Industrial Scale-Up of 3-Ethoxy Ethyl Acrylate via Green Trichloroacetyl Chloride Route
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. Patent CN109438237B, published in May 2021, introduces a groundbreaking preparation method for 3-ethoxy ethyl acrylate, a vital building block in the synthesis of fluoroquinolone antibiotics such as levofloxacin and ciprofloxacin. This novel approach addresses the longstanding limitations of traditional synthesis methods by utilizing trichloroacetyl chloride and vinyl ethyl ether as primary feedstocks. The significance of this patent lies not only in its chemical efficiency but also in its alignment with green chemistry principles, offering a pathway that drastically reduces the reliance on precious metal catalysts and complex purification steps. For R&D directors and procurement managers, understanding the nuances of this technology is essential for optimizing supply chains and ensuring the consistent availability of high-purity intermediates. The method promises a yield exceeding 80% with purity levels reaching 98.8%, demonstrating its viability for stringent pharmaceutical applications where impurity profiles are critically monitored.
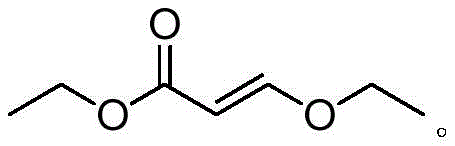
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-ethoxy ethyl acrylate has been plagued by significant economic and environmental hurdles that hinder large-scale industrial adoption. Traditional routes often rely on ethyl propiolate and ethanol, which, while offering a short reaction pathway, involve raw materials that are prohibitively expensive and difficult to source in bulk quantities. Another common method utilizes palladium chloride catalysts to facilitate the reaction between ethyl acrylate and ethanol; however, this introduces severe complications regarding heavy metal contamination. The removal of palladium residues requires sophisticated and costly purification technologies, which not only inflate the production cost but also pose environmental disposal challenges. Furthermore, alternative methods involving zinc or magnesium catalysts generate substantial amounts of inorganic waste, complicating the waste treatment process and increasing the overall carbon footprint of the manufacturing operation. These legacy methods often struggle to balance yield with purity, frequently resulting in by-products that are difficult to separate, thereby compromising the quality of the final intermediate required for sensitive API synthesis.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in patent CN109438237B leverages a unique three-step cascade reaction that bypasses the need for precious metals entirely. By initiating the synthesis with the addition of vinyl ethyl ether to trichloroacetyl chloride, the process establishes a robust carbon framework under mild thermal conditions ranging from 20°C to 40°C. This initial step is followed by an ethanolysis reaction mediated by an organic base, which effectively substitutes the chlorine atoms with ethoxy groups while generating a recoverable solid by-product. The final elimination step, catalyzed by a simple acid catalyst under a nitrogen atmosphere, ensures the formation of the double bond with high stereoselectivity. This approach not only simplifies the operational workflow but also enhances the overall atom economy of the reaction. The elimination of transition metals means that the downstream processing is significantly streamlined, reducing the number of unit operations required to achieve pharmaceutical-grade purity. This represents a paradigm shift in how this key intermediate can be manufactured, offering a sustainable alternative that aligns with modern regulatory expectations for green manufacturing.
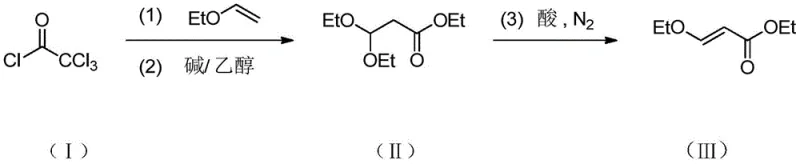
Mechanistic Insights into Trichloroacetyl Chloride Mediated Synthesis
The core of this innovative synthesis lies in the precise control of the addition-elimination mechanism, which is fundamentally different from the metal-catalyzed coupling reactions of the past. In the first stage, the electron-deficient carbonyl carbon of trichloroacetyl chloride undergoes a nucleophilic attack by the vinyl ethyl ether. This reaction is highly exothermic, necessitating strict temperature control between 20°C and 40°C to prevent polymerization or side reactions. The resulting intermediate is a chlorinated ester that serves as the precursor for the subsequent substitution. The use of trichloroacetyl chloride is strategic; the three chlorine atoms act as excellent leaving groups, facilitating the subsequent nucleophilic substitution by ethanol in the presence of an organic base such as triethylamine or diisopropylethylamine. This step is crucial as it installs the ethoxy functionality at the beta-position, which is the defining structural feature of the target molecule. The reaction kinetics are optimized by maintaining a molar ratio of organic base to trichloroacetyl chloride between 1:1 and 1:3, ensuring complete conversion while minimizing the formation of amine salts that could complicate filtration.
Impurity control is inherently built into the mechanistic design of this process, addressing a primary concern for R&D directors focused on quality. The final elimination step, which converts the saturated intermediate into the unsaturated 3-ethoxy ethyl acrylate, is driven by an acid catalyst such as potassium bisulfate at temperatures between 50°C and 100°C. The introduction of nitrogen gas during this phase serves a dual purpose: it acts as a carrier to remove low-boiling by-products and prevents oxidative degradation of the sensitive acrylate double bond. This inert atmosphere is critical for maintaining the color and stability of the product, which are key quality indicators for downstream API manufacturers. Furthermore, the by-product of the ethanolysis step, the organic base hydrochloride, precipitates as a solid filter cake. This physical separation allows for the easy removal of the base salt, which can then be regenerated and recycled, effectively closing the loop on reagent consumption. This mechanistic elegance ensures that the final distillation yields a product with purity levels consistently above 98%, meeting the rigorous specifications required for the synthesis of complex antibiotics like levofloxacin without the need for extensive chromatographic purification.
How to Synthesize 3-Ethoxy Ethyl Acrylate Efficiently
Implementing this synthesis route in a commercial setting requires a clear understanding of the operational parameters that drive efficiency and safety. The process is designed to be telescoped where possible, minimizing the transfer of intermediates and reducing the risk of exposure to hazardous materials. The initial addition of vinyl ethyl ether must be performed slowly over a period of 1 to 2 hours to manage the heat of reaction effectively, ensuring that the temperature does not exceed the 40°C threshold which could lead to thermal runaway. Following the reaction, the low-boiling by-products are removed under reduced pressure at temperatures below 40°C, a step that is energy-efficient and protects the thermal stability of the intermediate. The subsequent addition of ethanol and organic base is performed at mild temperatures (20-50°C), allowing for a prolonged reaction time of 1 to 10 hours to ensure complete conversion. The detailed standardized synthesis steps, including specific stirring rates, vacuum levels, and distillation cuts, are critical for reproducibility and are outlined in the technical guide below for process engineers.
- Dropwise add vinyl ethyl ether into trichloroacetyl chloride at 20-40°C and react for 1-10 hours to form the intermediate.
- Add organic base and ethanol, reacting at 20-50°C for 1-10 hours, followed by filtration to recover the organic base hydrochloride.
- Add acid catalyst, heat to 50-100°C under nitrogen flow, and perform reduced pressure distillation to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patent technology offers transformative benefits that extend far beyond simple chemical yield. The most significant advantage is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts. Traditional palladium-catalyzed routes require not only the initial purchase of expensive metals but also the implementation of costly scavenging processes to meet residual metal specifications. By switching to an organic base and acid catalyst system, manufacturers can achieve substantial cost savings in both material procurement and waste management. Furthermore, the raw materials utilized in this process, specifically trichloroacetyl chloride and vinyl ethyl ether, are commodity chemicals with stable supply chains, reducing the risk of supply disruptions that often plague specialty reagent-dependent processes. This stability is crucial for supply chain heads who must guarantee continuous production schedules for downstream API clients. The ability to recover and reuse the organic base and ethanol solvent further enhances the economic viability of the process, creating a circular economy within the manufacturing plant that minimizes variable costs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as palladium and zinc removes a major cost driver from the bill of materials. Precious metals are subject to volatile market pricing, and their removal from the process stabilizes the cost structure of the intermediate. Additionally, the simplified work-up procedure, which relies on filtration and distillation rather than complex extraction or chromatography, reduces labor hours and utility consumption. The recovery of the organic base hydrochloride allows for the regeneration of the amine, effectively turning a waste product into a reusable asset. This logical deduction of cost savings implies a significantly lower cost of goods sold (COGS) compared to legacy methods, providing a competitive edge in pricing negotiations with pharmaceutical buyers.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals rather than specialized catalysts ensures a more robust supply chain. Trichloroacetyl chloride and vinyl ethyl ether are produced by multiple global suppliers, mitigating the risk of single-source dependency. The mild reaction conditions also reduce the strain on equipment, leading to lower maintenance downtime and higher overall equipment effectiveness (OEE). For supply chain managers, this translates to more predictable lead times and the ability to scale production rapidly in response to market demand without the bottleneck of catalyst availability. The process is inherently safer due to the lower operating temperatures and pressures, reducing the likelihood of safety incidents that could halt production. This reliability is a key value proposition for long-term supply agreements with multinational corporations.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns perfectly with increasingly stringent environmental regulations. The reduction in heavy metal waste simplifies the effluent treatment process, lowering the cost of environmental compliance and reducing the facility's ecological footprint. The solvent recovery system ensures that ethanol emissions are minimized, contributing to better air quality and regulatory adherence. From a scalability standpoint, the batch process described is easily adaptable to large-scale reactors, with the heat transfer and mixing requirements being well within the capabilities of standard industrial equipment. The high atom utilization rate means that less raw material is wasted, enhancing the sustainability profile of the manufacturing operation. This makes the technology not only commercially attractive but also socially responsible, appealing to stakeholders who prioritize ESG (Environmental, Social, and Governance) criteria in their supplier selection process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN109438237B, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs. The clarity provided here aims to bridge the gap between patent theory and industrial practice, ensuring that all stakeholders have a unified understanding of the process capabilities and limitations.
Q: What are the primary advantages of the trichloroacetyl chloride route over palladium-catalyzed methods?
A: The trichloroacetyl chloride route eliminates the need for expensive and toxic palladium catalysts, significantly reducing raw material costs and simplifying heavy metal removal processes, which is critical for pharmaceutical compliance.
Q: How does this process handle waste management and solvent recovery?
A: The process generates solid organic base hydrochlorides that can be regenerated and reused. Additionally, the ethanol solvent is recovered via reduced pressure distillation with high purity, minimizing liquid waste discharge.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (20-100°C) and utilize cheap, readily available raw materials like vinyl ethyl ether and trichloroacetyl chloride, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethoxy Ethyl Acrylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the fine chemical industry. As a leading CDMO and manufacturer, we possess the technical expertise to translate the innovations found in patent CN109438237B into commercial reality. Our facilities are equipped to handle complex organic syntheses with the highest standards of safety and quality control. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of 3-ethoxy ethyl acrylate meets the exacting requirements of the pharmaceutical sector. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and compliant with global regulatory standards.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality specifications. We encourage you to reach out to request specific COA data and route feasibility assessments to verify how our implementation of this green synthesis technology can benefit your operations. Whether you are looking to secure a long-term supply agreement or need support with process development, NINGBO INNO PHARMCHEM is your strategic partner in delivering high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
