Scalable Synthesis of 3-Bromo-2-Methylthiophene and 2-Methyl-3-Thiophenecarboxylic Acid for Industrial Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for synthesizing critical heterocyclic intermediates, particularly those serving as building blocks for antibiotics and antiviral agents. Patent CN101987842A introduces a transformative methodology for preparing 2-methyl thiophene derivatives, specifically targeting the synthesis of 3-bromo-2-methylthiophene and its subsequent conversion to 2-methyl-3-thiophenecarboxylic acid. This technology addresses the longstanding inefficiencies associated with traditional organolithium chemistry by leveraging a selective metal powder reduction strategy. By shifting from hazardous, cryogenic butyllithium protocols to a thermally driven metal-mediated debromination, this invention offers a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing. The implications for supply chain stability and cost structure in the production of cephalosporin intermediates are profound, marking a significant departure from legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-bromo-2-methylthiophene (Compound 3) has relied heavily on the use of n-butyllithium (n-BuLi) to effect halogen-metal exchange or directed lithiation. As illustrated in the conventional synthetic route, this approach necessitates extremely low reaction temperatures, often ranging from -40°C to -65°C, to maintain control over regioselectivity and prevent decomposition. 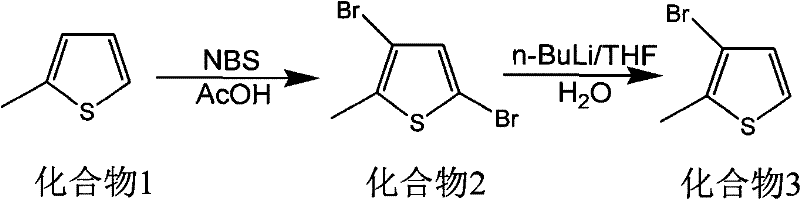 Such cryogenic conditions impose a massive energy burden on manufacturing facilities, requiring specialized refrigeration equipment and significantly increasing utility costs. Furthermore, n-butyllithium is pyrophoric and highly moisture-sensitive, introducing severe safety hazards and requiring rigorous exclusion of air and water, which complicates reactor design and operational procedures. The complexity of quenching these reactive intermediates and the subsequent workup often leads to lower overall yields and higher solvent consumption, rendering the process economically unattractive for ton-scale production.
Such cryogenic conditions impose a massive energy burden on manufacturing facilities, requiring specialized refrigeration equipment and significantly increasing utility costs. Furthermore, n-butyllithium is pyrophoric and highly moisture-sensitive, introducing severe safety hazards and requiring rigorous exclusion of air and water, which complicates reactor design and operational procedures. The complexity of quenching these reactive intermediates and the subsequent workup often leads to lower overall yields and higher solvent consumption, rendering the process economically unattractive for ton-scale production.
The Novel Approach
In stark contrast, the patented methodology utilizes a selective debromination strategy mediated by inexpensive metal powders such as zinc, iron, magnesium, or aluminum. This innovative route begins with the dibrominated precursor, 3,5-dibromo-2-methylthiophene (Compound 2), which is subjected to reduction in a polar organic solvent at elevated temperatures between 90°C and 110°C. 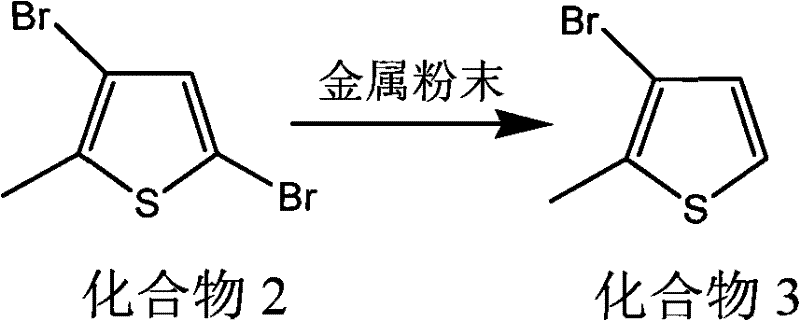 The use of metal powder allows for the selective removal of the bromine atom at the 5-position while retaining the bromine at the 3-position, a transformation that is difficult to achieve with high selectivity using other methods. This thermal process eliminates the need for cryogenic cooling entirely, allowing reactions to proceed under reflux conditions which are far easier to manage industrially. The simplicity of the workup, involving basic filtration to remove excess metal followed by vacuum distillation, drastically reduces processing time and solvent waste, positioning this method as a superior alternative for reliable pharmaceutical intermediate supplier operations.
The use of metal powder allows for the selective removal of the bromine atom at the 5-position while retaining the bromine at the 3-position, a transformation that is difficult to achieve with high selectivity using other methods. This thermal process eliminates the need for cryogenic cooling entirely, allowing reactions to proceed under reflux conditions which are far easier to manage industrially. The simplicity of the workup, involving basic filtration to remove excess metal followed by vacuum distillation, drastically reduces processing time and solvent waste, positioning this method as a superior alternative for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Metal-Mediated Selective Debromination
The core chemical innovation lies in the heterogeneous electron transfer mechanism facilitated by the metal surface. When Compound 2 is heated in the presence of activated metal powder and a proton source (such as acetic acid or formic acid), the metal acts as a reducing agent, donating electrons to the carbon-bromine bond. This single-electron transfer generates a radical anion intermediate which subsequently fragments to release a bromide ion and form a carbon-centered radical. The presence of the monobasic alkyl acid provides protons that rapidly quench the radical species at the 5-position, effectively replacing the bromine with hydrogen. This mechanism is highly dependent on the steric and electronic environment of the thiophene ring, which favors reduction at the 5-position over the 3-position due to the stabilizing influence of the adjacent methyl group and sulfur atom. Understanding this mechanistic nuance is critical for R&D directors aiming to optimize reaction parameters, as the choice of metal and acid catalyst directly influences the rate of electron transfer and the suppression of side reactions such as complete debromination or polymerization.
Following the formation of Compound 3, the pathway extends to the synthesis of 2-methyl-3-thiophenecarboxylic acid (Compound 4) via a Grignard carboxylation sequence. 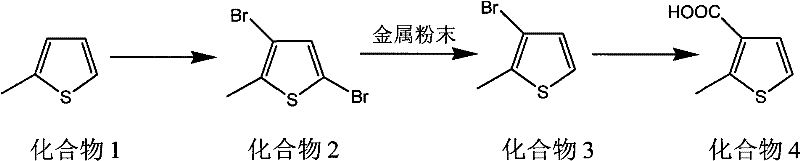 In this stage, the bromine atom at the 3-position serves as a handle for magnesium insertion, typically initiated by iodine or 1,2-dibromoethane in anhydrous THF. The resulting Grignard reagent is then reacted with carbon dioxide (dry ice) to install the carboxylic acid functionality. Unlike the initial lithiation step in prior art, this Grignard formation can be conducted at much milder temperatures (0°C to 65°C), further enhancing the safety profile. The impurity profile is tightly controlled because the starting material (Compound 3) is of high purity (>96%), and the Grignard reaction is highly specific. Any residual metal salts from the previous step are removed during the aqueous workup, ensuring that the final API intermediate meets stringent purity specifications required for downstream coupling reactions in antibiotic synthesis.
In this stage, the bromine atom at the 3-position serves as a handle for magnesium insertion, typically initiated by iodine or 1,2-dibromoethane in anhydrous THF. The resulting Grignard reagent is then reacted with carbon dioxide (dry ice) to install the carboxylic acid functionality. Unlike the initial lithiation step in prior art, this Grignard formation can be conducted at much milder temperatures (0°C to 65°C), further enhancing the safety profile. The impurity profile is tightly controlled because the starting material (Compound 3) is of high purity (>96%), and the Grignard reaction is highly specific. Any residual metal salts from the previous step are removed during the aqueous workup, ensuring that the final API intermediate meets stringent purity specifications required for downstream coupling reactions in antibiotic synthesis.
How to Synthesize 3-Bromo-2-Methylthiophene Efficiently
The operational protocol for this synthesis is designed to maximize throughput while minimizing technical risk. The process begins with the bromination of 2-methylthiophene to the dibromo intermediate, followed immediately by the metal reduction step without the need for isolating the unstable dibromo compound in some embodiments. This telescoping capability reduces unit operations and solvent usage. The reaction mixture is heated to reflux, monitored by GC or GC-MS to ensure complete consumption of the starting material, and then cooled to precipitate metal salts.
- Brominate 2-methylthiophene (Compound 1) using NBS or bromine in acetic acid to form 3,5-dibromo-2-methylthiophene (Compound 2).
- React Compound 2 with metal powder (Zn, Fe, Mg, or Al) in a polar solvent like THF at 90-110°C to selectively remove the 5-position bromine.
- Purify the resulting 3-bromo-2-methylthiophene (Compound 3) via filtration and vacuum distillation to achieve high purity suitable for downstream Grignard reactions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from organolithium chemistry to metal powder reduction represents a strategic opportunity to de-risk the supply of critical thiophene intermediates. The elimination of n-butyllithium removes a significant bottleneck related to the storage and handling of hazardous pyrophoric materials, which often require specialized logistics and insurance coverage. Furthermore, the reliance on commodity metals like zinc or iron powder ensures a stable and abundant supply of reagents, insulating the manufacturing process from the volatility often seen in the market for specialized organometallic reagents. This shift fundamentally alters the cost structure of production, moving away from high-cost, low-temperature batch processes toward robust, thermal-driven reactions that are inherently more scalable.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic reduction in energy consumption. By operating at reflux temperatures (approx. 105°C) instead of cryogenic conditions (-65°C), facilities can eliminate the need for expensive glycol chilling systems and the associated electricity loads. Additionally, the replacement of expensive n-butyllithium with low-cost metal powders significantly lowers the raw material bill of materials. The simplified workup procedure, which avoids complex chromatographic separations in favor of filtration and distillation, further reduces labor costs and solvent disposal fees, leading to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemical process translates directly into improved delivery reliability. Cryogenic reactions are prone to failure due to minor fluctuations in temperature control or moisture ingress, which can lead to batch losses and delayed shipments. The metal powder method is far more forgiving of minor operational variances, ensuring consistent batch-to-bystand quality and yield. Moreover, the use of common solvents like THF and acetic acid, combined with readily available metal reagents, means that production is less susceptible to supply disruptions of exotic chemicals, thereby securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route offers distinct advantages. The reaction generates fewer hazardous byproducts compared to the quenching of large quantities of organolithium reagents, simplifying wastewater treatment and waste disposal compliance. The ability to run the reaction at higher concentrations and elevated temperatures improves the volumetric productivity of the reactors, allowing manufacturers to produce larger quantities of high-purity pharmaceutical intermediates in existing infrastructure without the need for capital-intensive retrofitting for cryogenic capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical benefits of adopting this metal-mediated technology over traditional lithiation methods.
Q: Why is the metal powder reduction method superior to the traditional n-BuLi route?
A: The traditional route requires cryogenic conditions (-65°C) and hazardous n-butyllithium, leading to high energy costs and safety risks. The patented metal powder method operates at elevated temperatures (90-110°C) using inexpensive metals like zinc or iron, drastically simplifying operations and reducing safety hazards.
Q: What is the expected purity and yield for the final thiophene derivatives?
A: According to the patent data, the novel method achieves yields up to 80% for 3-bromo-2-methylthiophene and over 85% for 2-methyl-3-thiophenecarboxylic acid, with product purity consistently exceeding 95% after simple vacuum distillation.
Q: Can this process be scaled for commercial API intermediate production?
A: Yes, the process is specifically designed for industrialization. It eliminates the need for complex cryogenic reactors and uses robust heterogeneous reagents (metal powders), making it highly suitable for large-scale manufacturing of pharmaceutical intermediates like Cefoxitin precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-2-Methylthiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, safe, and scalable synthetic routes in the modern pharmaceutical landscape. Our technical team has extensively evaluated the metal powder reduction technology described in CN101987842A and integrated its principles into our manufacturing capabilities for thiophene derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial execution is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-bromo-2-methylthiophene or 2-methyl-3-thiophenecarboxylic acid meets the exacting standards required for GMP-grade API synthesis.
We invite global partners to leverage our expertise in process chemistry to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the superior economic and operational advantages of our thiophene intermediate offerings before committing to long-term supply agreements.
