Revolutionizing Tadalafil Production: A Low-Cost L-Tryptophan Route for Global Supply Chains
Revolutionizing Tadalafil Production: A Low-Cost L-Tryptophan Route for Global Supply Chains
The pharmaceutical landscape for Erectile Dysfunction (ED) treatments continues to evolve, with Tadalafil remaining a cornerstone blockbuster drug due to its extended duration of action and favorable pharmacokinetic profile. However, the economic viability of generic production has long been hindered by the exorbitant cost of chiral starting materials. Patent CN103772384A introduces a paradigm-shifting methodology that fundamentally alters the cost structure of Tadalafil manufacturing. By ingeniously utilizing L-tryptophan methyl ester hydrochloride—a naturally abundant and significantly cheaper precursor—instead of the traditional D-isomer, this technology enables the synthesis of high-purity Tadalafil through a novel chiral inversion mechanism. This approach not only addresses the critical pain point of raw material expenditure but also simplifies the purification workflow, making it an ideal candidate for reliable tadalafil intermediate supplier partnerships aiming for large-scale commercialization.
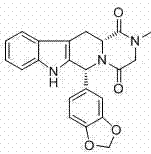
For R&D directors evaluating process feasibility, the structural integrity and stereochemical fidelity of the final product are paramount. The disclosed method ensures that the final active pharmaceutical ingredient meets stringent regulatory specifications, with optical rotation values aligning perfectly with pharmacopoeial standards. This technical breakthrough represents a significant leap forward in process chemistry, transforming a cost-prohibitive synthesis into a commercially robust operation suitable for the demands of the global generic drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tadalafil has relied heavily on D-tryptophan methyl ester hydrochloride as the foundational chiral building block. As detailed in prior art such as US5859006 and US7550479, this dependency creates a severe economic bottleneck because the D-isomer is an unnatural amino acid derivative that commands a premium price, often exceeding 1200 RMB per kilogram. In many existing manufacturing protocols, this single raw material constitutes more than 70% of the total synthetic material cost, rendering the overall process margin-sensitive and vulnerable to supply chain fluctuations. Furthermore, traditional routes frequently suffer from diastereomeric impurities that necessitate resource-intensive purification techniques, such as column chromatography or multiple recrystallization cycles, which drastically reduce overall throughput and increase solvent waste. The reliance on non-natural starting materials also introduces supply chain fragility, as the production capacity for D-tryptophan derivatives is inherently more limited compared to their natural L-counterparts.
The Novel Approach
The innovative strategy outlined in CN103772384A circumvents these economic and technical barriers by initiating the synthesis with L-tryptophan methyl ester hydrochloride, a commodity chemical available at less than one-fifth the cost of its D-enantiomer. This route capitalizes on the reversible nature of the Pictet-Spengler reaction and the specific chemical property of chiral inversion at the ester ortho-position under alkaline conditions. By strategically employing a strong base to invert the stereochemistry at a critical intermediate stage, the process effectively converts the inexpensive L-configured precursor into the required D-configuration necessary for the biologically active cis-isomer of Tadalafil. This clever manipulation of stereochemistry allows manufacturers to bypass the expensive D-tryptophan supply chain entirely while simultaneously improving the isolation efficiency of the key intermediate compounds through simple filtration rather than complex chromatography.
Consequently, this novel approach delivers a dual advantage: it dramatically lowers the Cost of Goods Sold (COGS) through raw material substitution and enhances operational efficiency by streamlining the purification of intermediates like Compound 3a. The ability to drive the equilibrium towards the less soluble, desired cis-product through thermodynamic control ensures high yields without the need for excessive reagents or harsh separation conditions, marking a substantial improvement over legacy manufacturing technologies.
Mechanistic Insights into Base-Catalyzed Chiral Inversion
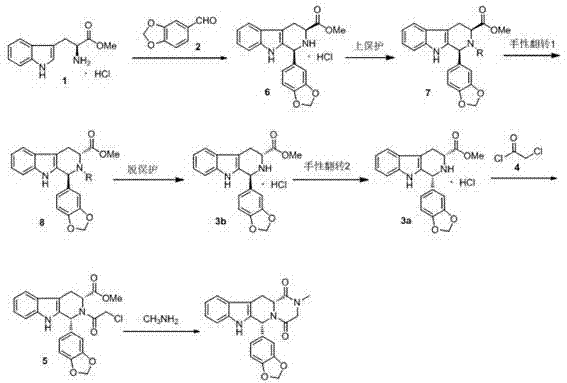
The core scientific innovation of this process lies in the precise execution of base-catalyzed chiral inversion, a sophisticated transformation that allows for the stereochemical correction of the molecule mid-synthesis. Initially, L-tryptophan methyl ester hydrochloride undergoes a Pictet-Spengler condensation with piperonal to form Compound 6, preserving the L-configuration. Following N-protection to yield Compound 7, the mixture is subjected to strong basic reagents such as potassium tert-butoxide or lithium diisopropylamide at elevated temperatures ranging from 50°C to 150°C. Under these specific alkaline conditions, the proton at the alpha-carbon adjacent to the ester group becomes acidic enough to be abstracted, forming a planar enolate intermediate. Upon reprotonation, the stereochemistry can flip, allowing the thermodynamically more stable or kinetically accessible D-configuration to emerge as Compound 8. This step is critical because it effectively 'repairs' the chirality of the cheap starting material to match the stereochemical requirements of the final drug substance.
Following the inversion, the process leverages the reversibility of the Pictet-Spengler reaction to further enrich the desired diastereomer. When Compound 3b (the deprotected amine) is heated with a catalytic amount of piperonal, the system equilibrates, favoring the formation of the cis-isomer Compound 3a due to its significantly lower solubility in the reaction medium. This phenomenon allows Compound 3a to precipitate out of the solution as a high-purity solid, effectively pulling the equilibrium towards the product and leaving impurities in the mother liquor. This mechanism of 'crystallization-induced diastereomer transformation' ensures that the final intermediate entering the cyclization phase possesses exceptional stereochemical purity, which is essential for meeting the rigorous impurity profiles demanded by regulatory agencies for generic API approval.
How to Synthesize Tadalafil Efficiently
The synthesis protocol described in the patent offers a robust, scalable pathway that integrates standard unit operations with specialized stereochemical control steps. The process begins with the condensation of L-tryptophan methyl ester and piperonal, followed by protection, inversion, deprotection, and final ring closure. Each step has been optimized to maximize yield and minimize waste, utilizing common organic solvents like toluene, dichloromethane, and methanol. The detailed procedural parameters, including specific molar ratios, temperature ramps, and workup procedures, are critical for reproducing the high purity (>99.5%) reported in the examples. For process chemists looking to implement this route, understanding the nuances of the inversion step and the crystallization dynamics of Compound 3a is key to success.
- React L-tryptophan methyl ester hydrochloride with piperonal in organic solvent at 70-100°C to form Compound 6, followed by Boc protection to yield Compound 7.
- Perform chiral inversion on Compound 7 using a strong base (e.g., potassium tert-butoxide) at 50-150°C to obtain the inverted stereoisomer Compound 8.
- Deprotect Compound 8 to form 3b, then epimerize with piperonal to isolate the cis-intermediate 3a, followed by chloroacetylation and methylamine cyclization to finalize Tadalafil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this L-tryptophan-based route presents a compelling value proposition centered on cost stability and supply security. The most immediate impact is the drastic reduction in raw material expenditure; by substituting a high-cost specialty chemical with a commoditized natural amino acid derivative, manufacturers can insulate their production costs from the volatility associated with synthetic chiral pools. This shift does not merely offer marginal savings but fundamentally restructures the bill of materials, potentially lowering the overall manufacturing cost by a significant factor given that the tryptophan derivative historically dominates the cost basis. Furthermore, the simplified purification workflow, which relies on filtration and washing rather than column chromatography, reduces solvent consumption and processing time, leading to additional operational cost reductions in utility and waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive D-tryptophan derivatives removes the single largest cost driver in traditional Tadalafil synthesis. Since L-tryptophan methyl ester hydrochloride is priced at a fraction of the D-isomer, the direct material cost savings are substantial and immediate. Additionally, the high yields reported in the patent examples, such as 97% for Compound 6 and 95% for Compound 3a, indicate a highly efficient atom economy that minimizes waste and maximizes output per batch. The avoidance of chromatographic purification further reduces the cost burden associated with silica gel, solvents, and labor, contributing to a leaner and more profitable manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit immensely from the use of L-tryptophan, as it is a naturally occurring amino acid produced on a massive global scale for food and feed applications, ensuring a stable and abundant supply. Unlike D-tryptophan, which requires specialized synthetic resolution or fermentation processes with limited capacity, L-tryptophan is readily available from multiple vendors worldwide, reducing the risk of supply disruptions. This abundance translates to better negotiating power for procurement teams and shorter lead times for raw material delivery, ensuring continuous production schedules and reliable fulfillment of customer orders for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed for industrial scalability, utilizing robust reaction conditions and simple isolation techniques that translate easily from pilot plant to commercial scale. The reduction in solvent usage, particularly the elimination of large volumes required for column chromatography, aligns with green chemistry principles and reduces the environmental footprint of the manufacturing site. This compliance with environmental standards simplifies regulatory permitting and waste management, while the high purity of the intermediates reduces the need for reprocessing, thereby enhancing overall throughput and capacity utilization for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method compares to established industry standards. Understanding these details is crucial for stakeholders evaluating the feasibility of technology transfer or licensing agreements.
Q: Why is the L-Tryptophan route superior to the traditional D-Tryptophan method?
A: The primary advantage is cost efficiency. L-Tryptophan methyl ester hydrochloride costs less than one-fifth of the D-isomer, which traditionally accounts for over 70% of total material costs. Additionally, this route avoids expensive column chromatography purification steps.
Q: What is the achieved purity of Tadalafil using this novel synthesis method?
A: The patent data indicates that the final product achieves a purity of greater than 99.5% as measured by HPLC, with optical rotation values strictly complying with European Pharmacopoeia standards (+78.0° to +84°).
Q: How is stereochemical control maintained when starting with the wrong enantiomer?
A: The process utilizes a base-catalyzed chiral inversion at the ester ortho-position. By leveraging the reversibility of the Pictet-Spengler reaction and the lower solubility of the desired cis-isomer, the system thermodynamically drives the formation of the correct stereochemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tadalafil Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this L-tryptophan-based synthesis route for the global Tadalafil market. As a premier CDMO partner, we possess the technical expertise and infrastructure to rapidly adapt and optimize this patented methodology for your specific production needs. Our facilities are equipped to handle complex chiral syntheses with precision, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of Tadalafil intermediate or API meets the highest international standards for safety and efficacy, thereby mitigating the risks associated with technology transfer and scale-up.
We invite you to collaborate with us to leverage this cost-effective technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to this L-tryptophan route can improve your margins. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for securing your long-term supply of high-quality Tadalafil intermediates.
