Advanced Nalfuraphine Manufacturing: A High-Purity Boc2O/DMAP Catalytic Route for Global Supply Chains
The pharmaceutical landscape for treating uremic pruritus in chronic kidney disease patients has been significantly advanced by the development of selective kappa-opioid receptor agonists, specifically Nalfuraphine. As the demand for this potent therapeutic agent grows, the efficiency and purity of its synthetic route have become paramount concerns for global supply chains. Patent CN114621238A introduces a transformative methodology for the preparation of Nalfuraphine, shifting away from traditional, harsh activation methods toward a sophisticated Boc2O (Di-tert-butyl dicarbonate) and DMAP (4-Dimethylaminopyridine) catalytic system. This innovation addresses long-standing challenges in amide bond formation within complex morphinan scaffolds, offering a pathway that combines operational simplicity with exceptional chemical fidelity. By leveraging this novel catalytic approach, manufacturers can achieve yields exceeding 96% and purities approaching 99.96%, setting a new benchmark for the reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities.
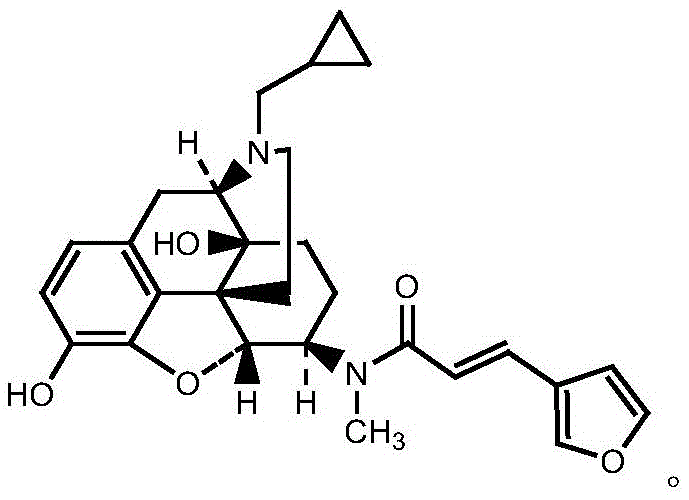
The structural complexity of Nalfuraphine, characterized by its rigid morphinan backbone and sensitive functional groups, demands a synthesis strategy that preserves stereochemical integrity while ensuring high conversion rates. The core of this invention lies in the direct amidation of 6 beta-N-methyl-naltrexone with 3-furan acrylic acid. Unlike conventional methods that rely on pre-activating the acid into a reactive and unstable acid chloride, this patent discloses an in-situ activation mechanism. This not only streamlines the process flow but also mitigates the risks associated with handling hazardous chlorinating agents. For R&D directors and process chemists, this represents a significant leap forward in process safety and robustness, ensuring that the final API precursor meets the stringent regulatory requirements for impurity profiles in modern drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nalfuraphine and its analogs has been plagued by inefficiencies inherent to traditional amide coupling strategies. As detailed in prior art such as CN1111900A and CN102325775A, the standard approach involves converting 3-furyl acrylic acid into 3-furyl acryloyl chloride using reagents like oxalyl chloride or formyl chloride. This two-step sequence introduces significant operational hazards, including the evolution of corrosive hydrogen chloride gas and the requirement for strict moisture control to prevent hydrolysis of the acid chloride intermediate. Furthermore, the reactivity of acid chlorides can lead to non-selective acylation of other nucleophilic sites on the morphinan ring, generating difficult-to-remove impurities that compromise the overall purity of the final product.
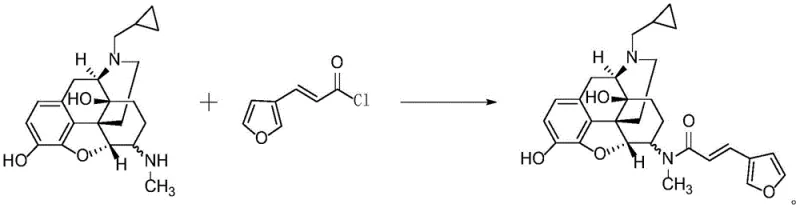
Alternative methods utilizing peptide coupling reagents, such as the BOP (Benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate) system described in WO2010006119A1, offer improved yields but suffer from critical downstream processing drawbacks. While the BOP-mediated reaction can achieve high conversion, the resulting crude product often exhibits poor purity, typically around 94-95%, necessitating labor-intensive and costly purification steps like column chromatography. For a commercial pharmaceutical intermediate supplier, the reliance on silica gel chromatography is a major bottleneck, drastically increasing solvent consumption, waste generation, and production time. These limitations highlight the urgent need for a method that balances high yield with intrinsic high purity, eliminating the need for extensive post-reaction cleanup.
The Novel Approach
The methodology disclosed in CN114621238A fundamentally reimagines the activation step by employing Boc2O as a mild and effective activating agent in conjunction with a nucleophilic catalyst, DMAP. In this system, Boc2O reacts with 3-furan acrylic acid to form a highly reactive mixed anhydride or active ester species in situ, which is then efficiently attacked by the secondary amine of the 6 beta-N-methyl-naltrexone substrate. This single-pot procedure operates under remarkably mild conditions, typically between 20°C and 35°C, avoiding the thermal stress that can degrade sensitive furan rings or epoxide moieties. The use of 2,6-lutidine or triethylamine as a base further buffers the reaction environment, ensuring that the pH remains optimal for amide formation without promoting side reactions.
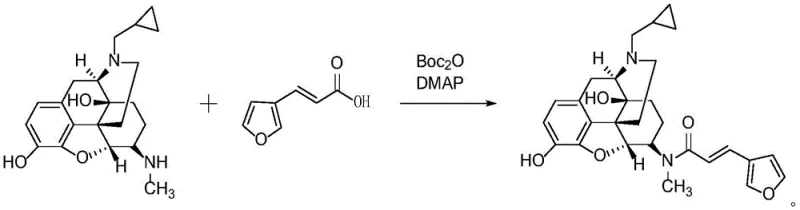
The impact of this novel approach on process chemistry is profound. By bypassing the isolation of unstable intermediates and avoiding corrosive chlorinating agents, the process becomes inherently safer and more environmentally friendly. The reaction demonstrates exceptional selectivity, targeting the specific nitrogen atom on the morphinan scaffold while leaving the phenolic hydroxyl groups and the ether bridge untouched. This chemoselectivity is the key driver behind the reported HPLC purities of over 99.8%, achieved directly after a simple slurry purification rather than chromatography. For procurement managers, this translates to a drastic reduction in raw material costs associated with purification media and a significant decrease in the environmental footprint of the manufacturing process, aligning perfectly with modern green chemistry initiatives.
Mechanistic Insights into Boc2O/DMAP Catalyzed Amidation
The success of this synthetic route hinges on the synergistic interaction between Boc2O and DMAP, which creates a highly efficient acyl transfer cycle. Mechanistically, DMAP acts as a powerful nucleophilic catalyst due to the resonance donation of the dimethylamino group into the pyridine ring, increasing the electron density at the nitrogen atom. This allows DMAP to attack the carbonyl carbon of the mixed anhydride formed between the acrylic acid and Boc2O, generating an even more reactive N-acylpyridinium intermediate. This cationic species is an exceptionally potent electrophile, capable of acylating the sterically hindered secondary amine of the naltrexone derivative much faster than the uncatalyzed reaction. This acceleration is crucial for driving the reaction to completion within a reasonable timeframe of 3 to 10 hours, even at near-ambient temperatures.
Furthermore, the choice of base plays a pivotal role in the mechanistic pathway. The use of hindered bases like 2,6-lutidine prevents the base itself from acting as a nucleophile and competing for the activated acid species, a common side reaction with less hindered amines. Instead, the base serves primarily to scavenge the tert-butyl carbonate byproduct and maintain the neutrality of the reaction medium. This precise control over the reaction micro-environment minimizes the formation of racemization byproducts or O-acylation impurities, which are common pitfalls in the synthesis of complex alkaloids. The result is a clean reaction profile where the desired amide bond is formed with high fidelity, preserving the critical stereochemistry at the C6 position of the morphinan core, which is essential for the biological activity of the final kappa-opioid agonist.
How to Synthesize Nalfuraphine Efficiently
The implementation of this Boc2O/DMAP protocol offers a streamlined workflow that is readily adaptable to both pilot and commercial scales. The process begins with the charging of acetonitrile, a solvent chosen for its excellent solubility profile for both the polar morphinan substrate and the organic reagents, into a reactor under an inert nitrogen atmosphere. The sequential addition of Boc2O, 3-furan acrylic acid, the amine substrate, DMAP, and the base initiates the catalytic cycle immediately upon mixing. The reaction is exothermic but manageable, requiring only mild heating to 28°C to maintain optimal kinetics. Following the reaction period, the work-up is remarkably straightforward: quenching with water followed by extraction with dichloromethane allows for the separation of the organic product from water-soluble byproducts like urea derivatives and salts.
- Charge a reactor with acetonitrile, Boc2O, 3-furan acrylic acid, 6 beta-N-methyl-naltrexone, DMAP, and 2,6-lutidine under inert gas protection.
- Maintain the reaction temperature between 20°C and 35°C and stir for 3 to 10 hours until completion.
- Quench with water, extract with dichloromethane, concentrate to an oil, and slurry in methanol to isolate the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this novel synthesis route presents a compelling value proposition centered on cost efficiency and operational reliability. The elimination of hazardous acid chloride reagents removes the need for specialized corrosion-resistant equipment and complex scrubbing systems for HCl gas, thereby reducing capital expenditure and maintenance costs. Moreover, the avoidance of chromatographic purification is a game-changer for cost reduction in pharmaceutical intermediate manufacturing. Chromatography is notoriously solvent-intensive and slow; replacing it with a simple crystallization or slurry step drastically cuts down on solvent purchase, recovery, and disposal costs, while simultaneously shortening the batch cycle time. This efficiency gain allows for higher throughput and better asset utilization in existing manufacturing facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive and wasteful purification methods with a high-yield precipitation technique. By achieving purities greater than 99.7% directly from the reaction work-up, manufacturers eliminate the substantial costs associated with silica gel, elution solvents, and the extended labor hours required for column chromatography. Additionally, the reagents used, such as Boc2O and DMAP, are commodity chemicals available from multiple global suppliers, ensuring competitive pricing and reducing the risk of supply bottlenecks associated with proprietary coupling reagents like BOP. This simplification of the bill of materials directly contributes to a lower cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The robustness of the Boc2O/DMAP system enhances supply chain continuity by reducing process sensitivity. Traditional acid chloride routes are highly sensitive to moisture, often leading to batch failures if humidity control is not perfect. In contrast, the new method tolerates a broader range of operating conditions, reducing the likelihood of off-spec batches and ensuring consistent delivery schedules. The use of common solvents like acetonitrile and dichloromethane further secures the supply chain, as these are standard inventory items in most fine chemical plants, unlike specialized reagents that may have long lead times. This reliability is critical for maintaining the continuous flow of materials required for downstream formulation and clinical trials.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns with the principles of green chemistry by minimizing waste generation. The E-factor (mass of waste per mass of product) is significantly improved due to the absence of chromatography silica and the reduced volume of solvents needed for purification. This makes the process easier to scale from kilogram to multi-ton quantities without encountering the heat transfer or mixing limitations often seen in heterogeneous chromatographic processes. Furthermore, the reduced solvent load simplifies waste treatment and lowers the environmental compliance burden, facilitating smoother regulatory approvals and audits for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of purity, yield, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is the Boc2O/DMAP system superior to acid chloride methods for Nalfuraphine?
A: The Boc2O/DMAP system avoids the use of corrosive reagents like oxalyl chloride and eliminates the generation of HCl gas, resulting in milder conditions and significantly higher product purity without the need for complex chromatographic purification.
Q: What represents the critical quality attribute in this synthesis?
A: The critical quality attribute is the HPLC purity, which consistently exceeds 99.7% using this method, compared to roughly 94-95% in prior art methods that require extensive post-reaction cleaning.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes common solvents like acetonitrile and operates at ambient temperatures (20-35°C), making it highly suitable for large-scale commercial manufacturing with simplified work-up procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nalfuraphine Supplier
As the global demand for effective treatments for chronic pruritus continues to rise, securing a stable and high-quality supply of Nalfuraphine is critical for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the Boc2O/DMAP system to deliver superior intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational clients. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Nalfuraphine meets the highest standards of chemical integrity and safety required for clinical and commercial applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-efficiency process. We encourage you to request specific COA data and route feasibility assessments to verify how our optimized manufacturing capabilities can accelerate your development timelines and reduce your overall production costs. Let us be your strategic partner in bringing this vital medication to the patients who need it most.
