chemists, often necessitating multi-step sequences with poor atom economy. Conventional thermal approaches frequently rely on stoichiometric amounts of hazardous reagents or expensive chiral transition metal complexes that are difficult to remove from the final product, posing risks for pharmaceutical applications. Furthermore, achieving high levels of enantioselectivity in such transformations typically requires cryogenic temperatures and inert atmospheres, which drastically increases energy consumption and operational complexity. These limitations have hindered the widespread adoption of difluoroalkylated cyclopentenones in medicinal chemistry, despite their potential utility. The reliance on harsh conditions also limits the functional group tolerance, preventing the synthesis of diverse analogues required for structure-activity relationship studies.
The novel approach detailed in the patent data overcomes these historical barriers through an elegant cascade sequence initiated by visible light. By employing a mer-Ir(ppy)3 photocatalyst under blue LED irradiation, the method generates reactive radical species under ambient conditions, facilitating a three-component coupling that constructs the core skeleton efficiently. This is followed by a stereoselective aza-Piancatelli rearrangement catalyzed by a chiral Bronsted acid and an achiral Lewis acid, specifically dysprosium triflate. This tandem strategy not only streamlines the synthetic route by reducing the number of isolation steps but also ensures exceptional stereochemical outcomes. The use of organocatalysts instead of heavy metals aligns with green chemistry principles, reducing the burden of metal residue testing and purification, which is a critical consideration for regulatory compliance in API manufacturing.
Mechanistic Insights into Visible Light Mediated Asymmetric Rearrangement
The mechanistic pathway of this transformation is a testament to modern catalytic design, beginning with the photoexcitation of the iridium catalyst which triggers a single-electron transfer process. This event activates the difluoroalkyl halide to generate a nucleophilic carbon-centered radical, which subsequently adds to the electron-rich furan olefin substrate. The resulting radical intermediate undergoes oxidation and trapping by the aromatic amine to form a key amino-furan intermediate. This species then serves as the precursor for the subsequent rearrangement step. The precision of this radical mediation ensures that side reactions such as homocoupling or polymerization are minimized, leading to clean reaction profiles that simplify downstream processing. The compatibility of the photocatalytic cycle with the subsequent acid catalysis is crucial, as it allows for a telescoped or sequential operation without the need for extensive intermediate workup.
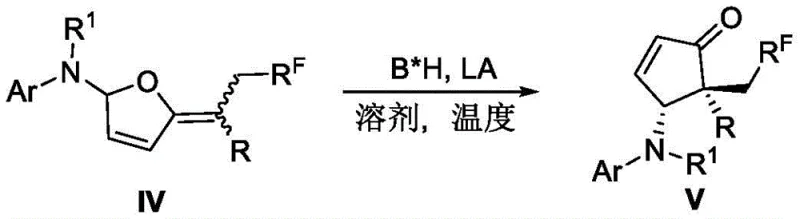
In the second stage of the mechanism, the chiral environment is established through the cooperative action of the chiral Bronsted acid and the Lewis acid activator. The Lewis acid coordinates with the oxygen atoms of the intermediate, increasing its electrophilicity and facilitating the ring-opening and reclosure events characteristic of the Piancatelli rearrangement. Simultaneously, the chiral Bronsted acid directs the facial selectivity of the nucleophilic attack, ensuring that the newly formed stereocenters possess the desired configuration. This dual activation mode is responsible for the high diastereoselectivity and enantiomeric excess observed across various substrates. The robustness of this catalytic system is evident in its tolerance to different electronic properties on the aromatic rings, allowing for the synthesis of a broad library of derivatives. Such mechanistic understanding provides confidence in the reproducibility of the process, a key factor for technology transfer from laboratory to pilot plant.
How to Synthesize Difluoroalkyl Cyclopentenones Efficiently
Executing this synthesis requires careful attention to the reaction parameters outlined in the patent to maximize yield and selectivity. The process begins with the preparation of the reaction mixture under an inert argon atmosphere to prevent quenching of the radical species by oxygen. The photocatalyst loading is kept minimal, typically around 1 mol%, which is economically favorable for large-scale operations. Following the light-mediated coupling, the reaction mixture is processed to remove the photocatalyst residues before introducing the chiral catalysts for the rearrangement step. The use of standard solvents like acetonitrile and chloroform ensures that the process remains cost-effective and easy to handle. Detailed standardized synthetic steps for replicating this high-value transformation are provided in the guide below.
- Perform a radical-mediated three-component difluoroalkyl amination reaction using furan olefins, aromatic amines, and difluoroalkyl halides under visible light irradiation with mer-Ir(ppy)3 photocatalyst.
- Subject the resulting intermediate to an asymmetric rearrangement reaction using a combination of chiral Bronsted acid and achiral Lewis acid (Dy(OTf)3) in chloroform at room temperature.
- Purify the final crude product via silica gel column chromatography to isolate the target difluoroalkyl-containing multifunctional cyclopentenone derivative with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic methodology offers substantial strategic benefits that extend beyond mere chemical novelty. The reliance on visible light and room temperature conditions translates directly into significant energy savings compared to processes requiring heating or cooling, contributing to a lower carbon footprint and reduced utility costs. Furthermore, the use of earth-abundant or easily recyclable catalysts minimizes the dependency on scarce precious metals, mitigating supply chain volatility associated with fluctuating metal prices. The high selectivity of the reaction reduces the formation of impurities, which simplifies purification protocols and decreases the consumption of chromatography media and solvents. These factors collectively contribute to a more sustainable and economically viable manufacturing process for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive chiral transition metal ligands and the use of low-loading photocatalysts significantly lower the raw material costs per kilogram of product. Additionally, the mild reaction conditions reduce the need for specialized high-pressure or cryogenic equipment, lowering capital expenditure requirements for production facilities. The high atom economy of the three-component coupling ensures that a greater proportion of starting materials are incorporated into the final product, minimizing waste disposal costs. These efficiencies allow for a more competitive pricing structure for the final active pharmaceutical ingredients derived from these intermediates.
- Enhanced Supply Chain Reliability: The starting materials, including furan olefins and difluoroalkyl halides, are commercially available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even with different batches of reagents. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream drug manufacturers. The scalability demonstrated in the patent data further assures partners that supply can be ramped up quickly to meet market demand without compromising quality.
- Scalability and Environmental Compliance: The process has been successfully demonstrated on a multi-gram scale with consistent results, indicating a clear path towards kilogram and ton-scale production. The use of visible light is inherently safer than UV radiation, reducing occupational health hazards and simplifying reactor design. Moreover, the reduced solvent usage and waste generation align with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. This forward-looking approach to process design ensures long-term viability in a regulatory landscape that prioritizes green chemistry and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines. Comprehensive responses help mitigate technical risks and clarify the operational requirements for successful adoption.
Q: What are the key advantages of this synthesis method over conventional thermal approaches?
A: This method utilizes mild room temperature conditions and visible light catalysis, eliminating the need for harsh thermal inputs and expensive transition metal chiral ligands, which significantly reduces operational costs and environmental impact.
Q: What level of stereoselectivity can be achieved with this protocol?
A: The protocol demonstrates excellent stereocontrol, consistently achieving high enantiomeric excess (ee) values often exceeding 90% and superior diastereoselectivity (dr), ensuring the production of high-purity intermediates suitable for drug development.
Q: Is this synthetic route scalable for commercial production?
A: Yes, the patent data includes successful scale-up experiments demonstrating consistent yields and selectivity on a multi-gram scale, indicating strong potential for adaptation to larger commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoroalkyl Cyclopentenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric synthesis technology for the development of advanced fluorinated therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity cyclopentenone derivatives meets the exacting standards required by global pharmaceutical companies. Our commitment to quality and consistency makes us a trusted partner for organizations seeking to secure their supply chain for critical intermediates.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your drug discovery programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. Please contact us to request specific COA data and route feasibility assessments that will demonstrate how we can support your goals for cost reduction in fine chemical manufacturing. By partnering with us, you gain access to a reliable pharmaceutical intermediate supplier dedicated to driving innovation and efficiency in your supply chain.
