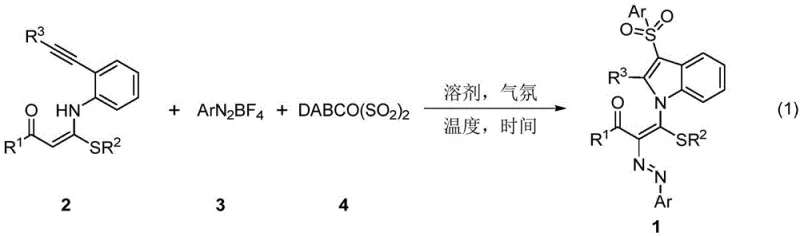
Advanced Metal-Free Synthesis of 3-Arylsulfonyl Indole Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic methodologies that balance structural complexity with manufacturing efficiency, particularly for nitrogen-containing heterocycles like indoles which serve as critical scaffolds in oncology therapeutics. Patent CN111269165A introduces a transformative approach for synthesizing 3-arylsulfonyl indole derivatives, utilizing readily available 1,5-enyne compounds as key building blocks. This innovation addresses long-standing challenges in heterocyclic chemistry by replacing hazardous gaseous sulfur dioxide and expensive transition metal catalysts with a stable solid surrogate, DABCO(SO2)2. The methodology not only streamlines the synthetic pathway but also enhances the safety profile of the operation, making it highly attractive for the production of high-purity pharmaceutical intermediates. By leveraging aryl diazonium salts in conjunction with the sulfur dioxide surrogate, the process achieves excellent regioselectivity and functional group tolerance under remarkably mild conditions. This represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to diversify their portfolio with safer, more sustainable chemistries that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-arylsulfonyl indole frameworks has relied heavily on catalytic systems involving precious metals such as Indium, Gold, or Palladium, which present substantial economic and environmental burdens. Traditional protocols often require the use of arylsulfonyl chlorides or direct sulfonation with gaseous SO2, both of which pose significant handling risks and corrosion issues in large-scale reactors. Furthermore, metal-catalyzed cyclizations frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that necessitate costly and time-consuming purification steps to meet stringent quality standards. The residual heavy metals from these catalysts can be notoriously difficult to remove to parts-per-million levels required for active pharmaceutical ingredients, often demanding additional scavenging technologies that drive up the overall cost of goods. Additionally, the reliance on specialized ligands and anhydrous conditions for noble metal catalysis adds layers of operational complexity that hinder the commercial scale-up of complex pharmaceutical additives and intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a metal-free radical cascade reaction that proceeds efficiently at ambient temperatures, drastically simplifying the operational requirements. By employing DABCO(SO2)2 as a convenient and safe source of sulfur dioxide, the process eliminates the need for high-pressure gas handling equipment and mitigates the toxicity risks associated with free SO2 gas. The reaction demonstrates exceptional substrate scope, accommodating a wide variety of substituents on the 1,5-enyne precursor and the aryl diazonium salt without compromising yield or selectivity. This approach effectively bypasses the need for expensive noble metals, thereby removing the risk of metal contamination entirely and simplifying the downstream workup procedure. The mild reaction conditions, typically ranging from 0°C to 40°C, allow for precise thermal control which is essential for maintaining product integrity during cost reduction in API manufacturing processes.
Mechanistic Insights into Metal-Free Radical Cyclization
The underlying mechanism of this transformation involves a sophisticated radical cascade initiated by the decomposition of the aryl diazonium salt, which generates an aryl radical species capable of attacking the alkyne moiety of the 1,5-enyne substrate. This initial addition triggers a cyclization event that constructs the indole core while simultaneously incorporating the sulfur dioxide molecule released from the DABCO complex. The absence of transition metals suggests a radical pathway rather than a traditional organometallic cycle, which explains the high tolerance for various functional groups that might otherwise poison a metal catalyst. The sulfur dioxide insertion occurs regioselectively at the 3-position of the indole ring, driven by the stability of the intermediate radical species formed during the cascade. Understanding this mechanistic nuance is crucial for R&D teams optimizing the process, as it highlights the importance of controlling radical initiation rates through temperature and concentration adjustments to minimize side reactions. The robustness of this radical mechanism ensures consistent performance across different batches, providing a reliable foundation for scaling up the synthesis of these biologically active molecules.
From an impurity control perspective, the metal-free nature of this reaction offers a distinct advantage by eliminating an entire class of potential contaminants associated with catalyst degradation or ligand dissociation. Without the presence of heavy metals, the impurity profile is dominated primarily by organic byproducts which are generally easier to separate using standard chromatographic techniques or crystallization. This simplification of the impurity landscape significantly reduces the analytical burden on quality control laboratories and accelerates the release of materials for clinical evaluation. Furthermore, the use of stable solid reagents like DABCO(SO2)2 ensures consistent stoichiometry and reactivity, minimizing batch-to-batch variability that can often plague processes relying on unstable gaseous reagents. For procurement managers, this translates to a more predictable supply chain with fewer variables affecting the final quality of the high-purity OLED material or pharmaceutical intermediate.
How to Synthesize 3-Arylsulfonyl Indole Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory equipment and commonly available solvents to facilitate rapid adoption in process development labs. The protocol typically involves charging a reaction vessel with the 1,5-enyne compound, the aryl diazonium tetrafluoroborate salt, and the DABCO(SO2)2 complex in a molar ratio optimized for maximum conversion, often around 1:3:3. The choice of solvent plays a critical role, with 1,2-dichloroethane or dichloromethane identified as the optimal media to ensure solubility of all components and efficient heat transfer during the exothermic radical initiation phase. Maintaining an inert atmosphere, preferably argon, is recommended to prevent oxidative side reactions that could degrade the sensitive diazonium species or the radical intermediates. Detailed standardized synthetic steps for replicating this high-yielding transformation are provided in the guide below, ensuring reproducibility for technical teams evaluating this route for commercial adoption.
- Prepare the reaction mixture by combining 1,5-enyne compound, aryl diazonium salt, and DABCO(SO2)2 in a solvent like 1,2-dichloroethane under an argon atmosphere.
- Maintain the reaction temperature between 0°C and 40°C, preferably at 25°C, and stir for 8 to 15 hours to ensure complete conversion.
- Upon completion, remove volatiles under reduced pressure and purify the crude product via silica gel column chromatography to isolate the target indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the shift towards this metal-free synthesis represents a strategic opportunity to de-risk the sourcing of critical indole-based intermediates while achieving substantial cost savings. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like Palladium and Gold can fluctuate wildly based on geopolitical factors and mining outputs. Moreover, the use of bench-stable solid reagents simplifies logistics and storage requirements, as there is no need for specialized high-pressure cylinders or cryogenic storage facilities often associated with gaseous reagents. This operational simplicity enhances supply chain reliability by reducing the dependency on specialized hazardous material transporters and allowing for broader sourcing options for raw materials. The mild reaction conditions also contribute to lower energy consumption during manufacturing, aligning with corporate sustainability goals and potentially reducing utility costs associated with heating or cooling large-scale reactors.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts and their associated ligands directly lowers the raw material costs, while the simplified purification process reduces solvent usage and waste disposal fees. By avoiding the need for metal scavengers and extensive filtration steps, the overall processing time is shortened, leading to higher throughput and better asset utilization in multipurpose plants. The use of commodity chemicals like DABCO derivatives and diazonium salts ensures a stable and competitive pricing structure for the key inputs, shielding the project from supply shocks common with specialty catalysts. This economic efficiency makes the process highly viable for producing cost-sensitive generic drug intermediates where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents is inherently less risky than managing the supply of toxic gases or air-sensitive organometallic complexes, ensuring continuous production even during logistical disruptions. The broad substrate scope allows for flexibility in raw material selection, enabling procurement teams to qualify multiple suppliers for the 1,5-enyne precursors and aryl diazonium salts to prevent single-source bottlenecks. The robustness of the reaction conditions means that minor variations in raw material quality are less likely to cause batch failures, thereby improving the overall yield consistency and on-time delivery performance. This resilience is critical for maintaining the continuity of supply for life-saving medications where interruptions can have severe consequences.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the environmental permitting process for new manufacturing lines, as wastewater treatment becomes less complex without the need for specialized metal precipitation units. The mild thermal profile of the reaction reduces the risk of thermal runaway incidents, enhancing plant safety and lowering insurance premiums associated with hazardous chemical operations. Scalability is further supported by the homogeneous nature of the reaction mixture, which facilitates efficient mixing and heat removal in large vessels without the mass transfer limitations often seen in heterogeneous catalysis. These factors collectively enable a smoother transition from pilot scale to full commercial production, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route, drawing directly from the experimental data and scope defined in the patent literature. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating the technology for integration into their existing manufacturing platforms. Understanding these details is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this metal-free synthesis over traditional methods?
A: Unlike conventional methods requiring expensive precious metal catalysts like Gold or Palladium, this protocol utilizes DABCO(SO2)2 as a safe sulfur dioxide source, eliminating heavy metal contamination risks and significantly reducing raw material costs while maintaining high regioselectivity.
Q: What are the optimal reaction conditions for scaling this process?
A: The process operates optimally at mild temperatures between 0°C and 40°C, typically at 25°C, using 1,2-dichloroethane as the preferred solvent under an inert argon atmosphere, which facilitates safe and energy-efficient commercial scale-up.
Q: How does this method impact the purity profile of the final API intermediate?
A: By avoiding transition metal catalysts, the resulting 3-arylsulfonyl indole derivatives exhibit superior purity profiles without the need for complex metal scavenging steps, thereby simplifying downstream purification and ensuring compliance with stringent pharmaceutical impurity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylsulfonyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free cyclization technology and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to the global market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and inert atmosphere requirements of this chemistry, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that transitioning to a new synthetic route requires a partner who can navigate the complexities of process optimization and regulatory compliance with precision and care. Our team of expert chemists is ready to collaborate with your R&D division to fine-tune the reaction parameters for your specific target molecule, ensuring maximum efficiency and yield.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free protocol for your specific project needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make data-driven decisions for your supply chain strategy. Let us help you secure a sustainable and cost-effective supply of high-quality 3-arylsulfonyl indole derivatives that will drive the success of your pharmaceutical development programs.
