Revolutionizing Chromone Derivative Production with Efficient Copper-Catalyzed Cyclization for Commercial Scale
Revolutionizing Chromone Derivative Production with Efficient Copper-Catalyzed Cyclization for Commercial Scale
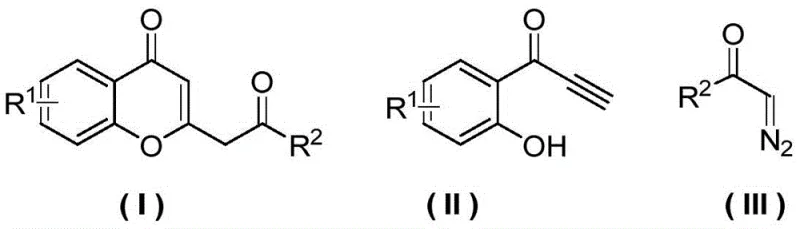
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in the Chinese patent CN113214205B, which discloses a novel preparation method for chromone derivatives. This technology leverages a sophisticated copper-catalyzed reaction between o-hydroxybenzoyl acetylene compounds and diazo compounds to construct the benzopyran-4-one core structure with exceptional precision. For R&D directors and procurement managers seeking a reliable chromone derivative supplier, this patent represents a pivotal shift away from traditional, labor-intensive methodologies towards a streamlined, high-yield process that promises substantial operational advantages.
The core innovation lies in the strategic selection of catalyst and ligand systems that facilitate a smooth carbene insertion and subsequent cyclization. Unlike older methods that often suffer from poor regioselectivity or require hazardous reagents, this approach operates under mild thermal conditions ranging from 25°C to 80°C. The ability to achieve yields exceeding 90% under optimized conditions demonstrates the robustness of this chemistry. By integrating this advanced synthetic strategy, manufacturers can significantly enhance the purity profile of their output while minimizing waste generation, aligning perfectly with modern green chemistry principles and the rigorous quality standards demanded by the global pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chromone scaffolds has relied heavily on classical condensation reactions such as the Pechmann condensation or the Baker-Venkataraman rearrangement. While these methods are well-documented, they frequently present significant bottlenecks for large-scale pharmaceutical intermediate manufacturing. Traditional routes often necessitate the use of strong acids or bases, leading to severe corrosion issues in reactor vessels and complicating the downstream neutralization and waste treatment processes. Furthermore, these conventional pathways typically involve multiple synthetic steps, including protection and deprotection sequences, which inevitably erode overall atom economy and drive up production costs due to increased solvent consumption and extended processing times.
Another critical drawback of legacy technologies is the difficulty in controlling impurity profiles, particularly when dealing with sensitive functional groups on the aromatic ring. Harsh reaction conditions can lead to polymerization, decomposition, or the formation of regio-isomers that are challenging to separate, thereby compromising the final product's purity. For supply chain heads, these inefficiencies translate into longer lead times and higher risks of batch failure. The reliance on stoichiometric amounts of reagents rather than catalytic systems further exacerbates the environmental footprint, making compliance with increasingly stringent environmental regulations a costly endeavor for producers relying on outdated synthetic protocols.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113214205B introduces a transformative catalytic cycle that bypasses these historical limitations. By utilizing a copper-catalyzed reaction between an o-hydroxybenzoyl acetylene substrate and a diazo compound, the process achieves the construction of the chromone ring in a single, concerted operation. This telescoped approach eliminates the need for intermediate isolation and reduces the total number of unit operations required. The reaction proceeds through a metal-carbene intermediate, which inserts efficiently into the alkyne bond, followed by an intramolecular nucleophilic attack by the phenolic hydroxyl group. This mechanism ensures high regioselectivity and minimizes the formation of side products, resulting in a much cleaner crude reaction mixture.
The operational simplicity of this new route is a major advantage for commercial adoption. It functions effectively in common organic solvents like dichloromethane or DMF and tolerates a wide range of substituents on both the aromatic ring and the diazo component. This versatility allows for the rapid generation of diverse chromone libraries, which is invaluable for drug discovery programs. Moreover, the mild reaction temperatures (25-80°C) reduce energy consumption compared to high-temperature reflux conditions often seen in traditional methods. For a cost reduction in pharmaceutical intermediate manufacturing, this transition from multi-step stoichiometric processes to a catalytic, one-pot synthesis offers a compelling value proposition by lowering raw material costs and improving throughput capacity.
Mechanistic Insights into Copper-Catalyzed Carbene Insertion and Cyclization
To fully appreciate the technical superiority of this method, one must delve into the mechanistic intricacies of the copper-catalyzed transformation. The reaction initiates with the activation of the diazo compound by the copper catalyst, specifically cuprous iodide (CuI), to generate a reactive copper-carbene species (Intermediate A). This step is critical, as the stability and electrophilicity of the carbene determine the success of the subsequent insertion. The patent data indicates that monovalent copper sources are vastly superior to divalent salts, suggesting that the redox properties of Cu(I) are essential for effective carbene formation without premature decomposition of the diazo precursor.
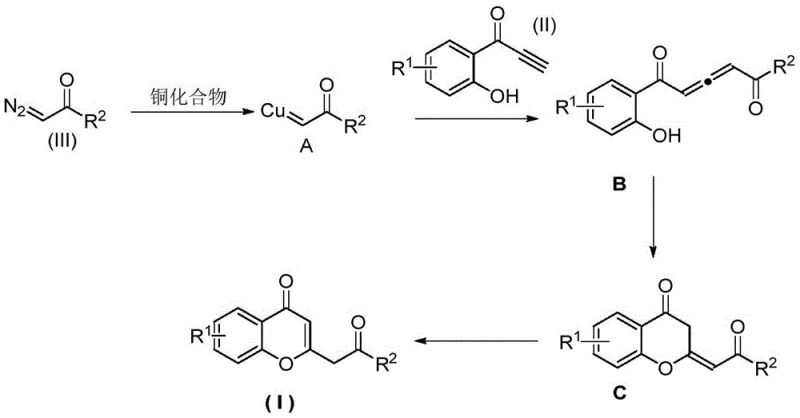
Following the formation of the copper-carbene complex, the mechanism proceeds via an insertion reaction into the carbon-carbon triple bond of the o-hydroxybenzoyl acetylene compound. This generates a vinyl-copper intermediate (Intermediate B), which possesses a unique electronic configuration that facilitates the next step. The proximal phenolic hydroxyl group then acts as an internal nucleophile, attacking the electrophilic center to close the pyran ring, forming Intermediate C. Finally, a protodemetalation or isomerization step releases the copper catalyst back into the cycle and yields the final chromone derivative (Product I). This elegant cascade ensures that the catalyst is regenerated efficiently, allowing for low catalyst loadings (molar ratios as low as 1:0.05 relative to substrate) while maintaining high turnover numbers.
Impurity control is inherently built into this mechanism due to the specificity of the carbene insertion. Unlike radical processes that can lead to indiscriminate bonding, the metal-carbene pathway is highly directed. The choice of ligand plays a pivotal role here; the patent highlights that simple amines like triethylamine provide the optimal steric and electronic environment for the copper center. Strongly chelating ligands like 1,10-phenanthroline were found to inhibit the reaction, likely by over-stabilizing the copper complex and preventing the necessary coordination of the diazo compound. Understanding these subtle interactions allows process chemists to fine-tune the reaction for maximum purity, ensuring that the final high-purity chromone derivatives meet the strict specifications required for API synthesis.
How to Synthesize Chromone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to replicate the high yields reported in the patent literature. The process begins with the precise weighing of the o-hydroxybenzoyl acetylene substrate and the diazo compound, typically in a molar ratio ranging from 1:1 to 1:3 to drive the reaction to completion. These substrates are dissolved in an anhydrous organic solvent, with dichloromethane being the preferred medium due to its ability to solubilize both reactants and the catalyst effectively. The addition of the copper catalyst, preferably cuprous iodide, and the amine ligand must be conducted under an inert atmosphere to prevent oxidation of the sensitive copper species and the diazo compound.
- Prepare the reaction mixture by combining o-hydroxybenzoyl acetylene compound, diazo compound, cuprous iodide catalyst, and triethylamine ligand in dichloromethane solvent.
- Heat the reaction system to a temperature range of 25-80°C and maintain stirring for 0.5 to 8 hours to ensure complete conversion.
- Perform post-processing via extraction with ethyl acetate and brine, followed by concentration and silica gel column chromatography to isolate the pure chromone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing readily available o-hydroxybenzoyl acetylenes and diazo compounds, manufacturers can avoid the bottlenecks associated with sourcing exotic or highly regulated reagents often required in traditional chromone synthesis. Furthermore, the use of base copper salts like CuI, which are commodity chemicals, eliminates the dependency on expensive noble metal catalysts such as palladium or rhodium, leading to substantial cost savings in catalyst procurement and recovery.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of multiple synthetic steps and the reduction in solvent usage. Traditional multi-step routes incur cumulative costs at each stage, including labor, energy, and waste disposal. By consolidating the synthesis into a single catalytic step, the overall manufacturing cost is significantly lowered. Additionally, the high yields (consistently above 85% and reaching over 90% in optimized examples) mean that less starting material is wasted, directly improving the cost of goods sold (COGS). The avoidance of expensive purification techniques like preparative HPLC, relying instead on standard silica gel chromatography or crystallization, further enhances the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical manufacturers, and this robust synthetic route offers greater resilience. The reaction conditions are mild and tolerant to minor variations in temperature and mixing, reducing the risk of batch-to-batch variability that can disrupt supply schedules. The catalyst system is stable and does not require complex handling procedures, simplifying logistics and storage requirements. Moreover, the broad substrate scope means that if a specific starting material faces supply constraints, the chemistry is flexible enough to accommodate alternative analogs without requiring a complete process redevelopment, thereby securing the supply chain against market fluctuations.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the absence of hazardous high-pressure or high-temperature conditions. The exothermic nature of carbene formation is manageable under the described conditions, ensuring safe operation in large reactors. From an environmental perspective, the process generates less waste compared to stoichiometric methods, aligning with corporate sustainability goals. The use of recyclable solvents and the potential for catalyst recovery contribute to a lower E-factor (environmental factor), making it easier for facilities to maintain compliance with environmental regulations and reducing the financial burden of waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chromone synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in patent CN113214205B, offering clarity on catalyst selection, reaction scope, and process optimization. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the optimal catalyst system for this chromone synthesis?
A: According to patent CN113214205B, cuprous iodide (CuI) combined with triethylamine provides the highest catalytic efficiency and yield, significantly outperforming divalent copper sources.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction utilizes simple conditions (25-80°C) and common solvents like dichloromethane, making it highly suitable for commercial scale-up without requiring specialized high-pressure equipment.
Q: How does this method compare to traditional chromone synthesis?
A: This novel approach offers a direct one-pot cyclization via carbene insertion, avoiding the multi-step sequences and harsh acidic conditions often associated with conventional Pechmann or Baker-Venkataraman rearrangements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis described in patent CN113214205B for the production of high-value chromone derivatives. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust, commercial-grade manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies gained in the lab are preserved and amplified at an industrial scale. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our chromone derivative portfolio and to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain and accelerate your time to market with our reliable and efficient manufacturing solutions.
