Revolutionizing 1-Alkoxy Isoquinoline Production: A Cost-Effective Cobalt-Catalyzed C-H Activation Strategy for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways to access privileged heterocyclic scaffolds. A recent breakthrough detailed in patent CN115286573A introduces a transformative green synthesis method for 1-alkoxy isoquinoline compounds, a structural motif prevalent in bioactive molecules such as the potent anesthetic Quinicaine. This innovation addresses critical bottlenecks in traditional manufacturing by leveraging a cobalt-catalyzed intramolecular C-H activation strategy within a recyclable deep eutectic solvent (DES) system. For R&D directors and procurement leaders, this technology represents a paradigm shift from precious metal dependency to earth-abundant catalysis, promising substantial operational efficiencies and cost reduction in API manufacturing without compromising on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of azacyclic ethers like 1-alkoxy isoquinolines has relied on harsh and economically inefficient protocols. Traditional approaches often involve the nucleophilic substitution of azacyclic halides with alcohols under strong basic conditions, which typically necessitates elevated temperatures and suffers from mediocre yields. More sophisticated methods, such as those reported by Londregan et al., utilize isoquinoline N-oxides promoted by tripyrrolidinylphosphonium bromide hexafluorophosphate (PyBroP). While effective, PyBroP is prohibitively expensive, costing approximately 280 yuan per gram, and requires multi-step synthesis for substituted precursors. Furthermore, alternative transition-metal catalyzed routes frequently employ rhodium complexes, which are not only costly but often require symmetrical alkyne substrates, limiting structural diversity. These legacy methods also heavily depend on toxic, volatile organic solvents that pose significant environmental hazards and complicate waste management protocols.
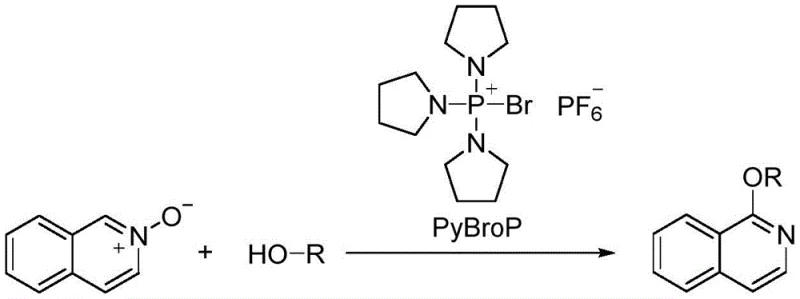
The Novel Approach
In stark contrast, the methodology disclosed in CN115286573A utilizes readily available benzonitriles and alcohols as starting materials, reacting them with vinylene carbonate in a one-pot procedure. The core innovation lies in the use of a cobalt catalyst coupled with a biomass-derived deep eutectic solvent, specifically a betaine/hexafluoroisopropanol mixture. This system facilitates a seamless addition reaction followed by intramolecular C-H activation and cyclization. The result is a direct, high-yielding pathway to 1-alkoxy isoquinolines that are unsubstituted at the 3,4-positions, providing a versatile platform for further functionalization. By eliminating the need for expensive phosphonium salts or rhodium catalysts and replacing volatile organic compounds with recyclable green solvents, this approach drastically simplifies the production workflow and enhances the overall sustainability profile of the synthesis.
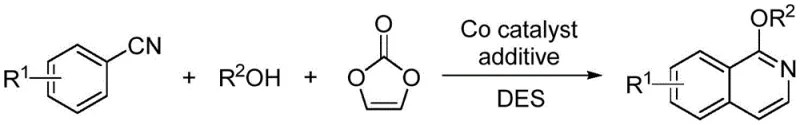
Mechanistic Insights into Cobalt-Catalyzed C-H Activation/Cyclization
The mechanistic elegance of this transformation centers on the cooperative action of the pentamethylcyclopentadienyl cobalt complex, specifically Cp*Co(CO)I2, and a copper oxidant additive. The reaction initiates with the coordination of the cobalt catalyst to the nitrile nitrogen, facilitating the nucleophilic attack by the alcohol to form an imidate intermediate. Subsequently, the cobalt center activates the proximal aromatic C-H bond, a critical step that enables the subsequent cyclization with vinylene carbonate. The vinylene carbonate serves as a two-carbon synthon, effectively inserting into the C-H bond to close the isoquinoline ring while releasing carbon dioxide. The copper additive, optimally copper acetate, acts as a terminal oxidant to regenerate the active cobalt species, ensuring the catalytic cycle continues efficiently. This mechanism avoids the formation of stable off-cycle intermediates, contributing to the high turnover numbers observed.
From an impurity control perspective, the use of deep eutectic solvents plays a pivotal role in stabilizing reactive intermediates and suppressing side reactions. The specific interaction between the hydrogen bond donor (hexafluoroisopropanol) and acceptor (betaine) creates a unique microenvironment that enhances the solubility of polar transition states while excluding non-polar impurities. Experimental data confirms that varying the molar ratio of the DES components significantly impacts yield, with a 1:2 ratio of betaine to hexafluoroisopropanol proving optimal. This precise tuning of the solvent matrix minimizes the formation of polymeric byproducts or over-oxidized species, ensuring that the final crude product possesses a clean impurity profile suitable for downstream pharmaceutical processing.
How to Synthesize 1-Methoxyisoquinoline Efficiently
To implement this green synthesis protocol effectively, precise control over reaction parameters is essential. The standard procedure involves charging a reactor with benzonitrile, methanol, and vinylene carbonate in the presence of 5 mol% Cp*Co(CO)I2 and 20 mol% copper acetate within the optimized deep eutectic solvent. The mixture is then heated to 110°C for 12 hours to drive the cyclization to completion. Following the reaction, the product is isolated via simple organic extraction, leaving the catalyst and solvent system in the aqueous raffinate for reuse. This streamlined workup eliminates the need for complex distillation or extensive chromatographic purification at the industrial scale. For detailed standardized synthesis steps and specific safety handling procedures, please refer to the guide below.
- Combine benzonitrile, alcohol, and vinylene carbonate in a deep eutectic solvent (e.g., Betaine/HFIP) with Cp*Co(CO)I2 catalyst and copper acetate additive.
- Heat the reaction mixture to 110°C and maintain for 12 hours to facilitate intramolecular C-H activation and cyclization.
- Cool the mixture to room temperature, extract the product with ethyl acetate, and purify via column chromatography if necessary.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed protocol offers compelling strategic advantages beyond mere technical feasibility. The shift from precious metals to base metals fundamentally alters the cost structure of raw materials, mitigating exposure to the volatile pricing of rhodium and specialized phosphonium reagents. Furthermore, the ability to recycle the solvent and catalyst system multiple times without significant loss of activity translates directly into reduced consumption of consumables and lower waste disposal costs. This aligns perfectly with corporate sustainability goals while simultaneously driving down the cost of goods sold (COGS) for high-value intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium catalysts and PyBroP promoters with inexpensive cobalt salts and biomass-derived solvents results in a drastic reduction in material costs. Since the catalytic system and solvent can be recycled for more than six cycles, the effective cost per kilogram of product is significantly lowered. Additionally, the use of commodity chemicals like benzonitrile and methanol as starting materials ensures a stable and low-cost supply chain, avoiding the premiums associated with specialized heterocyclic building blocks.
- Enhanced Supply Chain Reliability: Reliance on earth-abundant cobalt rather than scarce platinum-group metals reduces supply chain risks associated with geopolitical instability or mining constraints. The starting materials, including benzonitriles and vinylene carbonate, are widely produced industrial commodities with robust global availability. This ensures consistent production schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The process operates under relatively mild thermal conditions and avoids the use of chlorinated or highly volatile organic solvents, simplifying regulatory compliance and facility safety requirements. The deep eutectic solvent system is non-toxic and biodegradable, minimizing the environmental footprint of the manufacturing process. The straightforward extraction workup facilitates easy scale-up from laboratory to commercial production volumes, ensuring that the process remains efficient and manageable even at multi-ton scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Can the deep eutectic solvent and catalyst system be recycled?
A: Yes, the patent data indicates that the deep eutectic solvent/cobalt salt catalytic system can be recycled and reused for more than 6 cycles while maintaining high product yields, significantly reducing waste generation.
Q: What are the advantages of this method over traditional Rhodium-catalyzed routes?
A: This method utilizes abundant and low-cost cobalt catalysts instead of expensive rhodium complexes. Additionally, it avoids the use of toxic volatile organic solvents by employing biodegradable deep eutectic solvents, aligning better with green chemistry principles.
Q: Is this synthesis method compatible with diverse functional groups?
A: The process demonstrates excellent functional group tolerance, successfully accommodating substrates with methyl, methoxy, halogen, cyano, ester, trifluoromethyl, and nitro groups at various positions on the benzonitrile ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Alkoxy Isoquinoline Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with a technically proficient CDMO is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 1-alkoxy isoquinoline meets the highest international standards, ready for immediate integration into your drug development pipelines.
We invite you to leverage our expertise in green chemistry and process optimization to enhance your own manufacturing capabilities. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative cobalt-catalyzed technology can deliver value to your organization immediately.
