Advancing Pharmaceutical Intermediates: Efficient Synthesis of Trifluoromethyl Pyrazole Spirocyclopropanes
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to access complex spirocyclic scaffolds, particularly those incorporating fluorine motifs which are known to enhance metabolic stability and bioavailability. Patent CN114349704A introduces a groundbreaking methodology for the synthesis of trifluoromethyl-conjugated pyrazole spirocyclopropane compounds. This innovation addresses a critical gap in organic synthesis by providing a direct, one-pot tandem reaction that constructs these valuable architectures from readily available starting materials. The significance of this technology lies not only in its chemical elegance but also in its potential to streamline the supply chain for high-value intermediates used in drug discovery and development. By leveraging a copper-catalyzed oxidative coupling strategy, this method achieves high atom economy and operates under remarkably mild conditions, positioning it as a superior alternative to traditional multi-step syntheses.
This technical insight report analyzes the proprietary data within CN114349704A to evaluate its impact on manufacturing costs, process scalability, and supply chain reliability. For R&D directors, the robustness of the reaction across a wide substrate scope offers unprecedented flexibility in library design. For procurement and supply chain leaders, the use of commodity chemicals and ambient air as an oxidant presents a compelling case for cost reduction and operational simplicity. As a reliable pharmaceutical intermediate supplier, understanding these underlying technological shifts is essential for maintaining competitive advantage in the global market.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of spirocyclic pyrazole frameworks has been a formidable challenge in organic synthesis, often requiring harsh reaction conditions, expensive transition metal catalysts, or tedious multi-step sequences. Conventional approaches frequently rely on pre-functionalized substrates that are costly to produce and difficult to source in bulk quantities. Furthermore, many existing methods necessitate the use of inert atmospheres, such as nitrogen or argon, to prevent catalyst deactivation or side reactions, which adds significant complexity and expense to the manufacturing process. The introduction of trifluoromethyl groups into these structures often involves separate fluorination steps using hazardous reagents, further complicating the safety profile and environmental footprint of the synthesis. These limitations collectively result in prolonged lead times, lower overall yields, and higher production costs, creating bottlenecks for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
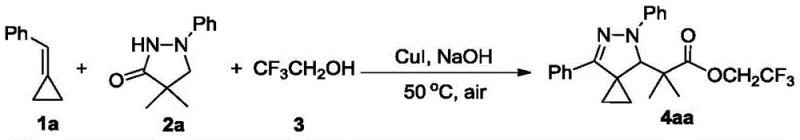
In stark contrast, the methodology disclosed in CN114349704A revolutionizes this landscape by employing a one-pot tandem reaction that seamlessly integrates cyclopropanation, oxidation, and esterification steps. The process utilizes substituted methylenecyclopropanes and 1-substituted pyrazolidinones as simple building blocks, reacting them directly with trifluoroethanol which serves dual roles as both a reactant and a solvent. This convergence of steps eliminates the need for isolating unstable intermediates, thereby maximizing material throughput and minimizing waste generation. The reaction proceeds efficiently under air atmosphere, utilizing molecular oxygen as the terminal oxidant, which is a green and economically advantageous feature. By avoiding the need for specialized equipment to maintain inert conditions, this novel approach drastically simplifies the operational requirements for manufacturing facilities, making it highly attractive for cost reduction in API manufacturing.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The core of this synthetic breakthrough lies in the synergistic interaction between the copper catalyst and the inorganic base under oxidative conditions. The proposed mechanism involves the activation of the methylenecyclopropane ring by the copper species, facilitating a ring-opening or functionalization event that allows for the subsequent attachment of the pyrazole moiety. The presence of trifluoroethanol is critical, as it participates in the transesterification or direct esterification of the intermediate carboxylic acid species formed during the oxidative process. The use of air as the oxidant suggests a radical pathway or a copper-oxygen species mediated cycle that regenerates the active catalyst, ensuring turnover without the accumulation of stoichiometric metal waste. This mechanistic efficiency is evidenced by the broad scope of the reaction, tolerating electron-withdrawing and electron-donating groups alike.
Impurity control is inherently managed by the selectivity of the copper catalytic system. The mild reaction temperature of 50°C prevents thermal degradation of sensitive functional groups, while the specific choice of ligands and additives minimizes side reactions such as polymerization of the cyclopropane or over-oxidation of the pyrazole ring. The patent data indicates that optimizing the base and additive combination is key to suppressing byproduct formation. For instance, the use of sodium hydroxide paired with copper iodide provides a balanced pH environment that promotes the desired transformation while neutralizing acidic byproducts. This level of control ensures that the resulting crude product requires minimal purification, often achievable through standard silica gel chromatography, which is a significant advantage for maintaining high purity specifications in a commercial setting.
How to Synthesize Trifluoromethyl Pyrazole Spirocyclopropane Efficiently
To implement this synthesis effectively, precise adherence to the optimized reaction parameters is essential. The process begins with the careful selection of high-purity starting materials to ensure consistent reaction kinetics. The molar ratios of the methylenecyclopropane, pyrazolidinone, base, and catalyst must be maintained within the specified ranges to achieve maximum conversion. The reaction mixture should be heated gradually to the target temperature to avoid exothermic spikes that could compromise safety or selectivity. Detailed standardized operating procedures for this synthesis are provided below to guide technical teams in replicating these results accurately.
- Mix substituted methylenecyclopropane, 1-substituted pyrazolidinone, inorganic base (NaOH), and copper iodide additive in trifluoroethanol.
- Seal the reaction vessel under air atmosphere and heat to 50°C for 12 hours with stirring.
- Quench with saturated ammonium chloride, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers transformative benefits for procurement strategies and supply chain resilience. The primary driver of value is the substantial reduction in raw material costs achieved by replacing expensive noble metal catalysts and specialized fluorinating agents with commodity copper salts and trifluoroethanol. This shift not only lowers the direct cost of goods sold but also mitigates the risk associated with the price volatility of precious metals. Furthermore, the ability to run the reaction under air atmosphere eliminates the capital expenditure and operational costs related to inert gas systems, allowing for more flexible deployment of manufacturing capacity across existing facilities without major retrofitting.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or rhodium catalysts in favor of abundant copper iodide represents a significant economic advantage. Additionally, the one-pot nature of the reaction reduces solvent consumption and energy usage by removing intermediate isolation and drying steps. This streamlined workflow translates directly into lower utility bills and reduced labor hours per kilogram of product. The high atom economy ensures that a greater proportion of the input mass ends up in the final product, minimizing waste disposal costs which are increasingly stringent in modern chemical manufacturing environments.
- Enhanced Supply Chain Reliability: The starting materials, including substituted methylenecyclopropanes and pyrazolidinones, are derived from common chemical feedstocks that are widely available from multiple global suppliers. This diversity in sourcing options reduces dependency on single-source vendors and protects against supply disruptions. The robustness of the reaction conditions means that production can be maintained even if slight variations in raw material quality occur, providing a buffer against supply chain fluctuations. Consequently, lead times for high-purity intermediates can be significantly shortened, enabling faster response to market demands.
- Scalability and Environmental Compliance: The mild thermal profile of the reaction, operating at just 50°C, makes it inherently safer and easier to scale from laboratory to pilot and eventually to full commercial production. There is no need for cryogenic cooling or high-pressure reactors, which simplifies engineering requirements. Moreover, the use of air as an oxidant aligns with green chemistry principles by avoiding stoichiometric oxidants that generate heavy metal waste. This environmental compatibility facilitates regulatory approval and supports corporate sustainability goals, making the process future-proof against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders assess the feasibility of integrating this method into their existing production pipelines.
Q: What are the optimal reaction conditions for synthesizing compound 4aa?
A: According to patent CN114349704A, the optimal conditions involve using CuI as the additive, NaOH as the base, in trifluoroethanol solvent at 50°C under air atmosphere for 12 hours, yielding 68%.
Q: Can this synthesis method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating substituents such as halogens, nitro groups, alkoxy groups, and alkyl chains on both phenyl rings without significant yield loss.
Q: Is this process suitable for large-scale manufacturing?
A: The process utilizes inexpensive inorganic bases and copper catalysts under air atmosphere, avoiding expensive noble metals or inert gas requirements, which significantly enhances its viability for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Pyrazole Spirocyclopropane Supplier
The technological advancements detailed in CN114349704A underscore the potential for producing high-value spirocyclic intermediates with exceptional efficiency and quality. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative synthetic routes for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into reliable industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to explore the full potential of these trifluoromethyl-conjugated compounds for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your project timelines while optimizing your budget.
