Advanced Synthesis of Triacetylganciclovir: Enhancing Purity and Commercial Viability for Antiviral APIs
Introduction to Advanced Triacetylganciclovir Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiviral intermediates, and patent CN101475572A presents a significant advancement in the production of triacetylganciclovir. This document details a refined chemical synthesis method that utilizes monoacetylguanine as a key starting material, reacting it with 1,3-dihalo-2-(acetoxymethoxy)propane under specific catalytic conditions. Unlike traditional approaches that often struggle with regioselectivity and purification challenges, this novel process operates effectively at temperatures ranging from 100 to 150°C in suitable organic solvents. The resulting intermediate, dihalo-2-acetyl-ganciclovir, is subsequently converted into the final triacetylated product through a controlled acetylation step. This technological breakthrough addresses long-standing issues regarding product purity and process scalability, making it a highly attractive option for manufacturers aiming to optimize their supply chains for ganciclovir production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of triacetylganciclovir has relied heavily on the use of diacetylguanine as the primary nucleophile in condensation reactions. However, this conventional pathway is fraught with significant technical drawbacks that impact both yield and quality. The presence of acetyl groups on both nitrogen atoms in diacetylguanine creates steric and electronic environments that hinder selective alkylation at the desired N9 position, often leading to the formation of unwanted N7-isomers and other structural impurities. Furthermore, prior art methods, such as those described in earlier patents, frequently result in products that are syrupy in nature rather than crystalline. This physical state makes purification extremely difficult, often requiring multiple recrystallization steps of the final ganciclovir API to meet quality standards, which drastically reduces overall process efficiency and increases production costs due to significant material loss during repeated refining cycles.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN101475572A employs monoacetylguanine, which possesses a free amino group at the N9 position that is highly reactive and selective for alkylation. This strategic choice of starting material fundamentally alters the reaction landscape, ensuring that the condensation with 1,3-dihalo-2-(acetoxymethoxy)propane proceeds with high regioselectivity. The process avoids the use of phase transfer catalysts and excess acetic anhydride in the second step, which were previously known to cause decomposition of the sensitive triacetyl structure. By generating a crystalline intermediate that can be easily isolated and purified through simple filtration and washing, this new approach transforms the manufacturing workflow. It effectively eliminates the need for exhaustive purification of the final API, thereby streamlining the entire production line and ensuring a consistent supply of high-quality material suitable for downstream pharmaceutical applications.
Mechanistic Insights into Acid-Catalyzed Alkylation and Acetylation
The core of this synthesis lies in a sophisticated acid-catalyzed alkylation mechanism that facilitates the coupling of the purine base with the side chain. In the first stage, a catalyst such as p-toluenesulfonic acid (tosic acid) activates the 1,3-dihalo-2-(acetoxymethoxy)propane by promoting the formation of a reactive carbocation or oxocarbenium ion species at the acetoxymethyl group. This electrophilic center is then attacked by the nucleophilic N9 nitrogen of the monoacetylguanine. The absence of an acetyl protecting group at this specific nitrogen atom in the starting material is crucial, as it prevents competitive alkylation at the N7 position, which is a common side reaction in less optimized processes. The reaction is typically conducted in high-boiling aromatic solvents like xylene or toluene, which provide the necessary thermal energy to overcome activation barriers while maintaining a homogeneous reaction mixture that supports efficient mass transfer and conversion rates.
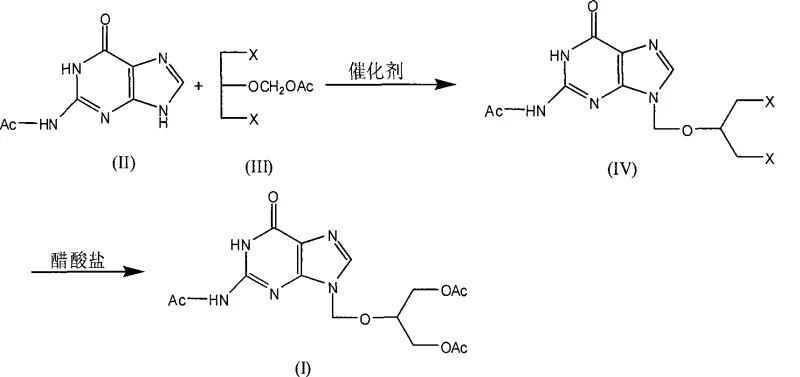
Following the initial condensation, the process moves to a nucleophilic substitution phase where the remaining halogen atoms on the side chain are displaced by acetate groups. This step utilizes an acetate salt, such as potassium acetate, in a polar aprotic solvent like dimethylformamide (DMF). The mechanism involves the acetate anion attacking the carbon-halogen bond, displacing the halide ion and forming the stable ester linkage required for the triacetyl structure. Crucially, the patent highlights that avoiding acetic anhydride in this step prevents the hydrolysis or degradation of the newly formed glycosidic bond, a vulnerability observed in older methods. The careful control of temperature between 70 and 150°C ensures complete conversion without inducing thermal decomposition, resulting in a product with a purity profile that exceeds 98% after standard recrystallization, demonstrating the robustness of this mechanistic pathway.
How to Synthesize Triacetylganciclovir Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the preparation of the reaction mixture containing monoacetylguanine and the halo-propyl derivative in a molar ratio optimized for complete consumption of the limiting reagent. Operators must maintain strict temperature profiles, initially heating the mixture to promote condensation and then adjusting conditions for the subsequent acetylation. Monitoring the reaction progress via thin-layer chromatography (TLC) or liquid chromatography is essential to determine the exact endpoint, ensuring that the intermediate is fully converted before workup. The isolation steps involve cooling the reaction mass to induce crystallization, followed by filtration and washing with solvents like acetone to remove residual starting materials and catalyst traces. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol provided below.
- Condense monoacetylguanine with 1,3-dihalo-2-(acetoxymethoxy)propane in an organic solvent like xylene using an acid catalyst such as tosic acid at 100-150°C.
- Isolate the intermediate dihalo-2-acetyl-ganciclovir through cooling, filtration, and washing with acetone to remove unreacted starting materials.
- React the isolated intermediate with an acetate salt (e.g., potassium acetate) in a polar solvent like DMF at 70-150°C, followed by extraction and recrystallization to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial benefits that directly address the pain points of procurement managers and supply chain directors. The shift from diacetylguanine to monoacetylguanine as a starting material represents a significant opportunity for cost optimization, as the monoacetyl derivative is generally less expensive to produce due to lower consumption of acetic anhydride during its own manufacture. Furthermore, the ability to isolate a crystalline intermediate simplifies the purification process, eliminating the need for complex chromatographic separations or multiple recrystallizations of the final API. This reduction in processing steps translates to shorter cycle times and lower utility consumption, contributing to a more sustainable and economically viable manufacturing operation. The robustness of the chemistry also implies a lower risk of batch failure, ensuring a more reliable supply of this critical antiviral intermediate for global markets.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the efficiency of the raw materials and the simplicity of the downstream processing. By utilizing monoacetylguanine, manufacturers avoid the costs associated with the extra acetylation step required for diacetylguanine, while also benefiting from higher reaction selectivity which reduces waste. The elimination of phase transfer catalysts and acetic anhydride in the second reaction step further lowers the bill of materials. Additionally, the crystalline nature of the product allows for efficient solid-liquid separation techniques like filtration, which are far less costly and time-consuming than the distillation or chromatography often required for syrupy residues, leading to substantial overall cost savings in large-scale production.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly improved by the robustness and reproducibility of this synthetic route. The use of readily available and stable reagents such as tosic acid and common organic solvents like xylene and DMF ensures that raw material sourcing is not a bottleneck. The high selectivity of the reaction minimizes the formation of hard-to-remove impurities, which historically have caused delays in quality control and release testing. By consistently producing an intermediate with purity levels exceeding 98%, manufacturers can reduce the lead time for batch release and ensure a steady flow of material to downstream API production facilities, thereby mitigating the risk of stockouts in the antiviral drug market.
- Scalability and Environmental Compliance: This process is inherently designed for scalability, utilizing standard unit operations such as reflux, filtration, and crystallization that are easily transferred from pilot plant to commercial scale. The avoidance of hazardous phase transfer catalysts and the reduction in solvent usage due to higher yields contribute to a greener manufacturing profile. The ability to recycle solvents like toluene and ethyl acetate further enhances the environmental sustainability of the process. Moreover, the high purity of the final product reduces the burden on wastewater treatment systems that would otherwise be loaded with organic impurities from extensive purification steps, aligning with increasingly stringent environmental regulations in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of triacetylganciclovir as described in the patent literature. These answers are derived from the specific experimental data and comparative analysis provided in the source documentation, offering clarity on the practical implementation of this technology. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing capabilities. The insights provided here cover aspects ranging from raw material selection to the specific advantages of the purification strategy employed.
Q: Why is monoacetylguanine preferred over diacetylguanine for synthesizing triacetylganciclovir?
A: Monoacetylguanine offers superior regioselectivity at the N9 position compared to diacetylguanine, significantly reducing the formation of N7-isomer impurities. Additionally, it eliminates the need for excessive acetic anhydride, lowering raw material costs and simplifying the purification process by yielding a crystalline product rather than a syrup.
Q: What catalysts are effective for this alkylation reaction?
A: The process utilizes strong acid catalysts to facilitate the condensation. Preferred catalysts include organic sulfonic acids like p-toluenesulfonic acid (tosic acid), as well as mineral acids like sulfuric acid or hydrochloric acid. Tosic acid is particularly favored for its balance of reactivity and ease of handling in organic solvents.
Q: How does this method improve the purity of the final ganciclovir API?
A: By producing a high-purity (>98%) crystalline triacetylganciclovir intermediate, this method minimizes the carryover of difficult-to-remove impurities into the final hydrolysis step. Traditional methods often yield syrupy intermediates with complex impurity profiles that require extensive recrystallization of the final API, whereas this route allows for efficient purification at the intermediate stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triacetylganciclovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antiviral medications. Our technical team has extensively analyzed advanced synthetic routes like the one described in CN101475572A and possesses the expertise to implement these methodologies at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of triacetylganciclovir meets the exacting standards required for pharmaceutical grade applications. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall production efficiency.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can reduce your total cost of ownership. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments tailored to your needs. Let us collaborate to drive innovation and efficiency in the global supply chain for antiviral therapeutics, ensuring that high-quality medicines reach patients faster and more affordably.
