Advanced Palladium-Catalyzed Synthesis of Furo[3,2-b]indole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. Patent CN114736212A introduces a significant breakthrough in this domain by disclosing a novel synthesis method for furo[3,2-b]indole derivatives, a privileged structure found in numerous natural products and drug candidates exhibiting anti-allergic, anti-tumor, and analgesic properties. This technology leverages a transition metal-catalyzed oxidative coupling strategy that fundamentally shifts the paradigm from hazardous, multi-step traditional syntheses to a streamlined, one-pot cyclization process. By utilizing readily available Ts-protected 2-(2-furyl)arylamine compounds as starting materials, the invention achieves the direct formation of the C-N bond between the amine nitrogen and the furan ring under remarkably mild conditions. For R&D directors and process chemists, this represents a critical advancement in accessing high-value chemical space with reduced operational complexity and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the furo[3,2-b]indole skeleton has been fraught with significant synthetic challenges that hinder efficient commercial production. Traditional methodologies often rely on the preparation of ortho-furan aryl azide compounds through tedious multi-step sequences, which not only lowers overall throughput but also introduces substantial safety risks due to the explosive nature of azides during handling and storage. Furthermore, existing protocols frequently necessitate extreme reaction conditions, with temperatures soaring as high as 160°C to drive intramolecular cyclization, leading to high energy consumption and potential thermal degradation of sensitive functional groups. Alternative approaches involving Ullmann-type couplings with brominated precursors suffer from similar drawbacks, including the need for stoichiometric amounts of copper and harsh bases, resulting in poor atom economy and difficult waste management. These legacy methods create bottlenecks in the supply chain, increasing the cost of goods sold (COGS) and limiting the ability to rapidly iterate on lead optimization campaigns for new drug discovery programs.
The Novel Approach
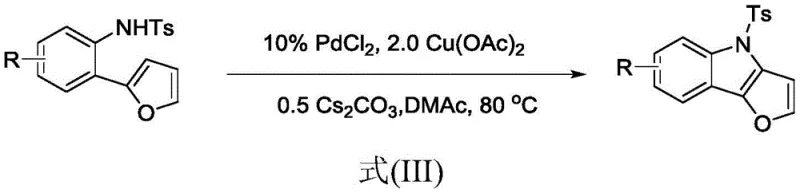
In stark contrast, the methodology described in patent CN114736212A offers a transformative solution by employing a palladium-catalyzed oxidative C-H activation strategy that operates under significantly milder parameters. This innovative route utilizes a robust catalytic system comprising PdCl2 and Cu(OAc)2 in the presence of a cesium carbonate base, enabling the direct coupling of the NH group with the C-3 hydrogen of the furan ring. The reaction proceeds efficiently at a moderate temperature of 80°C in polar aprotic solvents like DMAc, eliminating the need for dangerous high-heat operations. This approach not only simplifies the synthetic workflow by reducing the number of unit operations but also enhances the overall sustainability of the process by improving atom economy and minimizing hazardous waste generation. The versatility of this method is further demonstrated by its tolerance to various substituents, including halogens and alkyl groups, making it a powerful tool for generating diverse libraries of pharmaceutical intermediates.
![General reaction scheme showing the conversion of Ts-protected 2-(2-furyl)arylamine to furo[3,2-b]indole derivative using catalyst and oxidant](/insights/img/furo-indole-synthesis-pd-catalysis-pharma-supplier-20260303112419-02.webp)
Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst and the copper oxidant, which facilitates a unique oxidative cyclization mechanism. The process initiates with the coordination of the palladium species to the nitrogen atom of the Ts-protected amine, followed by the activation of the proximal C-H bond on the furan ring. This C-H activation step is critical, as it bypasses the need for pre-functionalized halogenated substrates, thereby streamlining the synthetic route. The copper co-catalyst, specifically Cu(OAc)2, serves as a terminal oxidant that regenerates the active palladium species, closing the catalytic cycle and driving the reaction forward to completion. The use of Cs2CO3 as a base is instrumental in deprotonating the intermediate species, ensuring smooth progression towards the final fused heterocyclic product. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as catalyst loading and stoichiometry, to maximize yield and purity for specific substrate classes.
From an impurity control perspective, this catalytic system offers distinct advantages over traditional thermal cyclizations. The mild reaction temperature of 80°C significantly reduces the formation of thermal decomposition byproducts and polymerization side reactions that often plague high-temperature processes. Furthermore, the specificity of the palladium-catalyzed C-H activation minimizes the generation of regioisomers, leading to a cleaner crude reaction profile that simplifies downstream purification. The patent data indicates isolated yields ranging from 60% to 80% across a variety of substrates, including those with electron-withdrawing and electron-donating groups, which underscores the robustness of the catalytic cycle. For quality assurance teams, this translates to a more predictable impurity profile and easier validation of the manufacturing process, ensuring consistent supply of high-purity intermediates for subsequent drug substance synthesis.
How to Synthesize 4-Ts-furo[3,2-b]indole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific stoichiometric ratios and atmospheric conditions outlined in the patent to achieve optimal results. The standard protocol involves dissolving the Ts-protected 2-(2-furyl)arylamine starting material in N,N-dimethylacetamide (DMAc) under an inert nitrogen atmosphere to prevent catalyst deactivation by oxygen. To this solution, precise amounts of palladium dichloride (10 mol%), copper acetate (2.0 equivalents), and cesium carbonate (0.5 equivalents) are added sequentially. The detailed standardized synthesis steps below provide a comprehensive guide for executing this transformation effectively, ensuring reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining Ts-protected 2-(2-furyl)arylamine, PdCl2 catalyst, Cu(OAc)2 oxidant, and Cs2CO3 base in DMAc solvent.
- Maintain the reaction under a nitrogen protective atmosphere at a temperature of 80°C for approximately 24 hours to ensure complete cyclization.
- Monitor reaction progress via TLC, then cool to room temperature and perform standard workup procedures to isolate the target furo[3,2-b]indole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic methodology presents a compelling value proposition centered around cost efficiency and operational reliability. By shifting away from hazardous azide chemistry and high-energy thermal processes, manufacturers can significantly reduce the overhead costs associated with specialized safety equipment and waste disposal. The reliance on commodity chemicals such as PdCl2 and Cu(OAc)2, which are widely available from global suppliers, mitigates the risk of raw material shortages and price volatility that often impact niche reagents. Furthermore, the simplified workup procedure, which typically involves cooling and filtration or extraction, reduces the demand for complex purification infrastructure, thereby lowering capital expenditure requirements for new production lines. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of multi-step precursor synthesis and the use of catalytic rather than stoichiometric amounts of expensive metals directly lower the variable costs per kilogram of product. By avoiding the need for high-temperature reactors capable of sustaining 160°C, facilities can utilize standard glass-lined or stainless steel equipment, reducing energy consumption and maintenance costs. Additionally, the higher atom economy of this direct C-H functionalization approach means less raw material is wasted as byproduct, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials for this process, specifically Ts-protected arylamines and furans, are commercially abundant and can be sourced from multiple vendors, ensuring a stable supply base. The mild reaction conditions reduce the likelihood of batch failures due to thermal runaway or equipment malfunction, leading to more consistent production schedules and reliable delivery times for downstream customers. This stability is crucial for maintaining continuous manufacturing flows in the fast-paced pharmaceutical industry, where delays in intermediate supply can halt entire drug development timelines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability potential, as evidenced by the successful synthesis of various derivatives with consistent yields in the patent examples. The reduced generation of hazardous waste and the avoidance of toxic azides align with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. This green chemistry profile not only protects the environment but also enhances the corporate social responsibility standing of the manufacturing entity, making it a preferred partner for sustainability-conscious global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this furo[3,2-b]indole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis routes?
A: This method avoids the use of hazardous aromatic azides and harsh high-temperature conditions (up to 160°C) required by conventional routes, offering a safer, milder process at 80°C with improved atom economy.
Q: What is the typical yield range for this oxidative cyclization process?
A: According to the patent data, the isolated yields for various substituted furo[3,2-b]indole derivatives typically range from 60% to 80%, demonstrating robust efficiency across different substrates.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the use of commercially available reagents like PdCl2 and Cu(OAc)2, combined with mild reaction conditions and simple post-processing, makes this route highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furo[3,2-b]indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust and scalable synthetic routes for complex heterocyclic intermediates like furo[3,2-b]indoles. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies such as the Pd-catalyzed oxidative cyclization described in CN114736212A, we empower our partners to accelerate their drug development pipelines with confidence.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, helping you identify opportunities for margin improvement without compromising quality. Please contact us today to request specific COA data for our available furo[3,2-b]indole derivatives and to discuss route feasibility assessments for your custom synthesis projects. Let us be your trusted partner in navigating the complexities of modern pharmaceutical manufacturing.
