Advanced Manufacturing of High-Purity 3-Bromofluorenone for Next-Gen OLED Applications
Advanced Manufacturing of High-Purity 3-Bromofluorenone for Next-Gen OLED Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands intermediates of exceptional purity and structural precision, driving the need for innovative synthetic methodologies. Patent CN107056595B introduces a groundbreaking preparation method for 3-bromofluorenone, a critical building block for high-performance electroluminescent materials. This technology addresses long-standing challenges in the fluorene compound sector by replacing hazardous and inefficient legacy processes with a streamlined, three-step protocol involving oximation, regioselective bromination, and hydrolysis. By leveraging an oxime protection strategy, this method not only enhances reaction selectivity but also ensures the final product meets the rigorous specifications required for commercial-scale electronic chemical manufacturing. For R&D directors and procurement specialists alike, this patent represents a pivotal shift towards more sustainable and cost-effective production of high-purity OLED material.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated fluorenones has been plagued by complex multi-step sequences that compromise both economic viability and operational safety. One prominent legacy route, documented in literature such as Tetrahedron Letters, utilizes phenanthrenequinone as a starting material, necessitating a bromination followed by a potassium hydroxide-induced rearrangement and subsequent oxidation. As illustrated in the reaction scheme below, this pathway is fundamentally flawed due to the sluggish kinetics of the rearrangement step, which acts as a severe bottleneck, resulting in low overall yields and significant difficulties in isolating high-purity products suitable for optoelectronic applications.
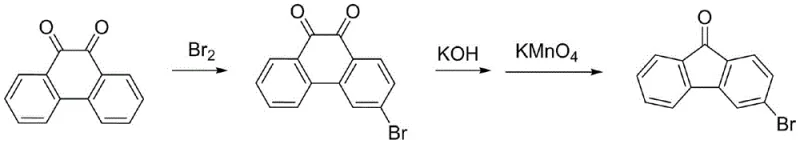
Furthermore, alternative historical approaches, such as those described in the Journal of the American Chemical Society, rely on the diazotization of amino-bromoacetophenones. These methods introduce substantial safety liabilities due to the formation of unstable diazonium salts, alongside the prohibitive cost and scarcity of specialized amine precursors. The purification of products from these routes is notoriously difficult, often failing to remove trace impurities that can quench luminescence in final OLED devices. Consequently, the industry has urgently required a reliable agrochemical intermediate supplier and electronic chemical partner capable of delivering a safer, more direct synthetic alternative.
The Novel Approach
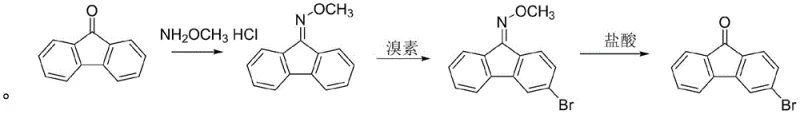
The methodology disclosed in CN107056595B offers a transformative solution by employing a strategic oxime protection group to direct electrophilic substitution. Instead of struggling with unactivated ketones or hazardous diazonium intermediates, the process begins with the conversion of fluorenone into fluorenone methoxy oxime. This intermediate serves as a highly activated substrate for bromination, allowing for precise regiocontrol without the need for extreme conditions. The subsequent hydrolysis step cleanly regenerates the carbonyl functionality, delivering the target 3-bromofluorenone with remarkable efficiency. This novel approach eliminates the need for transition metal catalysts often found in Suzuki coupling routes, thereby simplifying the downstream purification process and drastically reducing the environmental footprint associated with heavy metal waste disposal.
Mechanistic Insights into Oxime-Directed Electrophilic Bromination
The core innovation of this technology lies in the electronic modulation of the fluorene ring system through oxime formation. In the initial step, the reaction of fluorenone with methoxylamine hydrochloride generates an O-methyl oxime, which acts as a powerful electron-donating group via resonance. This activation significantly increases the electron density at the ortho and para positions relative to the oxime nitrogen, facilitating a rapid and selective electrophilic aromatic substitution with molecular bromine. The presence of a Lewis acid catalyst, specifically aluminum trichloride, further enhances the electrophilicity of the bromine species, ensuring that the reaction proceeds smoothly at moderate temperatures ranging from 30°C to 60°C. This mechanistic advantage prevents the formation of poly-brominated byproducts, a common issue in direct ketone bromination, thereby securing a cleaner reaction profile.
From an impurity control perspective, the hydrolysis mechanism is equally critical for achieving the reported 99.9% GC purity. The acidic hydrolysis of the brominated oxime intermediate is conducted under controlled thermal conditions (50-100°C) using dilute hydrochloric acid. This step effectively cleaves the C=N bond to restore the ketone while simultaneously protonating and solubilizing any residual amine-based impurities or catalyst complexes into the aqueous phase. The result is a crude product with exceptionally low levels of dibromo contaminants (less than 100ppm), which minimizes the burden on final recrystallization steps. For manufacturers focused on cost reduction in electronic chemical manufacturing, this inherent selectivity translates directly into higher throughput and reduced solvent consumption during purification.
How to Synthesize 3-Bromofluorenone Efficiently
The implementation of this synthesis route requires strict adherence to the optimized reaction parameters defined in the patent to ensure reproducibility and safety. The process is divided into three distinct operational phases: the formation of the oxime ether, the catalytic bromination, and the final acidic hydrolysis. Each stage utilizes common industrial solvents such as methanol, dichloroethane, and dilute hydrochloric acid, making the technology highly accessible for existing chemical infrastructure. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to adopt this superior manufacturing protocol.
- Perform an oximation reaction by reacting fluorenone with methoxylamine hydrochloride in an alcoholic solvent at 40-100°C to form fluorenone methoxy oxime.
- Conduct a regioselective bromination using bromine and an aluminum trichloride catalyst in a chlorinated solvent at 30-60°C to obtain the brominated oxime intermediate.
- Execute a hydrolysis reaction by treating the brominated intermediate with 20-35% hydrochloric acid at 50-100°C to regenerate the ketone and yield 3-bromofluorenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond mere technical performance. By shifting away from complex multi-step sequences involving expensive precursors like nitro-benzoates or unstable diazonium salts, manufacturers can achieve significant cost savings through raw material optimization. The use of commodity chemicals such as fluorenone, methoxylamine hydrochloride, and bromine ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized intermediates that are prone to market volatility. Furthermore, the elimination of heavy metal catalysts removes the need for costly scavenging processes and extensive wastewater treatment, contributing to a leaner and more environmentally compliant operation.
- Cost Reduction in Manufacturing: The streamlined three-step process inherently reduces operational expenditures by minimizing unit operations and energy consumption. Unlike legacy routes that require cryogenic conditions or high-pressure reactors, this method operates at atmospheric pressure and moderate temperatures, leading to substantial utility savings. Additionally, the high selectivity of the oxime-directed bromination reduces the generation of hard-to-separate isomers, which lowers the cost of goods sold by maximizing the yield of the desired 3-bromofluorenone isomer without extensive chromatographic purification.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals ensures that production schedules are not disrupted by the lead times associated with custom-synthesized starting materials. The robustness of the reaction conditions allows for flexible batch sizing, enabling suppliers to respond rapidly to fluctuating demand from the OLED and pharmaceutical sectors. This reliability is crucial for maintaining continuous production lines in downstream applications where material consistency is paramount for device performance and longevity.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and reagents that are easily managed in large-scale reactor systems. The absence of toxic heavy metals and the use of aqueous acid for hydrolysis simplify waste stream management, aligning with increasingly stringent global environmental regulations. This green chemistry profile not only reduces disposal costs but also enhances the corporate sustainability metrics of the manufacturing entity, a key factor for modern supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-bromofluorenone synthesized via this advanced method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What is the purity level achievable with this new synthesis method?
A: The patented process consistently achieves a GC purity of 99.9%, with critical dibromo impurities controlled to less than 100ppm, meeting stringent requirements for OLED applications.
Q: How does this method improve safety compared to traditional diazotization routes?
A: Unlike conventional methods that rely on hazardous diazotization reactions requiring unstable diazonium salts, this novel approach utilizes mild oximation and hydrolysis steps, significantly reducing operational risks and thermal hazards.
Q: What is the overall yield efficiency of this production route?
A: The optimized three-step sequence delivers a total yield of approximately 70%, which represents a substantial improvement over older multi-step pathways that often suffer from low yields during rearrangement or cyclization stages.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromofluorenone Supplier
As the demand for high-performance organic electronic materials continues to surge, partnering with a technically proficient manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in complex organic synthesis to deliver 3-bromofluorenone of unmatched quality. Our facilities are equipped to handle diverse synthetic pathways, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for OLED and specialty chemical applications, guaranteeing consistency and reliability for our global partners.
We invite R&D directors and procurement leaders to collaborate with us to optimize their material sourcing strategies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest quality intermediates available in the market.
