Advanced Metal-Free Synthesis of 2,3,5,6-Tetrasubstituted Pyridines for Commercial Scale-Up
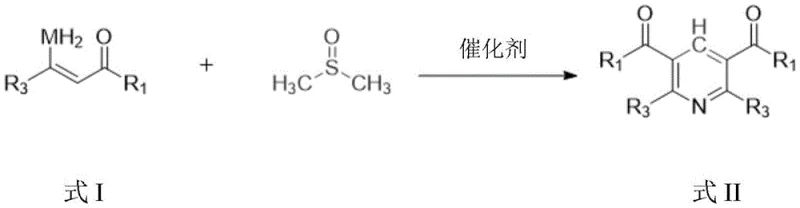
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN116143689A, which discloses a novel synthesis method for 2,3,5,6-tetrasubstituted pyridine compounds. This technology represents a paradigm shift from traditional transition metal-catalyzed processes to a more economical organocatalytic approach. By utilizing dimethyl sulfoxide (DMSO) as a single-carbon surrogate and imidazole hydrochloride as a promoter, this method achieves the construction of the pyridine core through the reaction of enaminones. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition: it bypasses the need for precious metal catalysts while maintaining high yields and exceptional functional group tolerance. As a reliable pharmaceutical intermediate supplier, understanding such technological advancements is crucial for optimizing supply chains and ensuring the continuous availability of high-purity building blocks for drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5,6-tetrasubstituted pyridine compounds has been fraught with significant challenges that hinder efficient commercial production. Traditional methodologies often rely on the condensation and oxidative aromatization of 1,3-dicarbonyl compounds with formaldehyde and inorganic ammonium salts. However, these classical routes frequently suffer from generally low yields and a notably narrow substrate scope, limiting their utility in diverse medicinal chemistry programs. Furthermore, more modern approaches, such as the ruthenium-catalyzed synthesis reported by Guan et al. in 2014, while effective, introduce the burden of using expensive transition metals. The reliance on metals like ruthenium not only escalates raw material costs but also necessitates rigorous and costly downstream processing to remove trace metal residues to meet stringent regulatory standards for pharmaceutical ingredients. These factors collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks in the supply of critical pharmaceutical intermediates.
The Novel Approach
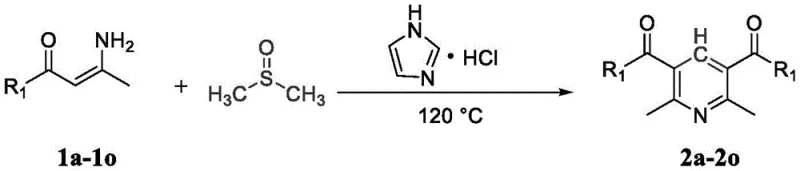
In stark contrast to these legacy methods, the technology described in patent CN116143689A introduces a streamlined, metal-free protocol that effectively addresses the aforementioned pain points. This novel approach promotes the reaction of enaminones with DMSO using imidazole hydrochloride, a readily available and inexpensive organocatalyst. The reaction proceeds under relatively mild thermal conditions, typically around 120°C, to afford the desired tetrasubstituted pyridines in moderate to good yields. This methodology not only simplifies the reaction setup by eliminating the need for inert atmospheres often required by sensitive metal catalysts but also drastically reduces the environmental footprint associated with heavy metal waste. The ability to synthesize a wide range of pyridines without metal catalysts signifies a major step forward in cost reduction in pharmaceutical intermediate manufacturing, offering a greener and more economically viable alternative for industrial applications.
Mechanistic Insights into Imidazole Hydrochloride-Promoted Cyclization
The core of this synthetic innovation lies in the unique role of imidazole hydrochloride acting as a Brønsted acid catalyst to facilitate the cyclization process. Unlike Lewis acid metal catalysts that coordinate with substrates to lower activation energy, imidazole hydrochloride likely promotes the reaction through protonation events that enhance the electrophilicity of the intermediates formed during the interaction between the enaminone and DMSO. DMSO serves a dual purpose here, acting both as the solvent and as the source of the single carbon atom required to close the pyridine ring. This mechanistic pathway is particularly advantageous because it avoids the formation of stable metal-substrate complexes that can sometimes lead to catalyst deactivation or difficult product separation. The reaction mechanism supports a broad tolerance for various electronic environments on the substrate, allowing for the successful incorporation of diverse functional groups without compromising the integrity of the catalytic cycle or the final product purity.
From an impurity control perspective, this metal-free system offers distinct advantages for producing high-purity pharmaceutical intermediates. In metal-catalyzed reactions, side products often include metal-coordinated species or products resulting from metal-mediated side reactions, which can be notoriously difficult to remove. By utilizing an organic salt catalyst, the impurity profile is significantly simplified, primarily consisting of organic byproducts that are more amenable to standard purification techniques like crystallization or silica gel chromatography. The patent data indicates that electron-donating groups such as methyl and methoxy, as well as electron-withdrawing groups like halogens, are well-tolerated, yielding products with yields ranging from 67% to 94%. This robustness ensures consistent quality across different batches, a critical factor for supply chain reliability.
How to Synthesize 2,3,5,6-Tetrasubstituted Pyridines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and efficiency. The general procedure involves charging a reaction vessel with the enaminone starting material, the imidazole hydrochloride catalyst, and DMSO as the reaction medium. The mixture is then heated to optimize the reaction kinetics, typically maintained at 120°C for a duration of approximately 8 hours. Reaction progress is conveniently monitored using thin-layer chromatography (TLC), allowing for precise determination of the endpoint. Upon completion, the workup procedure is straightforward, involving quenching with a saturated sodium chloride solution followed by extraction with ethyl acetate. The detailed standardized synthesis steps for this process are outlined below to ensure reproducibility and safety during scale-up operations.
- Charge a round bottom flask with enaminone substrate, imidazole hydrochloride catalyst, and DMSO solvent.
- Heat the reaction mixture to 120°C and stir for approximately 8 hours while monitoring progress via TLC.
- Quench with saturated NaCl solution, extract with ethyl acetate, dry over anhydrous Na2SO4, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend beyond simple chemical transformation. The primary advantage lies in the substantial optimization of the cost structure associated with producing complex heterocyclic intermediates. By eliminating the requirement for precious metal catalysts such as ruthenium or palladium, manufacturers can achieve significant reductions in raw material expenditures. Furthermore, the absence of heavy metals removes the necessity for specialized and expensive metal scavenging resins or complex purification protocols, thereby streamlining the production workflow and reducing overall processing time. This simplification directly contributes to cost reduction in API manufacturing, allowing for more competitive pricing structures in the final supply of active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts fundamentally alters the economic model of producing tetrasubstituted pyridines. Without the need for costly metals like ruthenium, the direct material cost is significantly lowered. Additionally, the downstream processing becomes more efficient as there is no need for rigorous heavy metal clearance steps, which often involve expensive reagents and extended processing times. This leads to a leaner manufacturing process with reduced operational overheads and waste disposal costs, ultimately enhancing the profit margin for high-volume production runs.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents such as enaminones, DMSO, and imidazole hydrochloride ensures a robust and resilient supply chain. Unlike specialized metal catalysts which may be subject to geopolitical supply constraints or price volatility, these organic reagents are commodity chemicals with stable global availability. This stability minimizes the risk of production delays caused by raw material shortages, ensuring consistent delivery schedules for clients. The robustness of the reaction conditions further supports reliable manufacturing, reducing the likelihood of batch failures due to catalyst sensitivity.
- Scalability and Environmental Compliance: The protocol has been validated on a multi-gram scale, demonstrating its potential for seamless translation to commercial production volumes. The metal-free nature of the reaction aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and waste management. By avoiding toxic metal waste, the process simplifies environmental compliance and reduces the burden on waste treatment facilities. This green chemistry approach not only mitigates regulatory risks but also enhances the corporate sustainability profile, making it an attractive option for environmentally conscious partners seeking commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2,3,5,6-tetrasubstituted pyridines using this novel metal-free protocol. These insights are derived directly from the experimental data and beneficial effects described in patent CN116143689A. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided reflect the specific capabilities of the imidazole hydrochloride-promoted reaction system, highlighting its versatility and operational advantages over traditional methods.
Q: What are the advantages of using imidazole hydrochloride over transition metal catalysts?
A: Imidazole hydrochloride eliminates the need for expensive and toxic transition metals like Ruthenium, significantly reducing raw material costs and simplifying downstream purification by removing heavy metal clearance steps.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates excellent tolerance for various functional groups including electron-donating groups (Me, OMe) and electron-withdrawing groups (Cl, Br, F), as well as heterocycles like furan and thiophene.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly mentions successful multi-gram scale reactions, indicating robust scalability and potential for commercial production without significant loss in efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrasubstituted Pyridines Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the metal-free pyridine synthesis described in CN116143689A can be effectively translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,3,5,6-tetrasubstituted pyridines supplied meets the highest standards required for drug development. Our infrastructure is designed to support both rapid prototyping for R&D and large-scale manufacturing for commercial launch.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable supply of high-quality heterocyclic intermediates.
