Advancing Fluorine Chemistry: Scalable Nickel-Catalyzed Synthesis of Trifluoroethyl Intermediates
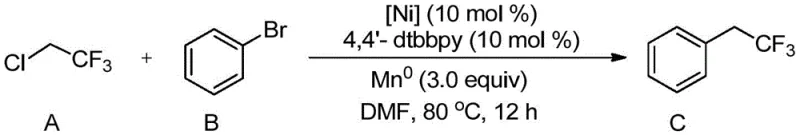
The introduction of fluorine atoms into organic molecules is a cornerstone strategy in modern medicinal chemistry and agrochemical design, primarily due to the unique ability of fluorine to modulate metabolic stability, lipophilicity, and binding affinity without significantly altering steric bulk. Patent CN110668913A discloses a groundbreaking preparation method for trifluoroethyl compounds that addresses long-standing inefficiencies in fluorine chemistry. This innovation enables the direct utilization of 2-chloro-1,1,1-trifluoroethane, an inexpensive and abundant industrial gas, as a trifluoroethylating agent. By employing a catalytic system comprising a nickel salt and a pyridine ligand, the method achieves efficient coupling with aryl and heteroaryl halides under remarkably mild conditions. This represents a significant paradigm shift from traditional stoichiometric methods, offering a sustainable and economically viable pathway for producing high-purity pharmaceutical intermediates and advanced material precursors.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl and heterocyclic aryl-2,2,2-trifluoroethyl compounds has been plagued by significant technical and economic hurdles. Traditional approaches often rely on the generation of trifluoromethyl copper species using excessive amounts of copper powder or cuprous iodide, which react with benzyl bromides. These methods typically require harsh reaction conditions, including high temperatures and prolonged reaction times, leading to energy inefficiency. Furthermore, the reliance on stoichiometric amounts of transition metals generates substantial heavy metal waste, complicating downstream purification and environmental compliance. Another prevalent method involves palladium-catalyzed reactions with 1,1,1-trifluoro-2-iodoethane; however, this reagent is derived from more expensive precursors and is inherently less stable. Additionally, classical Grignard-based couplings are severely limited by poor functional group tolerance, failing completely in the presence of active hydrogens, aldehydes, ketones, or esters, thus restricting the structural diversity accessible to process chemists.
The Novel Approach
The novel methodology described in the patent data overcomes these deficiencies through a cleverly designed nickel-catalyzed cross-coupling protocol. By activating the strong carbon-chlorine bond of 2-chloro-1,1,1-trifluoroethane directly, the process bypasses the need for expensive iodinated precursors. The reaction proceeds smoothly in polar solvents such as DMA or DMF at moderate temperatures ranging from 50°C to 90°C, significantly reducing thermal stress on sensitive substrates. The use of a nickel catalyst, specifically divalent or zero-valent nickel salts paired with bipyridine ligands, facilitates a catalytic cycle that is both robust and selective. This approach not only lowers the cost of raw materials drastically but also simplifies the operational workflow. The mild conditions ensure that complex molecules containing delicate functional groups remain intact, thereby expanding the chemical space available for drug discovery and development without the need for extensive protecting group strategies.
Mechanistic Insights into Nickel-Catalyzed Trifluoroethylation
The core of this transformation lies in the unique reactivity of the nickel catalytic system towards unactivated alkyl chlorides. Unlike palladium, which often struggles with the oxidative addition of alkyl chlorides due to high bond dissociation energy, nickel possesses a higher propensity for single-electron transfer (SET) processes. In this system, the nickel catalyst, likely reduced in situ by the metallic reductant (zinc or manganese powder), generates a low-valent nickel species capable of activating the C-Cl bond of the trifluoroethyl source. The presence of the pyridine-based ligand, particularly 4,4'-di-tert-butyl-2,2'-bipyridine (4,4'-dtbbpy), is critical for stabilizing the active nickel center and preventing catalyst deactivation via aggregation. This ligand environment modulates the electronic properties of the metal, facilitating the transmetallation or radical recombination steps necessary for C-C bond formation. The mechanistic pathway likely involves a radical intermediate, which explains the broad substrate scope and the tolerance for various functional groups that would otherwise interfere with ionic mechanisms.
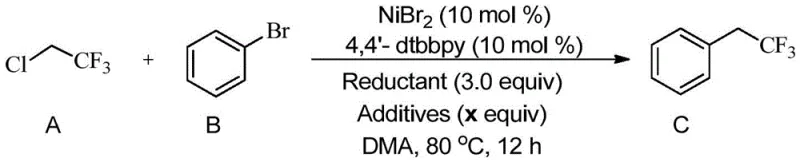
Furthermore, the inclusion of specific additives plays a pivotal role in enhancing the efficiency and selectivity of the reaction. Experimental data indicates that magnesium chloride (MgCl2) acts as a potent Lewis acid additive, potentially coordinating with the halide leaving group or the nitrogen atoms of the ligand to accelerate the catalytic turnover. The optimization of the reductant is equally important; while manganese powder shows activity, zinc powder often provides superior results in terms of both conversion rates and isolated yields. The synergy between the nickel precursor, the bulky bipyridine ligand, the metallic reductant, and the magnesium additive creates a highly tuned microenvironment within the reaction vessel. This precise tuning allows for the efficient coupling of electron-rich and electron-deficient aryl halides alike, ensuring consistent performance across a diverse library of substrates, which is essential for the reliable production of complex pharmaceutical intermediates.
How to Synthesize Trifluoroethyl Compounds Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic mixture and the handling of the gaseous reagent. The process begins by establishing an inert atmosphere, typically using argon or nitrogen, to protect the sensitive low-valent nickel species from oxidation. The catalyst system is assembled by mixing the nickel salt and ligand in the polar solvent, followed by the introduction of the 2-chloro-1,1,1-trifluoroethane gas, the concentration of which is carefully controlled to ensure optimal stoichiometry. Once the substrate and additives are introduced, the reaction is heated to the optimized temperature window. Detailed standard operating procedures regarding specific molar ratios, solvent volumes, and workup protocols are critical for reproducibility and safety. For a comprehensive understanding of the exact experimental parameters required to achieve high purity and yield, please refer to the standardized synthesis steps outlined below.
- Prepare the catalytic system by combining a nickel salt catalyst (such as NiBr2) with a pyridine-based ligand (like 4,4'-dtbbpy) in a polar solvent.
- Introduce 2-chloro-1,1,1-trifluoroethane gas into the reaction solvent to achieve the desired concentration, ensuring an inert atmosphere.
- Add the aryl or heteroaryl halide substrate, a metallic reductant (Zn or Mn), and additives (such as MgCl2), then heat to 50-90°C for 12-24 hours.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The primary driver for cost reduction is the substitution of expensive, specialized fluorinating reagents with 2-chloro-1,1,1-trifluoroethane, a commodity chemical produced on a massive industrial scale for refrigeration and other applications. This shift decouples the production cost from the volatile pricing of fine chemical fluorinating agents. Moreover, the replacement of precious metal catalysts like palladium with earth-abundant nickel significantly lowers the catalyst cost per kilogram of product. The elimination of stoichiometric copper waste also reduces the burden on waste treatment facilities, leading to substantial savings in environmental compliance and disposal costs. These factors combine to create a manufacturing process that is not only cheaper but also more predictable in terms of raw material availability.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior to legacy methods due to the drastic reduction in raw material expenses. By utilizing a cheap industrial gas as the trifluoroethyl source, manufacturers avoid the high premiums associated with iodinated or boronated fluorine reagents. Additionally, the use of non-precious nickel catalysts eliminates the need for costly metal recovery processes often required with palladium. The simplified workup procedure, which avoids complex chromatographic separations in many cases, further reduces labor and solvent consumption. Collectively, these efficiencies translate into a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for fluorinated intermediates.
- Enhanced Supply Chain Reliability: Supply chain security is paramount for continuous pharmaceutical production, and this method strengthens it by relying on widely available commodities. 2-Chloro-1,1,1-trifluoroethane and simple aryl halides are sourced from robust, multi-vendor supply chains, minimizing the risk of shortages that can plague specialized reagents. The mild reaction conditions reduce the dependency on specialized high-pressure or high-temperature equipment, allowing for production in a wider range of facilities. This flexibility ensures that manufacturing can be scaled or shifted geographically without significant capital investment, providing procurement managers with greater leverage and continuity in their supply networks.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its successful demonstration on gram scales with high yields, indicating a smooth path to multi-kilogram and ton-scale production. The reaction generates minimal heavy metal waste compared to copper-mediated methods, aligning with increasingly stringent environmental regulations. The use of common polar solvents like DMA or DMF, which are easily recycled, further enhances the green chemistry profile. For supply chain heads, this means fewer regulatory hurdles and a lower risk of production stoppages due to environmental non-compliance, ensuring a steady flow of high-quality materials to downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed trifluoroethylation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific pipeline candidates and for procurement teams assessing the long-term viability of the supply chain.
Q: What are the advantages of using 2-chloro-1,1,1-trifluoroethane over traditional trifluoromethylating agents?
A: 2-Chloro-1,1,1-trifluoroethane is a cheap, abundant industrial raw material compared to expensive and unstable reagents like 1,1,1-trifluoro-2-iodoethane. It allows for direct C-Cl bond activation under mild conditions.
Q: Does this nickel-catalyzed method tolerate sensitive functional groups?
A: Yes, the method exhibits excellent functional group compatibility, tolerating esters, ketones, ethers, and heterocycles that are often incompatible with Grignard-based traditional methods.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The reaction uses earth-abundant nickel catalysts, operates at mild temperatures (50-90°C), and utilizes simple workup procedures, making it highly scalable and cost-effective for manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoroethyl Compounds Supplier
The technological advancements detailed in patent CN110668913A represent a significant opportunity for the chemical industry to produce high-value fluorinated building blocks more efficiently. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent discoveries into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical clients. We understand the critical nature of fluorine chemistry and have the expertise to manage the specific safety and handling requirements of gaseous reagents and nickel catalysts, ensuring a safe and compliant manufacturing environment.
We invite potential partners to engage with our technical procurement team to discuss how this novel synthesis route can be adapted to your specific project needs. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this nickel-catalyzed method for your specific molecule. We encourage you to contact us to request specific COA data from our pilot runs and detailed route feasibility assessments. Let us collaborate to secure your supply chain and optimize your production costs for next-generation trifluoroethyl intermediates.
