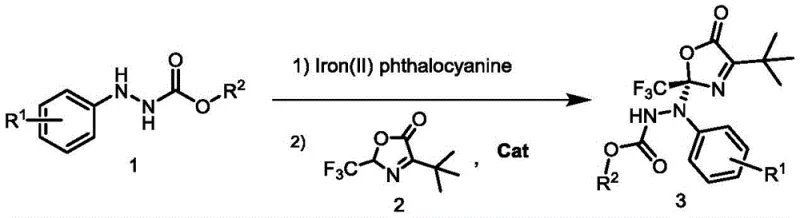
Scalable One-Pot Synthesis of Optically Active Benzocarboxylates for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing chiral centers, particularly those incorporating perfluoroalkyl groups which significantly enhance the metabolic stability and bioactivity of drug candidates. Patent CN113214180A introduces a groundbreaking one-pot two-step synthesis method for generating optically active benzocarboxylate compounds, addressing a critical gap in asymmetric synthesis technology. This innovative approach utilizes nitrogen acyl aryl hydrazine as a readily available starting material, which is first oxidized to an intermediate azobenzene compound in the presence of Iron(II) phthalocyanine. Subsequently, this intermediate undergoes an asymmetric addition reaction with oxazolone, catalyzed by a chiral bifunctional tertiary amine urea, to yield the final high-value product. The significance of this patent lies in its ability to merge oxidation and asymmetric C-N bond formation into a single operational sequence, thereby streamlining the production of complex chiral scaffolds essential for modern drug discovery.
This technological advancement represents a paradigm shift for manufacturers acting as a reliable pharmaceutical intermediate supplier, as it drastically simplifies the synthetic route compared to traditional multi-step processes. By leveraging earth-abundant iron catalysts and mild reaction conditions, the method not only improves atom economy but also aligns with green chemistry principles, reducing the environmental footprint associated with the production of high-purity API intermediates. The versatility of the reaction allows for various substituents on the aryl ring, including halogens and alkyl groups, providing a broad substrate scope that is invaluable for medicinal chemists exploring structure-activity relationships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitroxylamino derivatives containing perfluoroalkyl groups and amino groups has been fraught with significant challenges, particularly regarding the formation of carbon-nitrogen bonds at the 2-position of the azlactone ring. Conventional methods often rely on pre-formed azo compounds or require harsh oxidative conditions that are incompatible with sensitive functional groups, leading to poor yields and extensive byproduct formation. Furthermore, achieving high enantioselectivity in these transformations typically necessitates the use of expensive noble metal catalysts or stoichiometric amounts of chiral auxiliaries, which complicates downstream purification and inflates manufacturing costs. The lack of efficient, direct methods to access these chiral motifs has long been a bottleneck in the development of novel agrochemical and pharmaceutical agents, forcing R&D teams to settle for suboptimal synthetic routes that hinder rapid scale-up.
The Novel Approach
In stark contrast, the methodology disclosed in CN113214180A offers a streamlined solution by integrating the oxidation of hydrazines and the subsequent asymmetric addition into a seamless one-pot operation. This novel approach eliminates the need for isolating unstable azo intermediates, thereby reducing material loss and exposure to potentially hazardous compounds. The use of Iron(II) phthalocyanine as a biomimetic oxidation catalyst allows the reaction to proceed efficiently under ambient air at room temperature, a condition that is exceptionally rare for such transformative organic syntheses. Coupled with a highly effective chiral urea catalyst, this system delivers excellent enantioselectivity and yield, effectively overcoming the selectivity issues that have plagued previous attempts at azlactone functionalization. This represents a major leap forward in cost reduction in pharmaceutical intermediate manufacturing, as it minimizes solvent usage, energy consumption, and catalyst loading.
Mechanistic Insights into Fe(Pc)-Catalyzed Oxidation and Asymmetric Addition
The success of this transformation hinges on the synergistic interplay between the iron-based oxidation catalyst and the organocatalyst. In the first step, Iron(II) phthalocyanine activates molecular oxygen from the air to facilitate the dehydrogenative oxidation of the nitrogen acyl aryl hydrazine. This generates the reactive azobenzene intermediate in situ, which serves as the electrophilic partner for the subsequent addition. The choice of Iron(II) phthalocyanine is strategic; its macrocyclic structure stabilizes high-valent iron-oxo species necessary for hydrogen abstraction while remaining robust under the reaction conditions. This biomimetic oxidation pathway avoids the generation of toxic heavy metal waste streams often associated with traditional oxidants like chromium or manganese salts.
Following the oxidation, the chiral bifunctional tertiary amine urea catalyst takes center stage to control the stereochemistry of the C-N bond formation. As illustrated in the catalyst screening data, the specific architecture of the catalyst, particularly the cyclohexane backbone and the urea/thiourea moieties, is critical for inducing chirality. 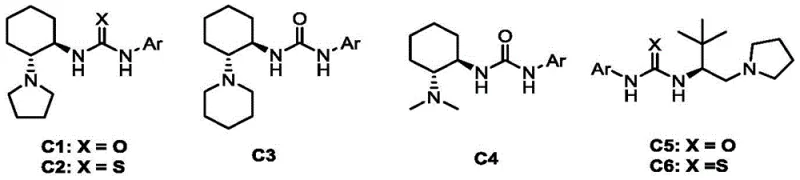
The catalyst likely operates through a dual-activation mode: the tertiary amine moiety deprotonates the azlactone to form a nucleophilic enolate, while the urea group hydrogen-bonds with the azo intermediate, organizing the transition state within a chiral pocket. This precise spatial arrangement ensures that the nucleophilic attack occurs exclusively from one face of the planar azo compound, resulting in the observed high enantiomeric excess (up to 96% ee). Understanding this mechanistic nuance is vital for process chemists aiming to further optimize reaction parameters or adapt the protocol to novel substrates.
How to Synthesize Optically Active Benzocarboxylates Efficiently
The practical implementation of this synthesis is remarkably straightforward, making it accessible for both laboratory-scale discovery and pilot-plant production. The protocol involves dissolving the hydrazine precursor in anhydrous mesitylene, followed by the addition of the iron catalyst and stirring under air to effect oxidation. Once the intermediate is formed, the chiral catalyst and oxazolone are introduced to trigger the asymmetric coupling.
- Oxidation Step: Dissolve nitrogen acyl aryl hydrazine in anhydrous mesitylene under air atmosphere. Add Iron(II) phthalocyanine (10 mol%) and stir at 25°C for 8 hours to generate the intermediate azobenzene compound.
- Asymmetric Addition Step: Slowly add oxazolone and chiral bifunctional tertiary amine urea catalyst (C3, 10 mol%) to the reaction mixture. Continue stirring at 25°C for 3 hours.
- Purification: Remove solvent under reduced pressure and purify the crude product via flash silica gel column chromatography using petroleum ether/ethyl acetate to obtain the final optically active benzocarboxylate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere chemical efficiency. The shift towards a one-pot process inherently reduces the number of unit operations required, which directly correlates to lower capital expenditure on equipment and reduced labor costs per kilogram of product. Furthermore, the reliance on air as the terminal oxidant and iron as the catalyst removes the supply chain volatility associated with precious metals like palladium or rhodium, whose prices can fluctuate wildly based on geopolitical factors. This stability ensures more predictable costing models for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction in solvent consumption due to the one-pot nature of the reaction lead to significant operational savings. By avoiding the isolation of intermediates, manufacturers can reduce waste disposal costs and minimize the loss of valuable material during transfer steps. The mild reaction conditions (25°C) also imply lower energy costs for heating or cooling, contributing to a leaner and more cost-effective production profile that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The starting materials, nitrogen acyl aryl hydrazines and oxazolones, are commercially available and structurally diverse, allowing for flexible sourcing strategies. The robustness of the catalyst system, which tolerates air and moisture better than many sensitive organometallic reagents, reduces the risk of batch failures due to minor environmental deviations. This reliability is crucial for maintaining continuous supply lines to downstream API manufacturers, preventing costly production stoppages.
- Scalability and Environmental Compliance: The use of Iron(II) phthalocyanine, a non-toxic and abundant metal complex, aligns perfectly with increasingly stringent environmental regulations regarding heavy metal residues in pharmaceutical products. The simplified workup procedure facilitates easier purification at scale, reducing the volume of organic solvents required for chromatography or crystallization. This eco-friendly profile not only simplifies regulatory filings but also positions the supply chain as sustainable, a key metric for modern corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers.
Q: What are the key advantages of using Iron(II) phthalocyanine in this synthesis?
A: Iron(II) phthalocyanine serves as an efficient, earth-abundant oxidation catalyst that operates under mild aerobic conditions (air, 25°C). This eliminates the need for expensive precious metal oxidants and harsh reaction environments, significantly lowering production costs and simplifying safety protocols for large-scale manufacturing.
Q: How does the chiral urea catalyst influence enantioselectivity?
A: The chiral bifunctional tertiary amine urea catalyst (specifically catalyst C3 derived from 1R,2R-cyclohexane-1,2-diamine) facilitates highly enantioselective addition to the azlactone. It creates a rigid chiral environment that directs the nucleophilic attack, achieving enantiomeric excess (ee) values up to 96%, which is critical for producing single-isomer pharmaceutical intermediates.
Q: Is this process suitable for commercial scale-up?
A: Yes, the process is highly scalable due to its one-pot nature, mild temperature requirements (room temperature), and use of air as the oxidant. The simple workup involving solvent removal and standard column chromatography translates well to industrial purification methods, ensuring consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Benzocarboxylates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the one-pot two-step synthesis method described in CN113214180A for producing high-value chiral intermediates. As a dedicated CDMO partner, we possess the technical expertise to translate this academic innovation into a robust commercial process. Our facilities are equipped to handle complex asymmetric syntheses, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of optically active benzocarboxylates meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your next drug development project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the market with high-quality, cost-effective chiral building blocks.
