Advanced Synthetic Routes for M6G Oxidized Impurities: A Technical Breakthrough for Pharmaceutical Quality Control
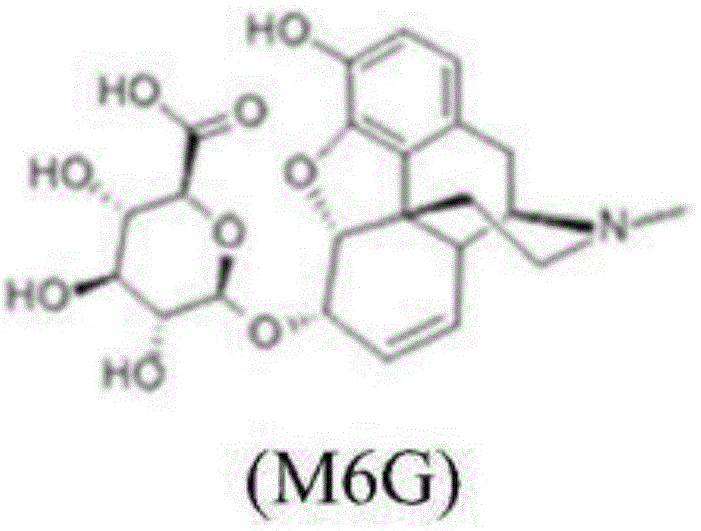
The pharmaceutical industry faces rigorous challenges in maintaining the purity and stability of opioid analgesics, particularly regarding their major metabolites. Patent CN110054656B, published in early 2021, addresses a critical gap in the availability of specific oxidized impurities derived from Morphine-6-Glucuronide (M6G). As a potent central nervous system μ-receptor agonist, M6G offers superior analgesic properties with reduced side effects compared to morphine, yet it is susceptible to oxidative degradation in aqueous solutions. This degradation leads to the formation of complex impurities, specifically 10-carbonyl and 10-hydroxy derivatives, which must be strictly monitored for regulatory compliance. The disclosed technology provides a robust, scalable synthetic route to generate these elusive reference standards, ensuring that manufacturers can meet stringent quality control requirements for opioid formulations.
For R&D directors and quality assurance teams, the ability to synthesize these specific degradation products is not merely an academic exercise but a regulatory necessity. The patent outlines a sophisticated chemical strategy that transforms stable precursors into the target oxidized impurities through a sequence of protection, selective oxidation, and deprotection. By securing a reliable source for these complex intermediates, pharmaceutical companies can accelerate their stability studies and impurity profiling, ultimately reducing the risk of regulatory delays during new drug applications. This technological advancement underscores the importance of having a reliable pharmaceutical intermediate supplier capable of navigating complex organic synthesis challenges.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 10-carbonyl and 10-hydroxymorphine-6-glucuronide was fraught with failure and inefficiency. Existing literature, such as methods cited from Die Pharmazie (1984) and Pharmazie (1978), attempted to synthesize these compounds but consistently failed to produce the desired 10-oxidized products in identifiable quantities. The primary limitation of these conventional approaches lies in the lack of selectivity and the instability of the intermediates under standard oxidative conditions. Without proper protection of the amine functionality and careful control of the reaction environment, direct oxidation attempts often lead to decomposition or the formation of intractable mixtures rather than the specific C-10 oxidized targets. This inability to generate authentic samples hampers the development of accurate analytical methods, leaving manufacturers vulnerable to unidentified impurity peaks during HPLC analysis.
The Novel Approach
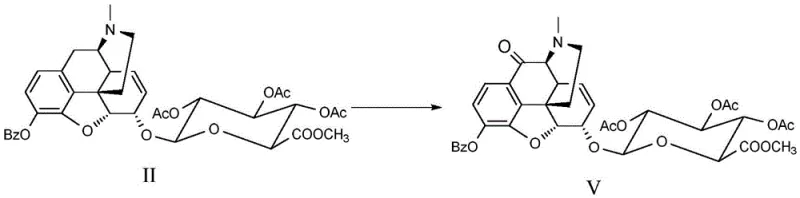
The patented methodology revolutionizes this landscape by introducing a stepwise protection-oxidation strategy that guarantees success where previous attempts failed. The core innovation involves converting the starting material (Formula II) into a carbamate-protected intermediate (Formula III) before subjecting it to oxidation. This protection step is crucial as it stabilizes the nitrogen atom, preventing unwanted side reactions during the harsh oxidative conditions required to functionalize the C-10 position. Subsequent oxidation using chromium trioxide or manganese dioxide in the presence of trifluoroacetic acid selectively installs the carbonyl group. This approach not only achieves the desired chemical transformation but does so with sufficient yield and purity to be viable for commercial reference standard production, representing a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Selective C-10 Oxidation and Carbamate Chemistry
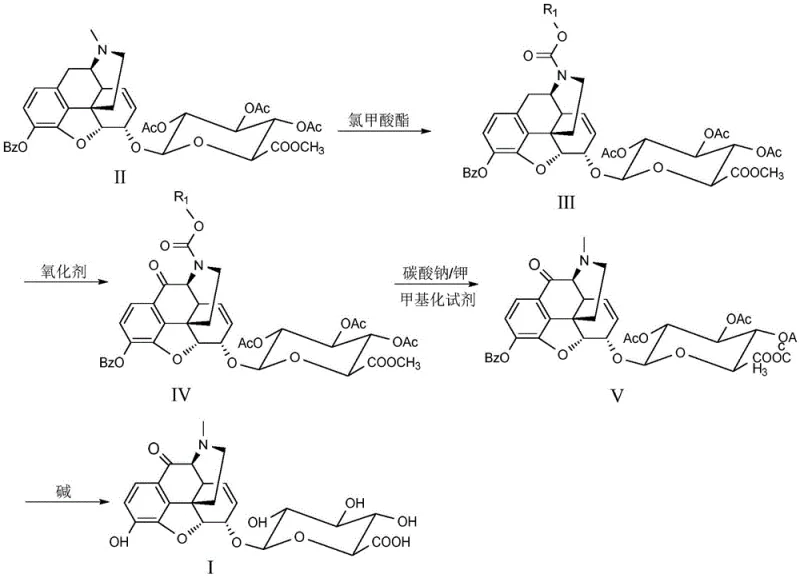
The mechanistic elegance of this synthesis lies in the precise manipulation of the morphine skeleton's reactivity. The process begins with the formation of a carbamate at the nitrogen center using chloroformates, which serves a dual purpose: it protects the amine from oxidation and modifies the electronic environment of the ring system to facilitate subsequent transformations. Once the protected intermediate (Formula III) is secured, the introduction of strong oxidants like CrO3 or MnO2, aided by superacidic conditions provided by trifluoroacetic acid or trifluoromethanesulfonic acid, drives the oxidation specifically at the benzylic C-10 position. This selectivity is paramount, as the molecule contains other oxidizable sites, including the phenolic hydroxyls and the alkene functionality. The acidic medium likely protonates intermediate species, directing the oxidant to the electron-rich C-10 position while the carbamate group prevents N-oxide formation.
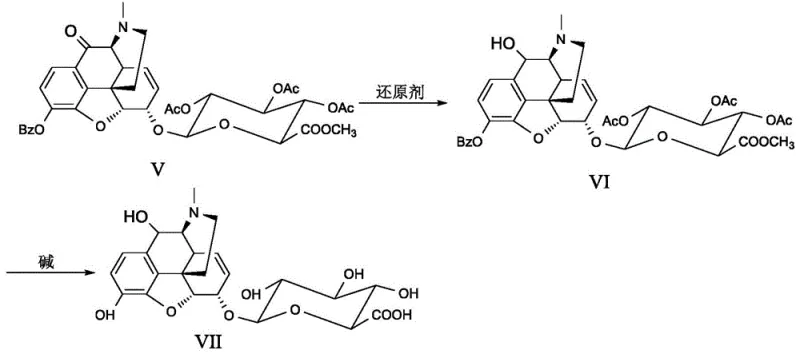
Following the oxidation to the ketone (Formula IV), the synthesis requires the restoration of the native N-methyl group found in natural morphine derivatives. This is achieved through a reductive amination process using formaldehyde and a reducing agent like sodium cyanoborohydride. This step effectively replaces the carbamate protecting group with the desired methyl group, yielding Formula V. The final stage involves a gentle hydrolysis using lithium hydroxide in aqueous methanol at low temperatures (-15 to 0°C). This mild basic condition is carefully chosen to cleave the ester bonds of the glucuronide and any remaining protecting groups without epimerizing the sensitive stereocenters or degrading the newly formed C-10 carbonyl. Such precise control over reaction conditions ensures the final product retains the stereochemical integrity required for biological activity and analytical accuracy.
How to Synthesize 10-Carbonyl Morphine-6-Glucuronide Efficiently
The synthesis of these high-value impurities requires strict adherence to the patented protocol to ensure reproducibility and safety. The process involves handling strong oxidants and acidic reagents, necessitating specialized equipment and rigorous temperature control. The initial protection step sets the foundation for the entire sequence, and any deviation in stoichiometry or temperature can compromise the yield of the critical oxidation step. Operators must be trained to manage the exothermic nature of the oxidation and the sensitivity of the intermediates to moisture and light. For a detailed breakdown of the operational parameters, reagent grades, and workup procedures necessary to achieve the reported yields and purities, please refer to the standardized synthesis guide below.
- Protect the amine functionality of the morphine backbone by reacting Formula II with chloroformates to form the carbamate intermediate (Formula III).
- Perform selective oxidation at the C-10 position using chromium trioxide or manganese dioxide in the presence of trifluoroacetic acid to yield the ketone intermediate (Formula IV).
- Restore the N-methyl group via reductive amination with formaldehyde, followed by global deprotection and hydrolysis using lithium hydroxide to obtain the final target compound (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the availability of this synthetic route translates directly into risk mitigation and operational efficiency. The ability to internally produce or source these specific impurities eliminates the dependency on scarce natural isolates or unreliable third-party vendors who may struggle with the complex chemistry. This self-sufficiency ensures a continuous supply of critical reference materials needed for batch release testing and stability monitoring. Furthermore, the robustness of the described method suggests that it can be scaled up from laboratory to pilot plant scales without significant re-engineering, providing a stable long-term supply chain for these essential quality control agents.
- Cost Reduction in Manufacturing: By utilizing a defined chemical synthesis rather than extraction or unpredictable degradation studies, manufacturers can significantly lower the cost per gram of these reference standards. The use of commodity chemicals like chromium trioxide, formaldehyde, and chloroformates keeps raw material costs low, while the high selectivity of the reaction minimizes waste generation and purification expenses. Eliminating the need for extensive trial-and-error optimization, which plagued previous methods, further reduces the overall R&D expenditure associated with impurity management.
- Enhanced Supply Chain Reliability: The reliance on a clear, patent-protected synthetic route ensures that supply is not subject to the vagaries of agricultural sourcing or complex isolation processes. The starting materials are readily available fine chemicals, reducing the risk of raw material shortages. This reliability is crucial for maintaining uninterrupted quality control operations, ensuring that every batch of opioid medication can be tested against verified standards without delay, thereby safeguarding the continuity of drug distribution.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard organic solvents like dichloromethane and methanol which are easily recovered and recycled in industrial settings. While the use of chromium reagents requires careful waste management, the high yield and purity of the product mean that less solvent and energy are consumed per unit of product compared to inefficient legacy methods. This efficiency aligns with modern green chemistry initiatives by maximizing atom economy in the critical oxidation steps and reducing the overall environmental footprint of impurity synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their internal reference standard programs or outsourcing strategies. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance to real-world application scenarios.
Q: Why is the synthesis of 10-carbonyl morphine-6-glucuronide critical for pharmaceutical development?
A: M6G is a major active metabolite of morphine. During stability testing and storage, M6G can oxidize to form impurities like 10-carbonyl and 10-hydroxy derivatives. Regulatory agencies require these specific impurities to be identified and quantified to ensure drug safety, making their synthesis essential for creating reference standards.
Q: How does the patented method overcome the limitations of prior art synthesis routes?
A: Previous methods described in literature (e.g., Die Pharmazie) failed to successfully produce the 10-carbonyl/hydroxy morphine-6-glucuronide targets. This patent introduces a robust protection-oxidation-deprotection strategy that prevents side reactions and allows for high-yield isolation of these unstable oxidized species.
Q: What are the key reagents used for the selective oxidation step in this process?
A: The process utilizes strong oxidizing agents such as chromium trioxide (CrO3) or manganese dioxide (MnO2) combined with acidic promoters like trifluoroacetic acid (TFA) or trifluoromethanesulfonic acid. This combination ensures selective oxidation at the C-10 position without degrading the sensitive glucuronide moiety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Carbonyl Morphine-6-Glucuronide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurities play in the global pharmaceutical supply chain. Our team of expert chemists has extensively analyzed the pathways described in CN110054656B and possesses the technical capability to execute this complex synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligrams for analytical method validation or kilograms for routine QC, we can deliver. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 10-carbonyl or 10-hydroxymorphine-6-glucuronide meets the highest international standards for reference materials.
We invite pharmaceutical partners to collaborate with us to secure their supply of these vital compounds. By leveraging our expertise in opioid intermediate synthesis, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your quality control operations remain robust, compliant, and efficient in the face of evolving regulatory landscapes.
