Advanced Manufacturing of 2-Cyano-5-Bromopyridine: A Breakthrough in Process Efficiency and Purity
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access critical heterocyclic building blocks, and the recent disclosure in patent CN113603639A presents a significant advancement in the synthesis of 2-cyano-5-bromopyridine. This vital intermediate serves as a cornerstone for various bioactive molecules, yet its historical production has been plagued by yield limitations and purification challenges. The patented methodology introduces a robust nucleophilic substitution strategy that utilizes 2-nitro-5-bromopyridine as a readily available starting material, effectively bypassing the bottlenecks associated with conventional synthetic routes. By leveraging a direct displacement of the nitro group with a cyanide source, this innovation not only streamlines the operational workflow but also delivers a product profile with exceptional purity levels exceeding 99 percent HPLC area. For R&D directors and procurement specialists alike, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols that can be seamlessly integrated into existing supply chains for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-cyano-5-bromopyridine has relied heavily on two primary synthetic strategies, both of which suffer from inherent structural and economic inefficiencies that hinder large-scale optimization. The first conventional pathway involves the cyanation of 2,5-dibromopyridine using reagents such as cuprous cyanide or sodium cyanide, a process that frequently results in the formation of substantial quantities of 2,5-dicyanopyridine as a stubborn byproduct. This side reaction creates a nightmare for downstream processing, as the separation of the mono-cyano target from the di-cyano impurity requires rigorous and costly purification techniques, ultimately depressing the overall isolated yield and compromising the final quality of the intermediate. The second traditional approach utilizes 2-formyl-5-bromopyridine, which undergoes oximation followed by dehydration; however, this route is characterized by an elongated step count, the high cost of the aldehyde precursor, and complex operational requirements that increase the risk of batch failure. These legacy methods collectively impose a heavy burden on manufacturing budgets and extend lead times, making them increasingly unattractive for modern, cost-sensitive pharmaceutical production environments.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent data utilizes a direct nucleophilic aromatic substitution on 2-nitro-5-bromopyridine, offering a streamlined and highly efficient alternative. This method capitalizes on the strong electron-withdrawing nature of the nitro group at the 2-position, which activates the pyridine ring for displacement by cyanide ions under relatively mild thermal conditions. The reaction proceeds cleanly in polar aprotic solvents such as sulfolane or dimethylformamide, generating nitrite salts as the primary byproduct, which are inherently water-soluble and easily removed during the aqueous workup phase. This fundamental change in reaction chemistry eliminates the formation of difficult-to-separate dicyano impurities and avoids the expensive multi-step sequences associated with aldehyde precursors. Consequently, the process achieves a dramatic simplification of the purification train, allowing for the isolation of the target 2-cyano-5-bromopyridine with yields consistently surpassing 80 percent and purity levels reaching upwards of 99 percent, thereby setting a new benchmark for operational excellence in heterocyclic synthesis.
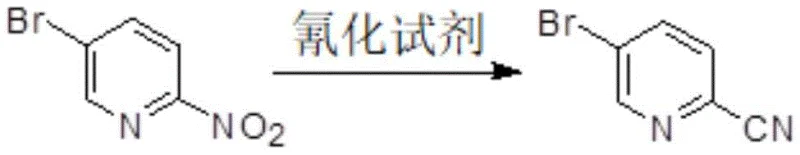
Mechanistic Insights into Nitro-Group Displacement Cyanation
The core of this technological breakthrough lies in the precise manipulation of nucleophilic aromatic substitution (SnAr) kinetics, where the 2-nitro-5-bromopyridine substrate acts as an electrophile activated by the powerful electron-withdrawing nitro moiety. When exposed to a cyanating agent such as sodium cyanide in a high-boiling polar solvent, the cyanide ion attacks the C-2 position of the pyridine ring, forming a transient Meisenheimer complex that subsequently collapses to expel the nitrite leaving group. This mechanism is highly favorable due to the stability of the nitrite anion in the reaction medium and the lack of competing nucleophilic sites on the bromine-substituted ring, which ensures high regioselectivity for the desired 2-cyano product. Furthermore, the choice of solvent plays a critical role in stabilizing the transition state and solubilizing the inorganic cyanide salt, with solvents like sulfolane providing an optimal dielectric environment that accelerates the reaction rate without promoting degradation or side reactions. Understanding this mechanistic pathway allows process chemists to fine-tune parameters such as temperature and stoichiometry to maximize conversion while minimizing the residence time required for complete reaction.
From an impurity control perspective, this mechanism offers a distinct advantage over metal-catalyzed cyanations or aldehyde condensations by fundamentally altering the byproduct profile. In traditional copper-catalyzed routes, trace metal contamination and over-cyanation are persistent issues that require specialized scavenging resins or chromatography to resolve. However, in this nitro-displacement route, the primary byproduct is an inorganic nitrite salt that partitions exclusively into the aqueous phase during the extraction workup, leaving the organic phase enriched with the target cyano compound. Additionally, the reaction conditions are carefully controlled to prevent the hydrolysis of the nitrile group or the displacement of the bromine atom, which could occur under excessively harsh basic conditions. The result is a crude product stream that is remarkably clean, requiring only a simple recrystallization from a solvent like toluene to achieve pharmaceutical-grade specifications, thus demonstrating a sophisticated alignment between reaction mechanism and practical purification logic.
How to Synthesize 2-Cyano-5-Bromopyridine Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot plant setting, operators must adhere to a strict protocol that balances reaction kinetics with safety and workup efficiency. The process begins with the careful charging of 2-nitro-5-bromopyridine and a stoichiometric amount of sodium cyanide into a reactor containing a polar aprotic solvent such as sulfolane, ensuring an inert atmosphere is maintained throughout to prevent oxidation or moisture ingress. The mixture is then heated to a temperature range of 130°C to 160°C, where it is stirred for a duration of approximately 3 to 5 hours, with reaction progress monitored via HPLC to ensure the starting material concentration drops below 1 percent. Following the completion of the substitution, the reaction mass is cooled and quenched with water, followed by extraction with an organic solvent like ethyl acetate to separate the product from the aqueous nitrite waste. The organic layer is concentrated under reduced pressure, and the resulting crude solid is dissolved in hot toluene and cooled to 0°C to induce crystallization, yielding the final white solid product with exceptional purity.
- Charge 2-nitro-5-bromopyridine and sodium cyanide into a reactor with sulfolane solvent under nitrogen protection.
- Heat the mixture to 130°C-160°C and stir for 2-5 hours until HPLC indicates less than 1% starting material remains.
- Quench with water, extract with ethyl acetate, concentrate, and recrystallize the crude product from toluene at 0°C to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that directly impact the bottom line and operational resilience. By shifting the raw material base to 2-nitro-5-bromopyridine, manufacturers can leverage a commodity chemical that is widely sourced and significantly less expensive than the specialized aldehyde or dibromo precursors used in older methods. This raw material substitution, combined with the elimination of expensive transition metal catalysts and complex purification resins, drives a substantial reduction in the overall cost of goods sold (COGS) for the final intermediate. Furthermore, the simplified one-step reaction sequence drastically reduces the manufacturing cycle time, allowing for faster turnover of production assets and a more responsive supply chain capable of meeting urgent demand fluctuations without the need for excessive safety stock. The robustness of the process also minimizes the risk of batch failures, ensuring a consistent and reliable flow of high-quality material to downstream customers.
- Cost Reduction in Manufacturing: The economic superiority of this method is driven by the replacement of costly reagents with inexpensive commodity chemicals and the removal of expensive metal catalysts. By utilizing sodium cyanide and sulfolane, the process avoids the high capital and operational expenditures associated with copper removal and recycling systems. Additionally, the high yield and purity reduce the volume of raw materials required per kilogram of finished product, leading to significant savings in material costs and waste disposal fees. The simplified workup procedure further lowers utility consumption by reducing the energy and time required for distillation and chromatography, creating a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing 2-nitro-5-bromopyridine is far less risky than procuring specialized aldehyde intermediates, as the nitro compound is produced on a massive scale for various industrial applications. This abundance ensures a stable supply base with multiple qualified vendors, mitigating the risk of shortages that can plague niche chemical markets. The shorter synthesis route also means that production lead times are compressed, enabling suppliers to fulfill orders more rapidly and maintain higher service levels. This agility is crucial for pharmaceutical clients who require just-in-time delivery to support their own clinical or commercial manufacturing schedules without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor equipment and common solvents that are easily managed in large-scale facilities. The generation of water-soluble inorganic byproducts simplifies wastewater treatment compared to organic-heavy waste streams from other routes, facilitating easier compliance with environmental regulations. Moreover, the ability to recover and recycle solvents like sulfolane and toluene further enhances the sustainability profile of the operation. This combination of scalability and environmental stewardship makes the technology attractive for long-term commercial partnerships focused on green chemistry principles and regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for 2-cyano-5-bromopyridine. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities and limitations. By clarifying these points, we aim to assist technical teams in evaluating the feasibility of adopting this route for their specific production needs and supply chain strategies.
Q: How does this new method improve purity compared to traditional dibromopyridine routes?
A: Traditional routes using 2,5-dibromopyridine often generate difficult-to-remove dicyanopyridine byproducts. This novel nitro-substitution route produces water-soluble nitrite byproducts that are easily washed away, achieving HPLC purity greater than 99%.
Q: What are the cost advantages of using 2-nitro-5-bromopyridine as a starting material?
A: 2-Nitro-5-bromopyridine is a widely available and inexpensive commodity chemical. By avoiding expensive aldehyde precursors or complex multi-step sequences, the overall manufacturing cost is significantly reduced while shortening the production cycle.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes common polar aprotic solvents like sulfolane or DMF and operates at moderate temperatures (50°C-200°C). The simple workup involving aqueous extraction and crystallization makes it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-5-Bromopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and commercialization of life-saving medicines. Our technical team has thoroughly analyzed the potential of this novel cyanation technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to full industrial maturity. We are committed to delivering 2-cyano-5-bromopyridine with stringent purity specifications that meet or exceed global pharmacopeial standards, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific solvent systems and thermal conditions required for this synthesis, ensuring a seamless transition from pilot scale to full commercial manufacturing.
We invite pharmaceutical partners and chemical buyers to engage with our technical procurement team to discuss how this optimized route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency and product quality.
