Advanced Manufacturing of L-Gluconic Acid Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks efficient pathways for synthesizing rare sugar intermediates, particularly L-configured carbohydrates which are essential for specific therapeutic applications such as laxatives and bowel cleansing agents. Patent CN101553477A introduces a groundbreaking methodology for the preparation of L-gluconic acid and its derivatives, addressing the historical challenges associated with the scarcity and high cost of L-sugars. This technology leverages a novel three-step sequence starting from the readily available D-glucono-1,5-lactone, effectively inverting the stereochemistry to access the L-series. By optimizing reaction conditions, specifically regarding solvent systems and temperature controls, this process offers a robust alternative to traditional multi-day syntheses. For R&D directors and procurement specialists, understanding this patent is crucial as it represents a significant leap forward in the reliable supply of high-purity pharmaceutical intermediates. The ability to transform abundant D-sugar feedstocks into valuable L-isomers with improved kinetics establishes a new benchmark for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-glucose and its precursors has been plagued by inefficient methodologies that rely on scarce natural starting materials or excessively harsh reaction conditions. Traditional routes often utilize L-arabinose extracted from sugar beet pulp, a process that involves complex fermentation and purification steps that are inherently variable and costly. Furthermore, prior art methods for converting intermediates, such as the reaction described by Lundt et al., strictly required anhydrous conditions to prevent side reactions, necessitating the use of expensive dry solvents and rigorous moisture control protocols. These conventional approaches typically demanded reaction times extending up to three days to achieve completion, creating significant bottlenecks in production schedules. The reliance on ice-cold bases and prolonged stirring not only increased energy consumption but also limited the throughput capacity of manufacturing facilities. Such inefficiencies resulted in a supply chain vulnerable to delays and elevated costs, making the commercial viability of L-sugar derivatives a persistent challenge for fine chemical producers.
The Novel Approach
In stark contrast to these legacy methods, the process disclosed in the patent utilizes a streamlined pathway that dramatically enhances reaction kinetics and operational simplicity. The innovation begins with the controlled bromination of D-glucono-1,5-lactone, followed by a critical cyclization step that surprisingly benefits from the presence of catalytic amounts of water rather than strict anhydrous environments. This counter-intuitive finding allows for the use of standard industrial solvents like methyl isobutyl ketone (MIBK) without the need for costly drying procedures. The subsequent hydrolysis step is conducted at elevated temperatures between 45°C and 55°C, reducing the reaction time from days to merely six hours or less. This acceleration is achieved while maintaining high selectivity, preventing the degradation of the sensitive lactone ring which was a common issue in previous low-temperature protocols. By integrating these optimizations, the novel approach delivers a scalable solution that aligns perfectly with the demands of modern high-purity pharmaceutical intermediate production.
Mechanistic Insights into Fluoride-Mediated Epoxide Formation and Hydrolysis
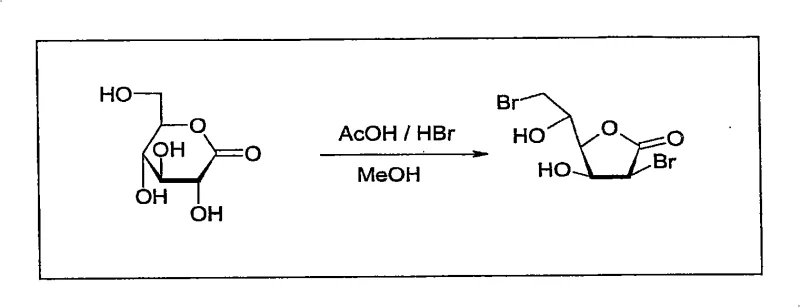
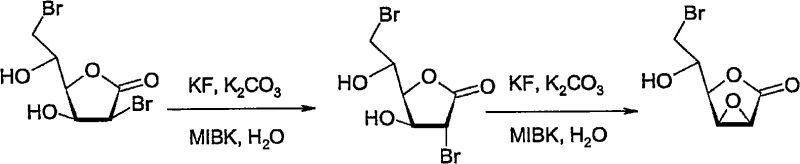
The core of this synthetic strategy lies in the precise manipulation of stereochemistry through a bromohydrin intermediate and a subsequent fluoride-mediated cyclization. The process initiates with the treatment of D-glucono-1,5-lactone with hydrogen bromide in acetic acid, generating a 2,6-dibromo-2,6-dideoxy-D-mannono-1,4-lactone species. This intermediate is then subjected to a Lewis base treatment, typically using potassium fluoride and potassium carbonate in an organic solvent. Mechanistically, the fluoride ion acts as a nucleophile to displace one of the bromine atoms, triggering an intramolecular substitution that forms a strained 2,3-anhydro epoxide ring. Crucially, the patent reveals that the presence of catalytic water, approximately 0.5% to 1% by weight, facilitates this transformation by enhancing the solubility and reactivity of the fluoride salt without causing hydrolytic degradation of the product. This delicate balance allows for rapid conversion at moderate temperatures around 40°C, ensuring that the reactive epoxide is generated efficiently before proceeding to the final ring-opening step.
Following the formation of the 6-bromo-6-deoxy-2,3-anhydro-D-manno-1,4-lactone epoxide, the final transformation involves a base-catalyzed hydrolysis that inverts the configuration to yield the L-gluconic acid skeleton. The reaction mixture is treated with a strong base, such as sodium or potassium hydroxide, to achieve a pH of at least 12, preferably between 13 and 14. Under these alkaline conditions at 45°C to 55°C, the epoxide ring undergoes nucleophilic attack by hydroxide ions, leading to ring opening and the formation of the L-configured polyhydroxy acid. The elevated temperature is critical here, as it overcomes the activation energy barrier rapidly, completing the reaction in under 6 hours compared to the 72 hours required by older methods. Impurity control is managed by the specific pH range; too low a pH results in incomplete conversion, while the controlled high pH ensures clean conversion without excessive degradation of the sugar backbone. This mechanistic precision ensures that the final product meets the stringent purity specifications required for downstream pharmaceutical applications.
How to Synthesize L-Gluconic Acid Efficiently
The synthesis of L-gluconic acid via this patented route offers a practical guide for chemists aiming to implement this technology in a pilot or production setting. The process is designed to be telescoped where possible, minimizing isolation steps and maximizing overall yield. Operators begin by preparing the dibromo intermediate in acetic acid, followed by solvent exchange into MIBK for the cyclization step. The critical control point is the water content during the fluoride treatment, which must be monitored to ensure it remains within the catalytic window to drive the epoxide formation. Once the epoxide is formed, the aqueous base is introduced directly or after minimal workup to effect the final rearrangement. The detailed standardized synthesis steps, including exact molar ratios and safety protocols for handling hydrogen bromide and strong bases, are outlined below to ensure reproducibility and safety compliance.
- React D-glucono-1,5-lactone with hydrogen bromide in acetic acid at 50-55°C, followed by methanol quenching to form 2,6-dibromo-2,6-dideoxy-D-mannono-1,4-lactone.
- Treat the dibromo intermediate with potassium fluoride and potassium carbonate in methyl isobutyl ketone (MIBK) containing catalytic water to form the epoxide.
- Hydrolyze the epoxide using a strong base at pH > 12 and 45-55°C for less than 6 hours to obtain L-gluconic acid or its salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible operational improvements that directly impact the bottom line and supply reliability. The elimination of strict anhydrous conditions removes the need for specialized drying equipment and expensive dry solvents, which are often subject to volatile pricing and supply constraints. Furthermore, the drastic reduction in reaction time from several days to less than a single shift allows for higher asset utilization rates, meaning existing reactor capacity can produce significantly more material over the same period. This efficiency gain reduces the working capital tied up in inventory and shortens the lead time for high-purity pharmaceutical intermediates. By simplifying the process workflow, manufacturers can also reduce the risk of batch failures associated with complex moisture-sensitive operations, thereby enhancing the overall consistency of supply for their global customers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of costly unit operations associated with maintaining anhydrous environments. By utilizing catalytic water instead of rigorously dried solvents, the process eliminates the energy and time costs associated with solvent drying and molecular sieve regeneration. Additionally, the use of common, inexpensive reagents like potassium fluoride and sodium hydroxide replaces more exotic or expensive catalysts found in alternative routes. The shortened reaction cycle time further contributes to cost optimization by reducing utility consumption, such as steam for heating and electricity for stirring over prolonged periods. These cumulative factors result in a substantially lower cost of goods sold, allowing for more competitive pricing strategies in the global market for specialty sugars.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this chemical pathway, which relies on widely available commodity chemicals rather than niche reagents. The ability to complete the synthesis in a fraction of the traditional time means that production schedules can be more flexible and responsive to sudden spikes in demand from downstream API manufacturers. Reduced processing time also minimizes the window for potential equipment downtime or maintenance issues to impact delivery timelines. Moreover, the simplified workup procedures, which involve standard aqueous extractions and crystallizations, reduce the complexity of waste management and logistics. This operational simplicity ensures a steady and predictable flow of materials, mitigating the risks of stockouts that can disrupt the production of critical pharmaceutical formulations.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process offers distinct advantages that align with modern green chemistry principles. The use of MIBK, a solvent with favorable recovery properties, combined with aqueous workups, facilitates efficient solvent recycling and reduces the volume of hazardous waste generated. The avoidance of cryogenic conditions (ice-cold bases) in favor of moderate heating reduces the energy footprint of the manufacturing process. As the process scales from laboratory to commercial tons, these efficiencies become even more pronounced, allowing for the commercial scale-up of complex pharmaceutical intermediates without proportional increases in environmental burden. The high selectivity of the reaction also minimizes the formation of difficult-to-remove byproducts, simplifying purification and ensuring that the final effluent streams are easier to treat and dispose of in compliance with regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-gluconic acid synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the specific advantages of the catalytic water system and the optimized temperature profiles that define this innovative approach.
Q: What is the key advantage of using catalytic water in the epoxide formation step?
A: The presence of catalytic water significantly accelerates the reaction rate and improves yield compared to strict anhydrous conditions, eliminating the need for expensive dry solvents.
Q: How does this process improve upon conventional L-glucose synthesis methods?
A: Conventional methods often require up to 3 days for reaction completion, whereas this novel process completes the critical hydrolysis step in under 6 hours at moderate temperatures.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common solvents like MIBK and aqueous workups, combined with shorter reaction times, makes the process highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Gluconic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient carbohydrate chemistry in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of L-gluconic acid or its derivatives meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of stereoselective synthesis, delivering consistent quality that supports your R&D and commercial manufacturing needs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this process can optimize your supply chain economics. Please contact us to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable supply of high-value pharmaceutical intermediates.
