Advanced Green Synthesis of Alkyl Acid Testosterone for Industrial Pharmaceutical Applications
Advanced Green Synthesis of Alkyl Acid Testosterone for Industrial Pharmaceutical Applications
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly in the complex realm of steroid hormone synthesis. Patent CN111848713A introduces a groundbreaking preparation method for alkyl acid testosterone that directly addresses the critical pain points of environmental pollution and operational inefficiency found in legacy technologies. By leveraging a novel esterification strategy that utilizes water-insoluble organic solvents, this innovation enables a streamlined workflow that significantly reduces wastewater generation while maintaining exceptional product quality. For R&D directors and procurement leaders seeking a reliable pharmaceutical intermediates supplier, this technology represents a vital evolution in the production of high-value hormonal APIs, offering a pathway to both regulatory compliance and enhanced economic margins through solvent recovery systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of testosterone derivatives has relied heavily on acyl chloride methods that utilize water-soluble organic solvents, a approach fraught with significant logistical and environmental drawbacks. As detailed in prior art such as CN105732754A, these traditional routes necessitate the addition of large volumes of water post-reaction to precipitate the product, a step that inevitably generates massive quantities of acidic wastewater contaminated with residual solvents. This not only creates a severe burden on waste treatment facilities but also renders solvent recovery economically unfeasible, leading to inflated raw material costs and a larger carbon footprint. Furthermore, alternative methods described in patents like CN103910777A often require cumbersome post-treatment steps involving acid scavengers such as alumina or calcium oxide, which complicate the filtration process and introduce potential sources of particulate contamination that can compromise the final purity profile of the active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN111848713A revolutionizes the production landscape by employing water-insoluble organic solvents such as dichloromethane or dichloroethane, fundamentally altering the workup dynamics. Instead of relying on water precipitation, the reaction mixture can be directly subjected to phase separation after a simple water quench, allowing the organic layer containing the product to be easily isolated while the aqueous waste layer is minimized. This strategic shift not only drastically cuts down the volume of hazardous waste requiring disposal but also facilitates the efficient recycling of the organic solvent, thereby closing the loop on material usage and driving down variable production costs. The result is a robust, scalable process that delivers a total molar yield exceeding 85%, proving that environmental stewardship and high-efficiency manufacturing are not mutually exclusive goals in modern hormone manufacturing.
Mechanistic Insights into DMAP-Catalyzed Esterification
The core of this synthetic breakthrough lies in the precise orchestration of a nucleophilic catalysis cycle driven by 4-dimethylaminopyridine (DMAP) within a non-polar environment. In this mechanism, the DMAP catalyst acts as a potent acyl transfer agent, reacting rapidly with the alkyl acyl chloride to form a highly reactive N-acylpyridinium intermediate that is far more electrophilic than the parent acid chloride. This activated species then efficiently attacks the sterically hindered 17β-hydroxyl group of the testosterone backbone, overcoming the kinetic barriers that often plague steroid functionalization. The use of organic bases such as pyridine, triethylamine, or DIPEA serves a dual purpose: neutralizing the hydrochloric acid byproduct to drive the equilibrium forward and maintaining the catalyst in its active free-base form, ensuring consistent reaction rates even at the lower temperature ranges of 5°C to 40°C specified in the protocol.
From an impurity control perspective, this mechanistic pathway offers superior selectivity compared to uncatalyzed thermal esterifications, which often promote degradation of the sensitive enone system in the A-ring of the steroid nucleus. By operating under mild thermal conditions and utilizing a highly specific catalytic cycle, the formation of side products such as elimination byproducts or over-acylated species is effectively suppressed. The subsequent workup, involving a simple aqueous wash, efficiently removes the amine salts and residual catalyst without the need for aggressive chromatographic purification, resulting in a crude product with purity levels consistently above 99%. This high level of chemical fidelity is crucial for meeting the stringent specifications required for high-purity pharmaceutical intermediates intended for human therapeutic use.
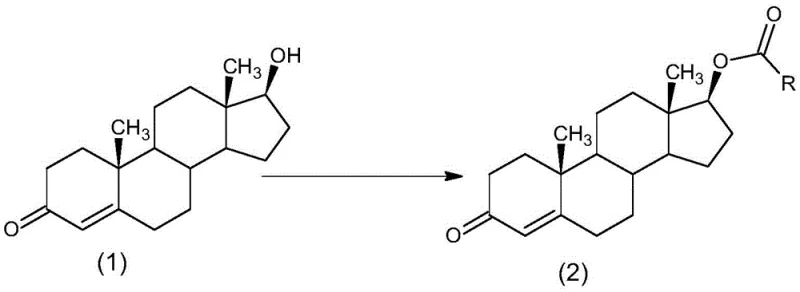
How to Synthesize Alkyl Acid Testosterone Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and stoichiometric balance to maximize the benefits of the green chemistry design. The process begins by dissolving the steroid substrate in a water-immiscible solvent under an inert atmosphere to prevent moisture interference, followed by the sequential addition of the organic base and the DMAP catalyst. Once the reaction mixture is homogenized, the alkyl acyl chloride is introduced at a controlled rate to manage the exotherm, ensuring the temperature remains within the optimal 5°C to 40°C window for complete conversion. While the general principles are straightforward, achieving reproducible high yields on a commercial scale demands precise control over mixing dynamics and quenching protocols, details of which are critical for process engineers to master.
- Dissolve 17β-hydroxyandrost-4-en-3-one in a water-insoluble organic solvent such as dichloromethane under inert gas protection.
- Add an organic base like pyridine or triethylamine along with a catalytic amount of DMAP to the reaction mixture.
- Introduce the alkyl acyl chloride at controlled temperatures (5-40°C), then quench with water and separate layers to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible operational improvements that extend far beyond the laboratory bench. The primary economic driver is the drastic simplification of the isolation process; by eliminating the need for water precipitation and the associated filtration of wet cakes, manufacturers can significantly reduce processing time and energy consumption associated with drying operations. Moreover, the ability to recycle the organic solvent creates a closed-loop system that insulates the production cost from volatile fluctuations in solvent market prices, providing a stable and predictable cost structure for long-term supply contracts. This efficiency gain is compounded by the reduction in waste disposal fees, as the volume of hazardous acidic wastewater is minimized, aligning production costs with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The elimination of water-soluble solvents and the subsequent ability to recycle dichloromethane or dichloroethane leads to substantial raw material savings over the lifecycle of the product. By avoiding the loss of solvent into aqueous waste streams, the effective consumption of chemicals per kilogram of API is lowered, directly improving the gross margin profile. Additionally, the high reaction yield exceeding 85% ensures that the expensive steroid starting material is utilized with maximum efficiency, reducing the cost of goods sold and enhancing the overall competitiveness of the supply chain.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route, characterized by mild reaction conditions and tolerance to standard industrial reagents, ensures high batch-to-batch consistency and reduces the risk of production failures. The simplified workup procedure reduces the dependency on specialized filtration equipment or extensive chromatography columns, allowing for faster turnaround times between batches. This agility enables suppliers to respond more rapidly to demand spikes, ensuring a continuous flow of high-purity hormonal intermediates to downstream formulation partners without the bottlenecks typical of older, more labor-intensive technologies.
- Scalability and Environmental Compliance: Designed with industrial scale-up in mind, this process avoids the engineering challenges associated with handling large volumes of slurry during water precipitation, making it ideal for reactor trains ranging from pilot plant to multi-ton production. The significant reduction in wastewater generation simplifies the environmental permitting process and lowers the operational burden on effluent treatment plants, ensuring long-term regulatory compliance. This sustainability advantage future-proofs the supply chain against tightening environmental laws, securing the license to operate for decades to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced esterification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new method improve environmental compliance compared to traditional acyl chloride methods?
A: Unlike conventional methods that use water-soluble solvents requiring massive water precipitation and generating acidic wastewater, this patent utilizes water-insoluble solvents like DCM. This allows for direct phase separation and solvent recycling, drastically reducing wastewater volume and environmental impact.
Q: What represents the key advantage in terms of yield and purity for this synthesis route?
A: The process achieves a total molar yield exceeding 85% with product purity levels often surpassing 99%. The use of DMAP as a catalyst ensures efficient conversion while minimizing side reactions, resulting in a cleaner crude profile that simplifies downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing of testosterone esters?
A: Yes, the methodology is specifically designed for industrial scalability. The simplified workup procedure involving layer separation rather than filtration of precipitates, combined with the ability to recycle expensive organic solvents, makes it highly economically viable for metric-ton production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Acid Testosterone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is critical for the long-term viability of the pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111848713A are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of testosterone ester meets the highest global standards for potency and impurity profiles, giving our partners absolute confidence in product quality.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your hormone manufacturing operations.
