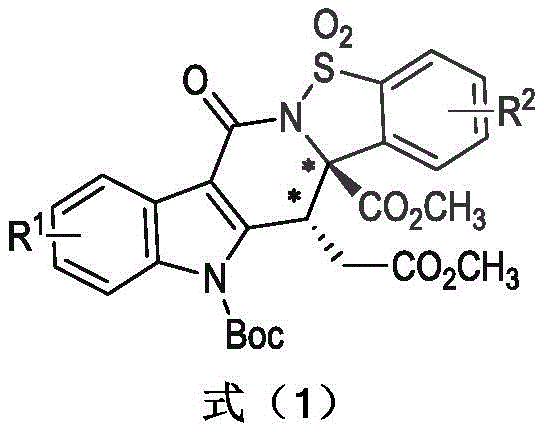
Advanced Organocatalytic Synthesis of Chiral Indolosulfonamide Intermediates for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to access complex chiral scaffolds with high stereochemical fidelity. Patent CN110938084A introduces a groundbreaking organocatalytic approach for the synthesis of chiral indolosulfonamide compounds, leveraging N-heterocyclic carbene (NHC) catalysis to achieve exceptional enantioselectivity. This technology addresses the critical need for efficient construction of nitrogen-containing heterocycles, which are prevalent in bioactive molecules. By utilizing simple aldehyde and imine precursors, this method enables the one-step assembly of intricate chiral architectures that were previously difficult to access with high optical purity. The significance of this innovation lies in its ability to bypass traditional transition metal catalysis, thereby eliminating the risk of heavy metal residue in the final active pharmaceutical ingredients. For R&D directors and process chemists, this represents a paradigm shift towards greener, more sustainable synthetic routes that align with modern regulatory standards for impurity control. The reported yields of up to 87% and enantiomeric ratios reaching 98:2 underscore the practical viability of this protocol for generating high-value intermediates required in drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing sulfonamide-fused indole systems often rely on multi-step sequences involving harsh reaction conditions and stoichiometric amounts of chiral auxiliaries or expensive transition metal catalysts. These conventional methods frequently suffer from poor atom economy and generate significant amounts of chemical waste, which poses challenges for environmental compliance and cost management in large-scale manufacturing. Furthermore, the use of transition metals necessitates rigorous purification steps to remove trace metal contaminants, which can be a bottleneck in the production of pharmaceutical intermediates destined for clinical use. The limited substrate scope of many existing protocols also restricts the ability to rapidly explore structure-activity relationships, as sensitive functional groups may not tolerate the aggressive conditions required for cyclization. Consequently, process chemists often face difficulties in scaling up these reactions without compromising yield or stereochemical integrity, leading to extended development timelines and increased operational costs for supply chain managers seeking reliable sources of complex heterocycles.
The Novel Approach
In contrast, the novel organocatalytic strategy disclosed in the patent utilizes N-heterocyclic carbenes to facilitate a highly enantioselective [4+2] annulation reaction under mild conditions. This approach eliminates the need for transition metals, thereby simplifying the workup process and significantly reducing the environmental footprint associated with heavy metal waste disposal. The reaction proceeds efficiently at room temperature in common solvents like tetrahydrofuran, using cesium carbonate as a benign base, which enhances the safety profile and operational simplicity of the process. The broad substrate tolerance allows for the incorporation of diverse electronic and steric properties on both the indole and sulfonimide components, enabling the rapid generation of diverse chemical libraries for biological evaluation. For procurement teams, this translates to a more resilient supply chain where raw materials are readily available and the synthesis is less susceptible to disruptions caused by the scarcity of precious metal catalysts. The high levels of stereocontrol achieved ensure that the resulting intermediates meet the stringent purity specifications required for downstream drug development, minimizing the need for costly recrystallization or chiral separation steps.
Mechanistic Insights into NHC-Catalyzed Cyclization
The core of this transformative synthesis lies in the unique reactivity of the N-heterocyclic carbene catalyst, which activates the aldehyde substrate through the formation of a Breslow intermediate. This key species undergoes an intramolecular proton transfer to generate a nucleophilic homoenolate equivalent, which then engages in a highly stereoselective addition to the sulfonimide electrophile. The chiral environment provided by the NHC catalyst dictates the facial selectivity of this bond-forming event, ensuring the precise installation of the chiral centers within the indolosulfonamide framework. Understanding this mechanistic pathway is crucial for R&D directors aiming to optimize reaction parameters or adapt the protocol for analogous substrates. The catalytic cycle is completed by the elimination of the NHC catalyst, regenerating the active species and releasing the target product with high fidelity. This mechanism avoids the formation of reactive radical species often associated with metal-catalyzed processes, thereby reducing the risk of side reactions and byproduct formation that could complicate purification. The detailed elucidation of this pathway provides a solid foundation for further methodological developments in organocatalysis, positioning this technology as a versatile tool for the construction of complex nitrogen-containing heterocycles.
![Reaction scheme and catalytic mechanism showing NHC activation and [4+2] annulation pathway](/insights/img/chiral-indolosulfonamide-organocatalysis-pharma-supplier-20260314191024-02.webp)
Impurity control is a paramount concern in the manufacture of pharmaceutical intermediates, and this organocatalytic method offers distinct advantages in this regard. The mild reaction conditions minimize the degradation of sensitive functional groups, such as esters and halogens, which are often present in advanced intermediates. By avoiding high temperatures and strong acids or bases, the process reduces the formation of decomposition products that can be difficult to separate from the desired compound. The high enantioselectivity ensures that the formation of unwanted stereoisomers is kept to a minimum, simplifying the chiral purity profile of the final material. For quality control teams, this means more consistent batch-to-batch reproducibility and a lower risk of failing specification tests due to unexpected impurities. The use of well-defined organic catalysts also reduces the variability often seen with metal catalysts, which can be sensitive to trace impurities in reagents or solvents. This robustness is essential for maintaining supply chain continuity, as it allows for the reliable production of high-purity materials that meet the rigorous standards of global regulatory agencies without the need for extensive reprocessing.
How to Synthesize Chiral Indolosulfonamide Efficiently
The synthesis of these valuable chiral intermediates begins with the preparation of the requisite substituted indole aldehyde precursors, which can be accessed through a sequence of reduction, oxidation, and olefination reactions. Once the aldehyde substrate is secured, it is reacted with a sulfonimide derivative in the presence of the NHC catalyst and a base in an inert solvent. The reaction mixture is stirred at room temperature for a defined period, after which the product is isolated via standard extraction and purification techniques. Detailed standardized synthesis steps see the guide below.
- Preparation of substituted indole aldehyde substrates via reduction, oxidation, and Wittig reaction sequences.
- Reaction of indole aldehyde with sulfonimide using NHC catalyst and Cs2CO3 base in THF solvent.
- Purification of the target chiral indolosulfonamide product via column chromatography and characterization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this organocatalytic technology offers substantial benefits for procurement and supply chain operations by streamlining the manufacturing process and reducing dependency on critical raw materials. The elimination of transition metal catalysts removes a significant cost driver and supply risk, as precious metals are subject to volatile market prices and geopolitical constraints. This shift towards organic catalysis aligns with the industry's move towards more sustainable and cost-effective manufacturing practices, allowing companies to optimize their cost structures without compromising on quality. The simplified workup and purification procedures reduce the consumption of solvents and consumables, further contributing to overall cost reduction in pharmaceutical intermediate manufacturing. For supply chain heads, the robustness of the reaction conditions ensures reliable production schedules and minimizes the risk of batch failures that could disrupt downstream operations. The ability to produce high-purity materials with consistent quality enhances the reliability of the supply chain, fostering stronger partnerships between manufacturers and their clients in the pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated heavy metal removal steps significantly lowers the direct material costs and processing time. This qualitative improvement in process efficiency translates to substantial cost savings over the lifecycle of the product, as fewer resources are required for purification and waste treatment. The use of inexpensive organic catalysts and common bases further enhances the economic viability of the process, making it an attractive option for large-scale production. By reducing the complexity of the synthesis, manufacturers can achieve better throughput and lower operational expenditures, providing a competitive edge in the market for high-value chiral intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available organic starting materials and reagents mitigates the risk of supply disruptions often associated with specialized metal catalysts. This increased availability ensures a more stable supply chain, allowing manufacturers to maintain consistent inventory levels and meet delivery commitments with greater confidence. The robustness of the reaction to varying substrate qualities also reduces the need for highly refined starting materials, further simplifying procurement logistics. For supply chain managers, this reliability is crucial for maintaining production schedules and ensuring the timely delivery of critical intermediates to downstream customers, thereby strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process highly scalable and environmentally friendly, aligning with increasingly stringent regulatory requirements. The reduced generation of hazardous waste simplifies compliance with environmental regulations and lowers the costs associated with waste disposal and treatment. This sustainability advantage is becoming a key differentiator in the chemical industry, as customers increasingly prioritize green manufacturing practices. The ease of scale-up allows manufacturers to quickly respond to market demand fluctuations, ensuring that supply can be ramped up efficiently without the need for extensive process re-engineering. This flexibility is essential for supporting the rapid development cycles of new drugs and agrochemicals, where time-to-market is a critical success factor.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organocatalytic synthesis method. These answers are derived from the specific technical details and beneficial effects outlined in the patent documentation, providing clarity on the practical application of this technology. Understanding these aspects is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing frameworks. The information provided here aims to facilitate informed decision-making regarding process adoption and supply chain optimization.
Q: What is the primary advantage of using NHC organocatalysis for indolosulfonamide synthesis?
A: The primary advantage is the ability to construct complex chiral frameworks with high enantioselectivity (up to 98:2 er) under mild conditions without using expensive transition metals, which simplifies downstream purification and reduces heavy metal contamination risks.
Q: How does this method improve substrate universality compared to traditional routes?
A: This method demonstrates excellent tolerance for various substituents on both the indole and sulfonimide rings, including electron-donating and electron-withdrawing groups like halogens, methoxy, and trifluoromethyl, allowing for diverse library synthesis.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes readily available starting materials and operates at room temperature with simple workup procedures, making it highly scalable and cost-effective for commercial production of high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indolosulfonamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the NHC-catalyzed route to deliver high-quality chiral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. Our commitment to technical excellence allows us to tackle complex synthetic challenges, providing our partners with a secure and reliable source of critical building blocks for drug development.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce costs. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits for your project. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. By partnering with us, you gain access to a wealth of chemical expertise and a dedicated team focused on driving your success through superior manufacturing solutions and responsive service.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
