Revolutionizing Cefotiam Side Chain Production: A High-Purity Tetrazole Intermediate Strategy
The pharmaceutical industry continuously seeks robust synthetic pathways for critical beta-lactam antibiotic intermediates, and patent CN101817796A presents a transformative approach to producing the cefotiam side chain, chemically known as 1-(2-dimethylaminoethyl)-5-mercapto tetrazole. This intellectual property discloses a novel methodology that fundamentally alters the traditional synthetic landscape by replacing hazardous methylation steps with a controlled oxidative cyclization process. By utilizing hydrogen peroxide as a key oxidant instead of toxic alkylating agents, the invention addresses long-standing safety and efficiency concerns inherent in tetrazole chemistry. The technical breakthrough lies in the seamless integration of dithiocarbamate formation followed by direct oxidation and azide-mediated ring closure, which collectively streamline the production workflow. For R&D directors and process chemists, this represents a significant opportunity to enhance the purity profile of the final API intermediate while mitigating the risks associated with handling volatile ethers and methyl iodide. The patent explicitly highlights the advantages of simple operational steps, reduced raw material costs, and the attainment of high product purity, making it a cornerstone technology for modern cephalosporin manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-(2-dimethylaminoethyl)-5-mercapto tetrazole has relied on a multi-step sequence involving the addition of carbon disulfide to N,N-dimethyl ethylenediamine, followed by a methylation reaction and subsequent cyclization with sodium azide. As illustrated in the reaction scheme below, the conventional route necessitates the use of diethyl ether as a solvent during the addition phase, which introduces severe explosion hazards and operational difficulties due to the solvent's high volatility and low flash point. Furthermore, the methylation step requires the use of strong bases to dissolve the dithiocarbamic acid intermediate, followed by the addition of methylating reagents, creating a complex reaction environment that demands rigorous temperature control. Post-reaction processing in the traditional method is equally burdensome, requiring distillation for solvent recovery, liquid-liquid extraction with organic solments, and multiple recrystallization steps to isolate the methyl ester intermediate. These cumulative inefficiencies result in a relatively low overall yield, reported in prior art to be approximately 76%, and generate significant chemical waste, thereby inflating both the environmental footprint and the cost of goods sold for manufacturers relying on this legacy technology.
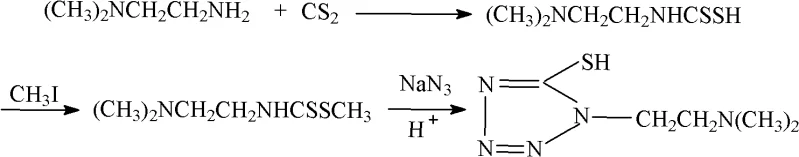
The Novel Approach
In stark contrast to the cumbersome legacy processes, the novel approach detailed in the patent data introduces a streamlined oxidative pathway that bypasses the need for explicit methylation reagents entirely. Instead of converting the dithiocarbamic acid into a methyl ester, the new method employs hydrogen peroxide to oxidize the intermediate directly within a water-insoluble organic solvent system, such as dichloromethane or ethyl acetate. This strategic shift eliminates the requirement for hazardous ether solvents and toxic methyl iodide, replacing them with safer, more industrially friendly alternatives that are easier to handle and recover. The reaction conditions are mild, typically maintained between 0°C and 40°C, which reduces energy consumption and minimizes the risk of thermal runaway incidents common in exothermic alkylation reactions. Following oxidation, the viscous residue is dissolved in methanol and reacted with aqueous sodium azide, facilitating a clean cyclization to form the tetrazole ring. This simplified workflow not only enhances operator safety but also drastically reduces the complexity of downstream processing, allowing for higher throughput and more consistent batch-to-batch quality in a commercial setting.
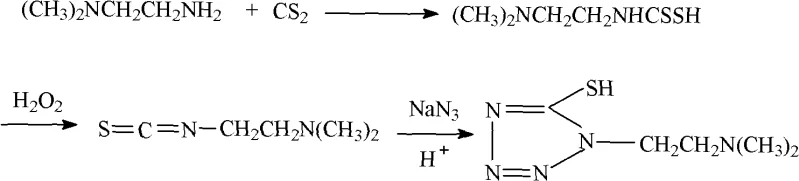
Mechanistic Insights into Oxidative Cyclization and Tetrazole Formation
The core mechanistic advantage of this patented process lies in the oxidative transformation of the dithiocarbamic acid derivative, which serves as a pivotal precursor for the tetrazole ring construction. In the presence of hydrogen peroxide, the sulfur-containing intermediate undergoes a selective oxidation that likely generates an reactive isothiocyanate-like species or a sulfenyl intermediate in situ, which is immediately primed for nucleophilic attack by the azide ion. This mechanism avoids the formation of stable methyl esters that require harsh conditions to cleave or rearrange, thereby preserving the integrity of the sensitive amine functionality on the ethyl side chain. The use of a biphasic or semi-biphasic system during the oxidation step, utilizing solvents like dichloromethane, allows for efficient heat dissipation and precise control over the oxidation state, preventing over-oxidation to sulfones or sulfonic acids which would act as difficult-to-remove impurities. By carefully controlling the stoichiometry of hydrogen peroxide, typically using a molar excess of 2 to 4 times relative to the substrate, the reaction ensures complete conversion of the starting material while maintaining a clean reaction profile that facilitates subsequent purification.
Impurity control is further enhanced by the specific sequence of workup operations designed to remove inorganic salts and organic byproducts effectively. After the cyclization reaction with sodium azide is complete, the mixture is treated with activated carbon, a critical step that adsorbs colored impurities and trace organic contaminants that could otherwise compromise the visual and chemical quality of the final product. The adjustment of the pH to a range of 4 to 6 using hydrochloric acid triggers the precipitation of the target tetrazole compound in its free acid or zwitterionic form, leveraging its solubility characteristics to separate it from the aqueous salt matrix. This acidification step is crucial for maximizing recovery yields, as evidenced by the experimental data showing total recoveries ranging from 60% to 64% with purities exceeding 99.3%. The ability to achieve such high purity through simple crystallization from water, rather than complex chromatographic separations, underscores the robustness of the chemical design and its suitability for GMP-compliant manufacturing environments where impurity profiles are strictly regulated.
How to Synthesize 1-(2-dimethylaminoethyl)-5-mercapto tetrazole Efficiently
The synthesis of this critical cefotiam side chain intermediate is executed through a highly optimized two-stage protocol that prioritizes safety, yield, and ease of operation. The process begins with the formation of the dithiocarbamic acid adduct under controlled low-temperature conditions, followed by an oxidative modification that sets the stage for ring closure. Detailed standard operating procedures for this synthesis, including specific reagent grades, agitation rates, and filtration parameters, are outlined in the comprehensive guide below to ensure reproducible results for process development teams. Adhering to these standardized steps allows manufacturers to replicate the high purity and yield metrics demonstrated in the patent examples, facilitating a smooth transition from laboratory scale to pilot plant operations.
- Dissolve carbon disulfide in an organic solvent and drip N,N-dimethyl ethylenediamine at 0-30°C to form the dithiocarbamic acid intermediate, followed by filtration.
- Oxidize the filtered intermediate using hydrogen peroxide in a water-insoluble organic solvent at 0-40°C, then evaporate the solvent to obtain a viscous residue.
- Dissolve the residue in methanol, drip into aqueous sodium azide at 30-70°C for cyclization, then decolorize and adjust pH to precipitate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this oxidative synthesis route offers profound strategic benefits that extend beyond mere technical feasibility. The elimination of methyl iodide, a reagent subject to strict regulatory controls and fluctuating market prices due to its toxicity and ozone-depleting potential, significantly de-risks the supply chain. By substituting this hazardous material with hydrogen peroxide, a commodity chemical with stable global availability and low cost, manufacturers can achieve substantial cost savings in raw material procurement without compromising on reaction efficiency. Furthermore, the removal of diethyl ether from the process solvent list mitigates the need for specialized explosion-proof infrastructure and reduces insurance premiums associated with storing large volumes of highly flammable liquids. These operational improvements translate directly into a more resilient supply chain capable of withstanding market volatility and regulatory shifts, ensuring continuous production of this vital antibiotic intermediate.
- Cost Reduction in Manufacturing: The economic impact of this process innovation is driven primarily by the simplification of the unit operations and the reduction in solvent consumption. Traditional methods require multiple distillation steps to recover ether and other organic solvents, which are energy-intensive and result in significant solvent loss; the new method utilizes solvents like ethyl acetate or dichloromethane which are easier to recover and recycle, thereby lowering utility costs. Additionally, the higher overall yield and purity reduce the burden on waste treatment facilities, as fewer byproducts and spent solvents need to be disposed of as hazardous waste. The avoidance of expensive methylating agents and the reduction in processing time contribute to a leaner manufacturing cost structure, enhancing the competitiveness of the final API in the global marketplace.
- Enhanced Supply Chain Reliability: Reliability in the supply of pharmaceutical intermediates is paramount, and this synthetic route bolsters security by relying on widely available, non-restricted raw materials. Hydrogen peroxide and sodium azide are produced at massive scales globally, ensuring that supply disruptions are unlikely compared to specialty reagents like methyl iodide which may have limited suppliers. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, means that production is less susceptible to minor variations in raw material quality or environmental conditions. This flexibility allows supply chain managers to source materials from a broader vendor base, fostering competition and further driving down costs while guaranteeing uninterrupted delivery schedules for downstream antibiotic manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden bottlenecks, but the oxidative nature of this reaction is inherently scalable due to its manageable exotherm and lack of gas evolution. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and halogenated waste streams. The ability to perform the reaction in common reactors without the need for specialized high-pressure or cryogenic equipment lowers the barrier to entry for contract manufacturing organizations looking to expand capacity. Consequently, the commercial scale-up of complex tetrazole intermediates becomes a straightforward engineering task rather than a scientific hurdle, enabling rapid response to market demand surges for cefotiam and related cephalosporins.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and product quality. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: Why is the hydrogen peroxide oxidation method superior to traditional methylation for cefotiam side chain synthesis?
A: The traditional method relies on methyl iodide and ether, which pose significant safety hazards and require complex solvent recovery. The new oxidative route using hydrogen peroxide eliminates toxic methylating agents, utilizes safer solvents like dichloromethane or ethyl acetate, and simplifies the workup process, leading to higher operational safety and reduced environmental impact.
Q: What purity levels can be achieved with this patented synthesis route?
A: According to the experimental data in patent CN101817796A, this method consistently achieves a product purity of greater than 99.3% after recrystallization. The simplified reaction pathway minimizes the formation of complex byproducts often associated with alkylation reactions, ensuring a cleaner impurity profile suitable for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is explicitly designed for industrial suitability. It avoids the use of highly volatile ether solvents and eliminates the need for difficult distillation steps required in the conventional methylation route. The use of common oxidants and straightforward filtration/crystallization steps makes the commercial scale-up of complex tetrazole intermediates significantly more feasible and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2-dimethylaminoethyl)-5-mercapto tetrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final pharmaceutical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering 1-(2-dimethylaminoethyl)-5-mercapto tetrazole with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the >99.3% purity benchmark established by advanced synthetic routes. Our facility is equipped to handle the specific solvent systems and oxidative conditions required for this process, guaranteeing a supply of cefotiam side chain that supports your regulatory filings and commercial launch timelines.
We invite global partners to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this oxidative synthesis method for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your antibiotic manufacturing operations.
