Scalable Asymmetric Synthesis of Bioactive Marine Natural Products for Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks reliable sources for complex bioactive molecules, particularly those derived from marine environments which often possess unique therapeutic profiles. Patent CN102093166B introduces a groundbreaking methodology for the total synthesis of the marine natural product (S)-trans-1-yn-4-en-eicosan-3-ol, a compound known for its potent anticancer and immunosuppressive activities. Historically, obtaining this molecule relied heavily on extraction from marine sponges like Cribrochalina vasculum, a process plagued by extremely low recovery rates of less than one part in 100,000. This patent delineates a sophisticated chemical pathway that bypasses these natural limitations through a highly efficient asymmetric addition reaction. By leveraging a chiral cyclopropane amino alcohol catalyst, the process constructs the critical chiral center in a single step with exceptional stereocontrol. For R&D directors and procurement specialists, this represents a pivotal shift from unsustainable harvesting to controllable, high-yield chemical manufacturing, ensuring a stable supply chain for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to acquiring (S)-trans-1-yn-4-en-eicosan-3-ol were fundamentally flawed due to their reliance on finite biological resources or overly complex synthetic routes. Early attempts involved extracting the compound directly from marine sponges, but the concentration in natural sources is so negligible that it renders commercial production impossible. Alternative synthetic strategies reported in prior art utilized carbohydrate derivatives as starting materials or employed enzymatic hydrolysis, both of which introduced significant operational complexities. These legacy methods often required multiple protection and deprotection steps, utilized expensive biocatalysts that were difficult to recycle, or suffered from poor stereochemical outcomes requiring extensive purification. Furthermore, routes starting from unsaturated ketones involving dihydroxylation reactions added unnecessary length to the synthesis, increasing the accumulation of impurities and lowering the overall throughput. For a supply chain manager, these factors translate into unpredictable lead times, volatile pricing, and an inability to scale production to meet clinical trial demands.
The Novel Approach
The methodology described in CN102093166B revolutionizes the production landscape by implementing a streamlined, linear synthesis starting from readily available bromopentadecane. The core innovation lies in the strategic use of an asymmetric addition reaction between an aliphatic aldehyde and trimethylsilylacetylene. This approach drastically reduces the step count compared to sugar-based syntheses and eliminates the need for rare biological enzymes. The process utilizes standard organometallic reagents like n-butyllithium and dimethylzinc, which are commodity chemicals available in bulk quantities globally. By constructing the chiral center late in the synthesis via the zinc-catalyzed addition, the route ensures that the stereochemical integrity is established just before the final deprotection, minimizing the risk of racemization during earlier transformations. This logical disconnection not only simplifies the operational workflow but also significantly enhances the atom economy of the process, making it an ideal candidate for cost reduction in fine chemical manufacturing.
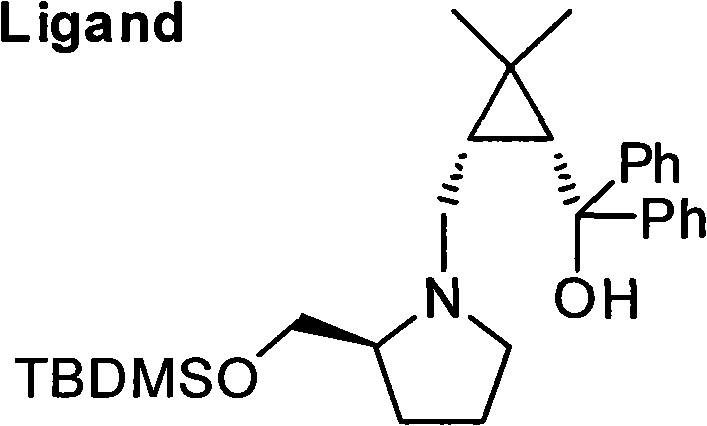
Mechanistic Insights into Chiral Zinc-Catalyzed Asymmetric Addition
The heart of this synthetic breakthrough is the enantioselective addition of an alkynyl zinc species to a prochiral aldehyde, mediated by a specialized chiral ligand. The mechanism begins with the in situ generation of a zinc acetylide from trimethylsilylacetylene and dimethylzinc. This nucleophilic species is then activated and directed by the chiral cyclopropane amino alcohol ligand, which coordinates to the zinc center to create a rigid, chiral environment. This coordination sphere effectively shields one face of the approaching aldehyde substrate, forcing the nucleophilic attack to occur from the opposite face with high fidelity. The result is the preferential formation of the (S)-configured alkynyl secondary alcohol. The steric bulk of the cyclopropane ring and the specific orientation of the hydroxyl and amine groups on the ligand are critical for inducing this high level of asymmetry. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters such as temperature and solvent polarity to maximize enantiomeric excess.
Following the asymmetric addition, the resulting intermediate possesses an optical purity of approximately 80% ee. While this is a strong outcome for a catalytic reaction, the patent further details a purification strategy that leverages the physical properties of the enantiomers. A simple recrystallization from n-hexane is sufficient to upgrade the optical purity to greater than 97% ee. This phenomenon suggests that the minor enantiomer remains soluble in the mother liquor while the major (S)-enantiomer crystallizes out efficiently. From an impurity control perspective, this is a highly desirable trait as it avoids the need for expensive chiral chromatography columns (like Chiralcel AD-H) on a production scale. The ability to achieve pharmaceutical-grade purity through crystallization rather than chromatography is a massive advantage for commercial scalability, reducing solvent consumption and processing time significantly while ensuring the final API intermediate meets rigorous regulatory specifications.
How to Synthesize (S)-trans-1-yn-4-en-eicosan-3-ol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process initiates with a metal coupling reaction at cryogenic temperatures to install the carbon backbone, followed by a selective reduction and oxidation sequence to prepare the electrophilic aldehyde. The critical asymmetric step is performed at room temperature, which is energetically favorable compared to cryogenic asymmetric reactions.
- Perform metal coupling of bromopentadecane with propargyl alcohol using n-BuLi and HMPA at low temperature to form pentadecyl propargyl alcohol.
- Reduce the alkyne intermediate using LiAlH4 to obtain trans-pentadecyl allyl alcohol, followed by PCC oxidation to generate the key aldehyde intermediate.
- Execute asymmetric addition using trimethylsilylacetylene and dimethylzinc catalyzed by a chiral cyclopropane amino alcohol ligand, followed by deprotection and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to this synthetic route offers profound strategic benefits regarding cost stability and supply continuity. The reliance on marine sponges created a bottleneck that was susceptible to environmental fluctuations and geopolitical issues regarding marine resource access. By shifting to a fully synthetic route based on petrochemical derivatives like bromopentadecane, the supply chain becomes decoupled from biological variability. The raw materials required for this synthesis are commodity chemicals produced in multi-ton quantities globally, ensuring that price volatility is minimized. Furthermore, the elimination of enzymatic steps removes the dependency on specialized biotech suppliers, consolidating the supply base to standard chemical manufacturers. This consolidation simplifies vendor management and reduces the risk of single-source failures, thereby enhancing the overall resilience of the supply chain for this critical anticancer intermediate.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive and scarce natural sources with abundant chemical feedstocks. The use of zinc-based catalysis instead of precious metals like palladium or rhodium significantly lowers the catalyst cost per kilogram of product. Additionally, the high yields observed in each step, particularly the 90-95% yields in the backbone construction phases, minimize material waste. The ability to purify the final product via recrystallization rather than preparative HPLC further drives down processing costs by reducing solvent usage and equipment time. These factors combine to create a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the final pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply security is paramount for clinical and commercial drug production. This synthetic route offers a distinct advantage by utilizing robust, well-understood chemical transformations that are easily transferable between different manufacturing sites. Unlike extraction processes which are location-specific, this chemistry can be replicated in any GMP-compliant facility equipped with standard reactors. The scalability of the reaction conditions, which do not require extreme pressures or temperatures beyond standard industrial capabilities, ensures that production can be ramped up rapidly to meet demand surges. This flexibility provides a buffer against supply disruptions and allows for just-in-time manufacturing strategies that reduce inventory holding costs.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the process is designed for efficiency. The atom economy of the asymmetric addition step is high, meaning most of the reactant mass ends up in the product, reducing the burden on waste treatment facilities. While the process does utilize organozinc reagents which require careful quenching, standard industrial protocols exist for handling these safely. The avoidance of heavy metal catalysts simplifies the purification of the final API, reducing the risk of toxic metal residues that could trigger regulatory delays. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy for technical decision-makers.
Q: What is the optical purity achievable with this synthetic route?
A: The patented process achieves an initial optical purity of 80% ee after the asymmetric addition step, which can be enhanced to greater than 97% ee through a single recrystallization step, meeting stringent pharmaceutical standards.
Q: How does this method compare to natural extraction?
A: Natural extraction from marine sponges yields less than one part in 100,000, making it commercially unviable. This chemical synthesis provides a robust, scalable alternative with high yields and consistent quality.
Q: Is the chiral catalyst cost-effective for large scale production?
A: Yes, the process utilizes a zinc-based catalytic system with a specialized amino alcohol ligand. Zinc reagents are significantly more affordable than precious metals like palladium or rhodium, reducing overall raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-trans-1-yn-4-en-eicosan-3-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation anticancer therapies. Our team of expert process chemists has thoroughly analyzed the pathway described in CN102093166B and is prepared to execute this synthesis with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale research to full commercial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >97% ee required for this chiral marine natural product. We understand that consistency is key, and our quality management systems are designed to deliver batch-after-batch reliability.
We invite you to collaborate with us to optimize this route for your specific needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic feasibility of this synthesis versus your current supply method, or you need specific COA data to support your regulatory filings, our technical procurement team is ready to assist. Contact us today to request route feasibility assessments and discuss how we can secure your supply chain for this valuable pharmaceutical intermediate.
