Advanced Synthesis of 21-Position Carboxylate Steroids for Oncology Drug Development
The pharmaceutical industry's relentless pursuit of potent anticancer agents has placed steroidal compounds at the forefront of oncology research, particularly those targeting estrogen-dependent tumors such as breast and uterine cancer. Patent CN101676296B introduces a groundbreaking methodology for the preparation of 21-position carboxylate steroid compounds, represented generally by Formula II, which exhibit significant antitumor activity comparable to natural extracts but with superior manufacturability. This technical disclosure addresses the critical bottleneck of sourcing rare bioactive steroids from marine organisms like Spongodes sp., offering a robust chemical synthesis pathway that ensures supply chain stability for drug developers. By leveraging a streamlined six-step transformation starting from accessible pregnane derivatives, this innovation provides a reliable pharmaceutical intermediates supplier with the capability to deliver high-purity materials essential for preclinical and clinical trials. The strategic value of this patent lies not only in the biological efficacy of the resulting molecules but also in the economic and operational feasibility of producing them on an industrial scale.
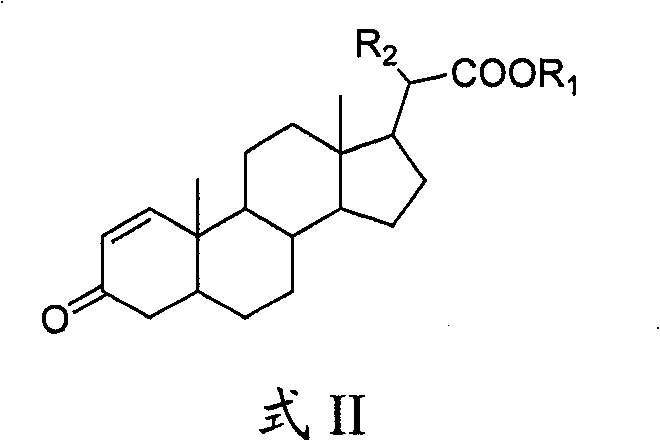
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of 21-position carboxylate steroids relied heavily on the isolation of trace amounts from natural marine sources, a process fraught with logistical and economic inefficiencies. Natural extraction is inherently limited by the seasonal availability of soft corals, the geographical constraints of harvesting in the South China Sea, and the extremely low concentration of the target compound within the biological matrix. Furthermore, the purification of natural extracts often requires extensive chromatographic separation to remove structurally similar impurities, leading to substantial material loss and inconsistent batch-to-batch quality. From a regulatory perspective, relying on natural sources introduces variability in the impurity profile that complicates the validation of analytical methods required for New Drug Applications (NDAs). Consequently, the conventional approach fails to meet the rigorous demands of modern cost reduction in pharmaceutical intermediates manufacturing, as the cost of goods sold (COGS) remains prohibitively high for widespread therapeutic application.
The Novel Approach
The methodology disclosed in CN101676296B revolutionizes the production landscape by establishing a fully synthetic route that bypasses the uncertainties of natural extraction. The core innovation involves a sequential modification of the steroid nucleus, beginning with the protection of the 3-hydroxyl group followed by a highly selective alkylation at the C21 position. This approach allows for the precise installation of diverse side chains, enabling the generation of a library of analogues for structure-activity relationship (SAR) studies without the need for new natural sources. The subsequent oxidative and eliminative steps efficiently construct the conjugated enone system crucial for biological activity, utilizing standard reagents that are commercially available in bulk quantities. This shift from extraction to synthesis represents a paradigm shift in commercial scale-up of complex pharmaceutical intermediates, transforming a scarce natural product into a commodity chemical that can be produced with predictable yields and defined specifications.
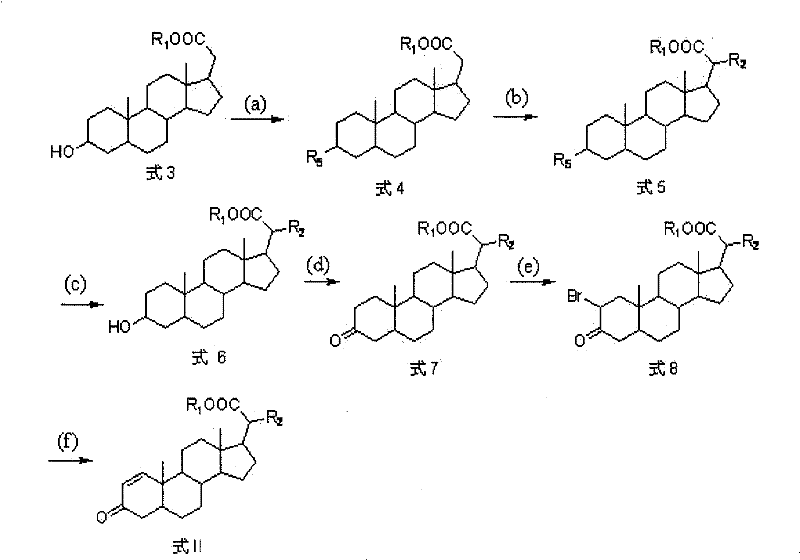
Mechanistic Insights into Steroid Side-Chain Alkylation and Enone Formation
The chemical elegance of this synthesis is anchored in the precise control of reactivity at the C21 position, a challenging task given the presence of multiple acidic protons on the steroid skeleton. The process initiates with the silylation of the 3-hydroxyl group using tert-butyldimethylchlorosilane (TBDMSCl), which serves a dual purpose: it prevents unwanted oxidation at the A-ring during subsequent steps and enhances the solubility of the intermediate in organic solvents. The pivotal alkylation step employs a strong base system generated in situ from n-butyllithium and diisopropylamine in the presence of hexamethylphosphoric triamide (HMPA). This specific combination facilitates the formation of a kinetic enolate at the C21 methyl group rather than the thermodynamically more stable enolates elsewhere on the ring system. The addition of C1 to C10 halogenated alkanes then proceeds via an SN2 mechanism, extending the side chain with high regioselectivity. Following deprotection of the silyl ether, the 3-hydroxyl group is oxidized to a ketone using chromium-based oxidants, setting the stage for the final structural modification.
The introduction of the Δ1-double bond is achieved through a two-step bromination-dehydrobromination sequence that exemplifies classic steroid functionalization strategies. Treatment of the 3-keto intermediate with pyridinium hydrobromide perbromide results in alpha-bromination at the C2 position, activated by the adjacent carbonyl. The subsequent elimination of hydrogen bromide using lithium carbonate and lithium bromide in a polar aprotic solvent like DMF drives the formation of the conjugated enone system. This specific arrangement is critical for the compound's interaction with steroid receptors and its antitumor potency. From an impurity control perspective, the use of lithium salts in the elimination step helps to suppress competing elimination pathways that could lead to non-conjugated isomers, thereby ensuring a clean impurity profile. This mechanistic understanding is vital for a reliable pharmaceutical intermediates supplier to maintain stringent quality control standards during the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 21-Position Carboxylate Steroids Efficiently
The execution of this synthetic route requires careful attention to reaction conditions, particularly temperature control during the lithiation step and the choice of solvents for the elimination reaction. The process is designed to be modular, allowing manufacturers to optimize individual steps based on their specific equipment capabilities and raw material availability. For instance, the oxidation step can be adapted to use greener oxidants if environmental compliance is a priority, while the core alkylation logic remains unchanged. The detailed standardized synthesis steps provided in the patent offer a clear roadmap for process chemists to replicate the results, ensuring that the transition from laboratory bench to pilot plant is seamless. By adhering to the specified molar ratios and reaction times, producers can achieve consistent yields that validate the economic viability of the method.
- Protect the 3-hydroxyl group of the starting steroid (Formula 3) using tert-butyldimethylchlorosilane to form the silyl ether (Formula 4).
- Perform alkylation at the C21 position using n-butyllithium and diisopropylamine with a C1-C10 halogenated alkane to generate Formula 5.
- Remove the silyl protecting group under acidic or fluoride conditions to yield the hydroxy intermediate (Formula 6), followed by oxidation to the ketone (Formula 7).
- Introduce the double bond via bromination with pyridinium hydrobromide perbromide to form Formula 8, followed by dehydrobromination using lithium carbonate to obtain the final enone (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers transformative benefits that extend beyond simple cost metrics. The primary advantage lies in the decoupling of production from biological sources, which eliminates the volatility associated with harvest seasons, weather patterns, and geopolitical risks in marine resource regions. This stability ensures a continuous supply of critical intermediates, reducing lead time for high-purity pharmaceutical intermediates and preventing production stoppages in downstream drug manufacturing facilities. Furthermore, the use of commodity chemicals such as TBDMSCl, n-butyllithium, and common solvents means that the raw material supply chain is robust and diversified, reducing the risk of single-source bottlenecks that often plague specialty chemical procurement.
- Cost Reduction in Manufacturing: The synthetic route significantly lowers the cost of goods by replacing expensive and inefficient extraction processes with high-yield chemical transformations. The elimination of complex chromatographic purifications required for natural extracts reduces solvent consumption and waste disposal costs, contributing to substantial cost savings. Additionally, the ability to synthesize the compound from inexpensive pregnane precursors allows for better margin management compared to purchasing rare natural isolates. The streamlined nature of the six-step sequence minimizes unit operations, further driving down operational expenditures in large-scale production environments.
- Enhanced Supply Chain Reliability: By establishing a fully synthetic supply chain, manufacturers can guarantee the availability of 21-position carboxylate steroids regardless of external ecological factors. This reliability is crucial for long-term drug development projects where consistency of supply is a regulatory requirement. The scalability of the process means that suppliers can rapidly ramp up production volumes to meet surging demand during clinical trial phases or commercial launch without the lead times associated with cultivating or harvesting biological materials. This agility provides a strategic buffer against market fluctuations and ensures business continuity for pharmaceutical partners.
- Scalability and Environmental Compliance: The reaction conditions employed in this patent are well-suited for industrial scale-up, utilizing standard reactor types and temperature ranges that do not require specialized cryogenic or high-pressure equipment. The process generates manageable waste streams that can be treated using conventional effluent handling systems, aligning with increasingly strict environmental regulations in the fine chemical sector. The avoidance of heavy metal catalysts in the key coupling steps simplifies the removal of trace metals from the final product, facilitating compliance with ICH Q3D guidelines for elemental impurities in drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these steroid intermediates. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners evaluating this technology. Understanding these nuances is essential for integrating this intermediate into existing drug discovery pipelines and ensuring that quality expectations are met from the outset of the partnership.
Q: What are the key advantages of this synthetic route over natural extraction?
A: This synthetic method avoids the low yields and complex purification associated with extracting rare steroids from marine sponges. It utilizes readily available pregnane derivatives and standard organic reagents, ensuring consistent supply and higher purity suitable for pharmaceutical applications.
Q: How is regioselectivity controlled during the alkylation step?
A: The process employs a specific base system comprising n-butyllithium and diisopropylamine in hexamethylphosphoric triamide (HMPA) at low temperatures (-78°C). This conditions the enolate formation specifically at the C21 position, minimizing side reactions at other active sites on the steroid nucleus.
Q: Is this process scalable for commercial production?
A: Yes, the reaction conditions utilize common solvents like THF and DMF and operate at manageable temperatures ranging from -78°C to reflux. The elimination of exotic catalysts and the use of robust chemical transformations make it highly amenable to scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 21-Position Carboxylate Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is executed with precision. We are committed to delivering 21-position carboxylate steroids that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in steroid chemistry allows us to troubleshoot potential scale-up issues proactively, guaranteeing a supply of material that supports your regulatory filings and clinical timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this synthetic source. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements and quality standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
