Scalable Synthesis of Sulfur-Functionalized Calix[4]arenes for Advanced Molecular Recognition
Scalable Synthesis of Sulfur-Functionalized Calix[4]arenes for Advanced Molecular Recognition
The landscape of supramolecular chemistry continues to evolve with the development of specialized macrocyclic compounds capable of precise molecular recognition. A significant advancement in this field is detailed in patent CN101555219A, which discloses a robust method for preparing 5,11,17,23-tetramercaptomethyl-25,26,27,28-tetrahydroxy-calix[4]arene. This specific derivative represents a critical evolution from traditional calixarenes by incorporating sulfur functionalities alongside the inherent hydroxyl groups. The strategic placement of mercapto groups at the upper rim of the calixarene cavity dramatically alters the electronic properties and coordination chemistry of the molecule. For research directors and procurement specialists in the fine chemical sector, this innovation offers a pathway to high-performance materials suitable for sensor development, metal ion extraction, and advanced catalytic systems. The patent outlines a streamlined three-step synthetic route that avoids the pitfalls of complex multi-step derivatizations often associated with functionalizing the calixarene upper rim.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of calixarenes, particularly at the para-positions of the phenolic rings, has been fraught with challenges regarding regioselectivity and reaction severity. Traditional methods often require harsh Lewis acids or extreme temperatures that can degrade the delicate macrocyclic structure or lead to polymerization byproducts. Furthermore, introducing sulfur-containing groups directly often suffers from poor yields due to the sensitivity of thiol groups to oxidation during the synthesis process. Many conventional routes rely on expensive protecting group strategies that add unnecessary steps, increase waste generation, and complicate the purification profile. For supply chain managers, these inefficiencies translate into higher costs of goods sold and longer lead times for obtaining research-grade quantities. The lack of a standardized, high-yield protocol for tetra-functionalized sulfur-calixarenes has previously limited their widespread adoption in industrial applications such as heavy metal remediation or specialized ligand design.
The Novel Approach
The methodology presented in the patent data introduces a highly efficient three-step sequence that circumvents these historical bottlenecks by utilizing a chloromethylation-thioacetylation-hydrolysis strategy. This approach begins with the controlled chloromethylation of the parent calix[4]arene, followed by a nucleophilic substitution with potassium thioacetate, and concludes with a mild hydrolysis to reveal the free thiol groups. 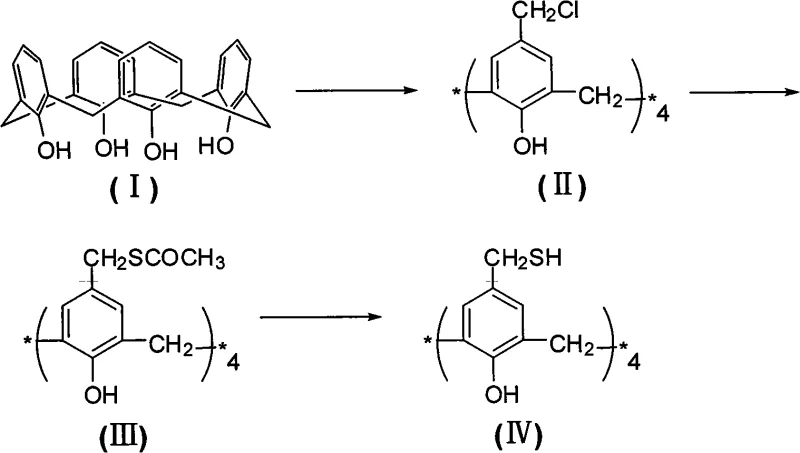 As illustrated in the reaction scheme, this pathway leverages the high reactivity of the chloromethyl intermediate while protecting the sensitive sulfur atoms as thioesters until the final step. This tactical protection prevents premature oxidation and ensures high fidelity in the final product structure. The process operates under atmospheric pressure and utilizes common organic solvents like acetic acid and acetonitrile, significantly reducing the engineering controls required for scale-up. By decoupling the introduction of the carbon scaffold from the installation of the sulfur functionality, the novel approach achieves yields close to or exceeding 70%, a substantial improvement over legacy methods.
As illustrated in the reaction scheme, this pathway leverages the high reactivity of the chloromethyl intermediate while protecting the sensitive sulfur atoms as thioesters until the final step. This tactical protection prevents premature oxidation and ensures high fidelity in the final product structure. The process operates under atmospheric pressure and utilizes common organic solvents like acetic acid and acetonitrile, significantly reducing the engineering controls required for scale-up. By decoupling the introduction of the carbon scaffold from the installation of the sulfur functionality, the novel approach achieves yields close to or exceeding 70%, a substantial improvement over legacy methods.
Mechanistic Insights into Chloromethylation and Nucleophilic Substitution
The core of this synthesis lies in the precise control of electrophilic aromatic substitution during the initial chloromethylation step. The reaction utilizes paraformaldehyde and dry hydrogen chloride gas in an acetic acid medium, where the phenolic hydroxyl groups activate the ortho-positions relative to themselves, which correspond to the para-positions of the methylene bridges. Maintaining the reaction temperature below 20°C is critical to prevent poly-chloromethylation or ring degradation, ensuring that exactly four chloromethyl groups are installed. Following this, the nucleophilic substitution with potassium thioacetate proceeds via an SN2 mechanism in anhydrous acetonitrile. The sulfur atom of the thioacetate anion attacks the benzylic carbon of the chloromethyl group, displacing the chloride ion. This step is thermodynamically driven and benefits from the polar aprotic nature of acetonitrile, which stabilizes the transition state. The final hydrolysis step employs sodium methoxide or ethoxide to cleave the thioester bond, generating the free mercapto group and a methyl or ethyl acetate byproduct. This base-catalyzed hydrolysis is rapid and clean, avoiding the need for harsh acidic conditions that could protonate the phenolic oxygens and disrupt the macrocycle.
Impurity control is inherently built into this mechanistic pathway through the physical properties of the intermediates. The chloromethyl intermediate precipitates from ice water, allowing for a simple filtration wash that removes unreacted formaldehyde and acid. The thioacetate intermediate is purified via recrystallization from isopropanol, which effectively removes inorganic salts like potassium chloride and excess thioacetate. Finally, the target mercapto compound is isolated by acidification and recrystallization from methanol. This sequence of precipitation and recrystallization steps ensures that the final product meets stringent purity specifications without requiring column chromatography. For R&D teams, this means the impurity profile is predictable and manageable, primarily consisting of partially substituted species which are removed during the crystallization phases. The stability of the thioester intermediate also allows for storage and quality control checks before the final deprotection, adding a layer of process robustness.
How to Synthesize 5,11,17,23-tetramercaptomethyl-25,26,27,28-tetrahydroxy-calix[4]arene Efficiently
The synthesis protocol described in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing temperature control and stoichiometric precision. The process begins with the suspension of the starting calixarene in acetic acid, cooled to 10°C, followed by the slow introduction of HCl gas to generate the chloromethyl derivative. After isolation, this solid is reacted with potassium thioacetate in refluxing acetonitrile under a nitrogen atmosphere to prevent oxidation. The resulting thioacetate solid is then dissolved in alcohol and treated with alkoxide at 0°C before warming to room temperature for hydrolysis. This structured approach minimizes side reactions and maximizes the recovery of the valuable macrocyclic skeleton.
- Perform chloromethylation of 25,26,27,28-tetrahydroxy-calix[4]arene with paraformaldehyde and HCl gas in acetic acid at low temperature.
- Conduct nucleophilic substitution using potassium thioacetate in anhydrous acetonitrile under reflux to introduce thioacetate groups.
- Hydrolyze the thioacetate intermediate using sodium methoxide or ethoxide in alcohol, followed by acidification to yield the final mercapto derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling advantages for procurement managers seeking cost reduction in fine chemical manufacturing. The reliance on commodity chemicals such as paraformaldehyde, acetic acid, and potassium thioacetate eliminates the need for exotic or proprietary reagents that often carry high price tags and long lead times. The absence of transition metal catalysts in the main synthetic steps further reduces the cost burden and simplifies the regulatory compliance associated with heavy metal residues in the final product. Additionally, the high yields reported in the patent examples suggest a material-efficient process that minimizes waste disposal costs. The ability to perform reactions at atmospheric pressure reduces the capital expenditure required for specialized high-pressure reactors, making the technology accessible for a wider range of manufacturing partners. These factors combine to create a supply chain that is both resilient and economically efficient.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the use of standard solvents like acetic acid and acetonitrile significantly lower the variable costs per kilogram. The high conversion rates mean less raw material is wasted, directly improving the margin profile for large-scale production runs. Furthermore, the simple workup procedures involving filtration and distillation reduce energy consumption compared to complex separation techniques.
- Enhanced Supply Chain Reliability: Since all starting materials are commercially available and sourced from broad chemical supply chains, the risk of single-source bottlenecks is minimized. The robustness of the reaction conditions, specifically the tolerance for standard laboratory glassware and atmospheric pressure, ensures that production can be easily transferred between different facilities without extensive re-validation. This flexibility is crucial for maintaining continuity of supply in a volatile global market.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the byproducts are primarily salts and simple organic esters that are easier to treat than heavy metal sludge. The use of recrystallization for purification reduces the volume of solvent waste compared to chromatographic methods. This aligns well with modern green chemistry initiatives and simplifies the environmental permitting process for scaling up to multi-ton production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sulfur-functionalized calixarenes. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for integrating this material into your specific research or production workflows.
Q: What are the key advantages of this sulfur-containing calixarene synthesis?
A: The process utilizes commercially available raw materials, operates at atmospheric pressure, and achieves high yields close to or exceeding 70%, making it cost-effective and scalable.
Q: How does the sulfur functionality enhance the molecule's application?
A: The introduction of mercapto groups significantly enhances the molecule's ability to recognize and complex with transition metal ions and heavy metal ions compared to traditional calixarenes.
Q: Is the purification process complex for these derivatives?
A: No, the purification involves straightforward recrystallization steps using common solvents like isopropanol and methanol, ensuring high purity without complex chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,11,17,23-tetramercaptomethyl-25,26,27,28-tetrahydroxy-calix[4]arene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity supramolecular intermediates play in the development of next-generation sensors and catalytic systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial application is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the structural integrity and functional group density of every batch. Our commitment to quality assurance means that you receive materials that perform consistently in your downstream applications, whether for metal ion recognition or advanced material science.
We invite you to collaborate with us to optimize your supply chain for these specialized calixarene derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and manufacturing goals. Let us be your partner in delivering high-performance chemical solutions with reliability and precision.
