Advanced Rhodium-Catalyzed Synthesis of Trifluoromethyl Indazole Derivatives for Commercial Scale-Up
Introduction to Novel Trifluoromethyl Indazole Synthesis
The pharmaceutical and agrochemical industries continuously seek efficient pathways to construct complex heterocyclic scaffolds, particularly those incorporating fluorine motifs which enhance metabolic stability and bioavailability. Patent CN116003326A introduces a groundbreaking methodology for the synthesis of trifluoromethyl indazole derivatives, addressing critical bottlenecks in traditional heterocycle construction. This innovation leverages a rhodium-catalyzed one-pot cascade reaction between 1-substituted pyrazolidinones and trifluoromethyl alkynyl ketones. Unlike conventional multi-step approaches that often suffer from low overall yields and harsh conditions, this technique operates under remarkably mild parameters, typically at 80°C in an air atmosphere. The strategic integration of a rhodium catalyst facilitates a seamless C-H activation and cyclization sequence, delivering high-purity intermediates essential for downstream drug development. For research teams focused on rapid lead optimization, this protocol represents a significant leap forward in synthetic efficiency.
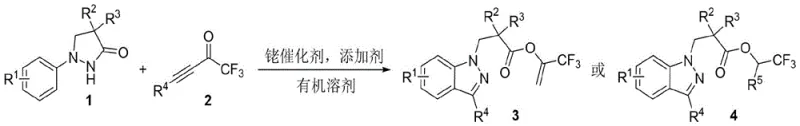
The structural versatility of the resulting indazole core cannot be overstated. By varying the substituents on the pyrazolidinone precursor and the alkynyl ketone, chemists can access a diverse library of compounds bearing different electronic and steric properties. The presence of the trifluoromethyl group is particularly valuable, as it serves as a bioisostere for methyl or hydroxyl groups while imparting unique lipophilic characteristics. This patent data underscores the robustness of the method, demonstrating compatibility with a wide array of functional groups including halogens, alkyl chains, and electron-withdrawing nitro or cyano groups. Such broad substrate scope is a hallmark of a reliable pharmaceutical intermediate supplier capability, ensuring that diverse chemical spaces can be explored without the need for extensive protecting group strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indazole rings functionalized with trifluoromethyl groups has been a laborious endeavor. Traditional synthetic routes often rely on the condensation of hydrazines with appropriate carbonyl precursors, followed by separate trifluoromethylation steps. These sequential processes are inherently inefficient, requiring isolation and purification of unstable intermediates which leads to material loss and increased waste generation. Furthermore, many classical trifluoromethylation reagents are hazardous, expensive, or require cryogenic conditions and strict anhydrous environments. The cumulative effect of these limitations is a prolonged development timeline and inflated production costs, which are unacceptable in the fast-paced environment of modern drug discovery. Additionally, the regioselectivity in forming the indazole core can be problematic, often yielding mixtures of isomers that are difficult to separate, thereby compromising the purity required for biological testing.
The Novel Approach
In stark contrast, the methodology disclosed in CN116003326A streamlines the entire process into a single operational step. By utilizing a rhodium catalyst, specifically dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer, the reaction achieves direct C-H functionalization and cyclization simultaneously. This one-pot strategy not only reduces the number of unit operations but also significantly improves the atom economy of the transformation. The reaction proceeds efficiently in common organic solvents such as dichloroethane or fluorinated alcohols, eliminating the need for exotic or highly toxic media. Perhaps most importantly for industrial application, the reaction tolerates the presence of oxygen and moisture, removing the burden of maintaining an inert atmosphere. This robustness translates directly into lower capital expenditure for reactor setups and simplified operator training, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Rhodium-Catalyzed Cascade Cyclization
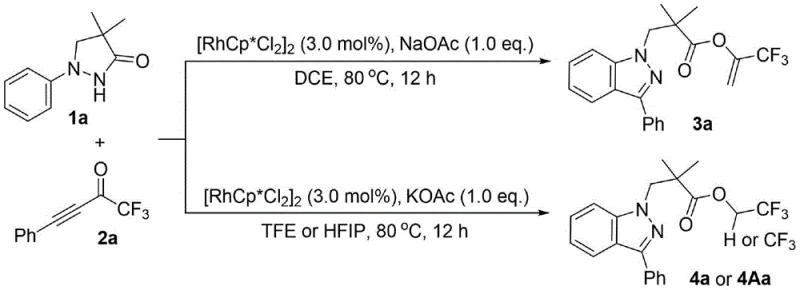
The success of this transformation hinges on the unique ability of the Cp*Rh(III) catalyst to activate the N-H bond of the pyrazolidinone and subsequently direct the insertion of the alkyne moiety. The mechanism likely initiates with the coordination of the rhodium center to the nitrogen atom, followed by concerted metalation-deprotonation (CMD) to form a rhodacycle intermediate. This activated species then undergoes migratory insertion with the trifluoromethyl alkynyl ketone. The subsequent steps involve intramolecular nucleophilic attack and elimination to restore aromaticity and form the indazole ring. The elegance of this system lies in its tunability; the nature of the final ester linkage is dictated by the solvent environment. In non-fluorinated solvents like DCE, the reaction yields the enol ester derivative (Product 3), whereas in fluorinated alcohols like TFE or HFIP, transesterification or direct trapping leads to the trifluoroethyl ester (Product 4). This solvent-controlled divergence provides chemists with a powerful tool to access distinct chemical entities from the same starting materials simply by changing the reaction medium.
From an impurity control perspective, this catalytic cycle is highly selective. The use of additives such as sodium acetate or potassium acetate plays a crucial role in facilitating the C-H activation step and stabilizing the catalytic species. The mild reaction temperature of 80°C minimizes thermal degradation of sensitive functional groups, ensuring a clean reaction profile. Analytical data from the patent indicates that side reactions such as polymerization of the alkyne or over-oxidation are negligible under these optimized conditions. This high level of chemoselectivity is vital for maintaining the integrity of complex molecules during scale-up. For R&D directors, understanding this mechanistic nuance allows for rational optimization of reaction parameters to maximize yield and minimize downstream purification burdens, ultimately accelerating the path from bench to pilot plant.
How to Synthesize Trifluoromethyl Indazole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring, although the protocol itself is forgiving. The standardized procedure involves mixing the pyrazolidinone substrate and the alkynyl ketone in the chosen solvent, followed by the addition of the rhodium catalyst and acetate additive. The mixture is then heated with stirring for approximately 12 hours. Workup is straightforward, typically involving filtration through a short pad of silica or simple aqueous extraction, followed by column chromatography. The detailed standardized synthesis steps are outlined below to ensure reproducibility across different laboratory settings.
- Combine 1-substituted pyrazolidinone compound 1 and trifluoromethyl alkynyl ketone compound 2 in an organic solvent such as dichloroethane or trifluoroethanol.
- Add the rhodium catalyst [RhCp*Cl2]2 and an additive like sodium acetate or potassium acetate to the reaction mixture under air atmosphere.
- Heat the reaction mixture to 60-120°C for approximately 12 hours to facilitate the cascade cyclization, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the simplification of the supply chain for raw materials. Since the reaction tolerates a wide variety of substituents, manufacturers can source generic, commodity-grade starting materials rather than relying on custom-synthesized, high-cost precursors. This flexibility mitigates the risk of supply disruptions and allows for dynamic sourcing strategies based on market availability. Furthermore, the elimination of inert atmosphere requirements means that existing manufacturing infrastructure can be utilized without costly modifications for nitrogen or argon blanketing. This compatibility with standard equipment drastically reduces the barrier to entry for commercial production.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis fundamentally alters the cost structure of producing trifluoromethyl indazoles. By collapsing multiple synthetic steps into a single operation, manufacturers save significantly on labor, energy, and solvent consumption. The high atom economy ensures that a greater proportion of raw material mass is converted into the final product, reducing waste disposal costs. Additionally, the use of earth-abundant additives like acetates instead of expensive ligands further drives down the bill of materials. These cumulative efficiencies result in substantial cost savings that can be passed down the value chain, enhancing the competitiveness of the final API.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. Because the process does not require strictly anhydrous or anaerobic conditions, the risk of batch failure due to environmental fluctuations is minimized. This reliability ensures consistent output and predictable lead times, which are critical metrics for supply chain planning. Moreover, the broad substrate scope means that if a specific substituted precursor becomes unavailable, alternative analogs can often be synthesized using the same platform technology without re-validating the entire process, providing a buffer against raw material volatility.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is facilitated by its thermal safety profile. Operating at moderate temperatures (60-120°C) avoids the hazards associated with exothermic runaway reactions common in traditional trifluoromethylation methods. The reduced solvent usage and higher yields align with green chemistry principles, lowering the environmental footprint of the manufacturing process. This compliance with increasingly stringent environmental regulations future-proofs the production line and reduces the regulatory burden associated with waste management and emissions reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this methodology for their specific projects.
Q: What are the key advantages of this Rh-catalyzed synthesis method?
A: The method described in patent CN116003326A offers a one-pot cascade reaction under mild conditions (80°C) and air atmosphere. It eliminates the need for inert gas protection and complex multi-step sequences, significantly simplifying the operational procedure while maintaining high atom economy and functional group tolerance.
Q: How does the solvent choice affect the product outcome?
A: The reaction exhibits excellent divergent selectivity based on solvent polarity. Using standard organic solvents like dichloroethane yields the enol ester derivative (Product 3), whereas switching to fluorinated alcohols like trifluoroethanol or hexafluoroisopropanol directs the reaction towards the trifluoroethyl ester derivative (Product 4), allowing precise control over the final molecular architecture.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the protocol is highly scalable due to its robustness. It utilizes commercially available raw materials, operates under air without stringent moisture exclusion, and tolerates a wide range of substituents including halogens and electron-withdrawing groups, making it ideal for reliable pharmaceutical intermediate supplier operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Indazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN116003326A. As a dedicated partner in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of handling complex organometallic chemistry, ensuring that every batch of trifluoromethyl indazole derivatives meets stringent purity specifications. We understand that transitioning a novel patent technology to commercial reality requires more than just chemical knowledge; it demands a holistic approach to process safety, quality assurance, and regulatory compliance.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this efficient synthesis route. Whether you require custom synthesis for clinical trials or bulk manufacturing for commercial launch, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this technology can optimize your supply chain and accelerate your time to market.
