Advanced Synthesis of Azepino[4,5-b]indole Intermediates for Commercial Scale-Up
Advanced Synthesis of Azepino[4,5-b]indole Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products like ibogaine and its analogs. Patent CN113121543B discloses a groundbreaking preparation method for azepino[4,5-b]indole compounds that addresses longstanding challenges in synthetic efficiency and operational simplicity. This technology leverages readily available tryptamine derivatives as starting materials, employing a strategic sequence of protection, allylation, olefin metathesis, and Lewis acid-catalyzed cyclization. For R&D directors and procurement specialists, this represents a significant opportunity to access high-purity azepino[4,5-b]indole intermediates through a route that eliminates the need for extreme temperatures or pressures, thereby enhancing process safety and reproducibility in a GMP environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the azepino[4,5-b]indole skeleton has relied on a disparate array of complex transformations, including electrophilic substitutions, palladium-catalyzed reductive Heck reactions, and photo-induced free radical processes. These traditional pathways often suffer from significant drawbacks, such as the requirement for expensive transition metal catalysts, stringent anhydrous conditions, and multi-step sequences that result in cumulative yield losses. Furthermore, many existing methods utilize specialized precursors that are not commercially available on a metric ton scale, creating bottlenecks in the supply chain for potential API manufacturers. The reliance on harsh reaction conditions also complicates waste management and increases the overall cost of goods sold, making these routes less attractive for large-scale commercial production of neuroactive pharmaceutical intermediates.
The Novel Approach
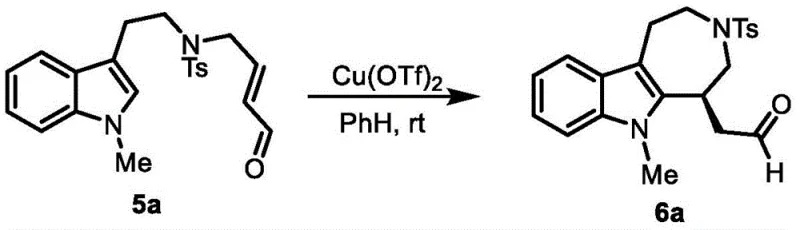
In stark contrast, the methodology outlined in the patent introduces a streamlined, linear synthesis that prioritizes atom economy and operational ease. By initiating the sequence with commodity-grade tryptamine, the process immediately lowers the barrier to entry regarding raw material costs. The core innovation lies in the combination of olefin cross-metathesis to install the necessary carbon framework followed by a mild Lewis acid-promoted intramolecular Michael addition to close the seven-membered ring. This approach avoids the use of stoichiometric oxidants or reductants typically associated with ring-expansion strategies. As illustrated in the general reaction scheme below, the transformation proceeds through well-defined intermediates that can be easily purified, ensuring a consistent quality profile for the final active pharmaceutical ingredient precursor.
![General reaction scheme for the synthesis of azepino[4,5-b]indole compounds from tryptamine derivatives](/insights/img/azepino-indole-synthesis-pharma-supplier-20260305012129-01.webp)
Mechanistic Insights into Cu(OTf)2-Catalyzed Cyclization
The cornerstone of this synthetic strategy is the final ring-closing step, which employs copper(II) triflate as a Lewis acid catalyst to facilitate a formal intramolecular Michael addition. Mechanistically, the copper center coordinates with the carbonyl oxygen of the pendant aldehyde chain, increasing the electrophilicity of the beta-carbon. This activation allows the electron-rich C3 position of the indole nucleus to attack the activated olefin, forming the new carbon-carbon bond that establishes the azepine ring system. Unlike traditional Friedel-Crafts alkylations that might require strong Bronsted acids and elevated temperatures, this copper-catalyzed variant operates efficiently at room temperature. This mildness is crucial for preserving sensitive functional groups elsewhere on the molecule, such as the tosyl-protected amine, preventing decomposition or side reactions that could compromise the purity of the final isolate.
Furthermore, the regioselectivity of this cyclization is inherently controlled by the geometry of the tethered chain and the electronic bias of the indole ring. The use of the Hoveyda-Grubbs II catalyst in the preceding metathesis step ensures the formation of the specific E-alkene geometry required for the subsequent cyclization to proceed with high stereochemical fidelity. The resulting product features a chiral center at the ring junction, and while the patent describes the formation of racemic mixtures in the examples, the mild conditions leave open the possibility for future asymmetric variants using chiral ligands. The structural integrity of the final product, as confirmed by NMR and mass spectrometry data in the patent examples, demonstrates the robustness of this cascade, yielding the target azepino[4,5-b]indole scaffold with high structural definition suitable for downstream derivatization.
How to Synthesize Azepino[4,5-b]indole Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing standard unit operations that are familiar to process chemists. The sequence begins with the selective protection of the primary amine on the tryptamine side chain using tosyl chloride, followed by N-alkylation of the indole nitrogen. Subsequent N-allylation installs the handle for the cross-metathesis reaction with crotonaldehyde, setting the stage for the final cyclization. Each step utilizes common organic solvents such as dichloromethane, DMF, and benzene, and relies on standard workup procedures like aqueous quenching and column chromatography. For detailed standardized operating procedures and specific molar ratios optimized for maximum yield, please refer to the technical guide below.
- Protect the side-chain amine of tryptamine with TsCl and protect the indole nitrogen with an alkyl group.
- Perform N-allylation on the side chain followed by cross-metathesis with crotonaldehyde using Hoveyda-Grubbs II catalyst.
- Execute the final intramolecular Michael addition cyclization using Cu(OTf)2 catalyst at room temperature to form the seven-membered ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of sourcing complex heterocyclic intermediates. By shifting away from precious metal catalysts like palladium in the key bond-forming steps and utilizing more abundant copper salts for the cyclization, the process significantly reduces the cost burden associated with catalyst recovery and heavy metal clearance. This transition not only lowers the direct material costs but also simplifies the regulatory filing process by minimizing the risk of residual metal contamination in the final drug substance. Additionally, the reliance on room temperature reactions for the critical cyclization step drastically reduces energy consumption compared to processes requiring prolonged heating or cryogenic cooling, contributing to a lower carbon footprint and reduced utility costs for the manufacturing facility.
- Cost Reduction in Manufacturing: The utilization of tryptamine as a starting material leverages a globally established supply chain for this commodity chemical, effectively decoupling production from volatile markets for exotic precursors. The elimination of expensive palladium catalysts in favor of copper triflate represents a substantial decrease in catalyst expenditure per kilogram of product. Moreover, the high yields reported in the patent examples suggest that fewer batches are required to meet production targets, optimizing reactor utilization time and labor costs. The simplified purification protocols, primarily relying on standard chromatography or recrystallization rather than preparative HPLC, further drive down the operational expenses associated with downstream processing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for ambient temperatures and standard atmospheric pressure, enhances the reliability of the supply chain by reducing the risk of batch failures due to equipment malfunction. Since the reagents involved, such as allyl bromide and crotonaldehyde, are widely available bulk chemicals, the risk of supply disruption is minimized compared to routes dependent on custom-synthesized building blocks. This stability allows for more accurate forecasting and inventory management, ensuring that pharmaceutical partners can maintain continuous production schedules without unexpected delays caused by raw material shortages or complex logistics.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the exothermic profiles of the reactions are manageable and do not require specialized high-pressure reactors. The use of common organic solvents facilitates solvent recovery and recycling programs, aligning with modern green chemistry principles and environmental regulations. By avoiding the generation of hazardous byproducts associated with radical reactions or strong oxidants, the waste stream is easier to treat and dispose of, reducing the environmental compliance burden. This makes the technology highly attractive for contract development and manufacturing organizations (CDMOs) looking to offer sustainable and scalable solutions for neuroactive drug development.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route, derived from the specific experimental data and claims within the patent documentation. Understanding these nuances is critical for process engineers evaluating the feasibility of technology transfer. The answers provided reflect the optimal conditions identified during the patent prosecution to ensure high purity and yield.
Q: What are the critical reaction conditions for the final cyclization step?
A: The final cyclization utilizes Cu(OTf)2 as a Lewis acid catalyst in solvents like benzene or toluene at room temperature for 20-60 hours, avoiding harsh thermal conditions.
Q: Can this method accommodate different substituents on the indole ring?
A: Yes, the protocol demonstrates high tolerance for various R groups including hydrogen, alkyl, methoxyl, and halogens, allowing for diverse derivative synthesis.
Q: Why is the Hoveyda-Grubbs II catalyst selected for the metathesis step?
A: Hoveyda-Grubbs II offers superior stability and activity for cross-metathesis reactions involving electron-deficient olefins like crotonaldehyde under mild heating.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azepino[4,5-b]indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes for complex heterocyclic scaffolds like the azepino[4,5-b]indole core. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with international quality standards. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the exacting requirements of global pharmaceutical clients. Our commitment to technical excellence allows us to adapt patented methodologies to fit specific client needs while maintaining cost-effectiveness and supply security.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this route for your specific project. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your development timeline, ensuring that your journey from discovery to market is supported by a partner dedicated to innovation and reliability.
