Revolutionary Room-Temperature Synthesis of Imidazo[1,2-a]pyridine Derivatives for Commercial Scale-up
Revolutionary Room-Temperature Synthesis of Imidazo[1,2-a]pyridine Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and economically viable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN113372346B introduces a groundbreaking four-component one-pot synthesis method for 3-aliphatic aminomethyl imidazo[1,2-a]pyridine compounds, a structural motif prevalent in numerous CNS drugs such as Zolpidem and Alpidem. This innovative approach utilizes 2-aminopyridine, alpha-bromoaryl ethyl ketone, secondary amines, and dichloromethane under mild conditions, effectively bypassing the harsh requirements of traditional methodologies. By enabling the reaction to proceed at room temperature without external metal catalysts or oxidants, this technology represents a significant leap forward in green chemistry and process efficiency for reliable pharmaceutical intermediate supplier networks globally.
![General reaction scheme for the four-component one-pot synthesis of 3-aliphatic aminomethyl imidazo[1,2-a]pyridine compounds](/insights/img/imidazo-pyridine-synthesis-pharma-supplier-20260304011400-05.webp)
The strategic implementation of this synthesis protocol allows for the direct construction of complex heterocyclic systems with high atom utilization rates, addressing the growing demand for sustainable manufacturing practices in the fine chemical sector. The ability to generate diverse derivatives through simple variation of the aryl and amine components provides medicinal chemists with a powerful tool for rapid library generation and lead optimization. Furthermore, the elimination of toxic reagents and the use of environmentally benign solvents like ethanol align perfectly with modern regulatory standards for environmental compliance and worker safety. As we delve deeper into the technical specifics, it becomes evident that this patent offers a transformative solution for cost reduction in API manufacturing while ensuring high purity and consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminomethyl imidazo[1,2-a]pyridine derivatives has relied heavily on oxidative coupling strategies that impose severe limitations on process scalability and economic feasibility. Previous literature, as summarized in the background of the patent, describes methods requiring transition metal catalysts such as vanadium acetate or expensive hypervalent iodine reagents like PhI(OAc)2, which significantly inflate raw material costs. These conventional routes often necessitate high reaction temperatures ranging from 90°C to 130°C and the use of hazardous solvents such as dioxane or DMSO, creating substantial energy burdens and safety risks during commercial scale-up of complex pharmaceutical intermediates. Additionally, the requirement for excessive stoichiometric amounts of oxidants and amine sources leads to poor atom economy and generates large volumes of chemical waste that require costly disposal procedures.
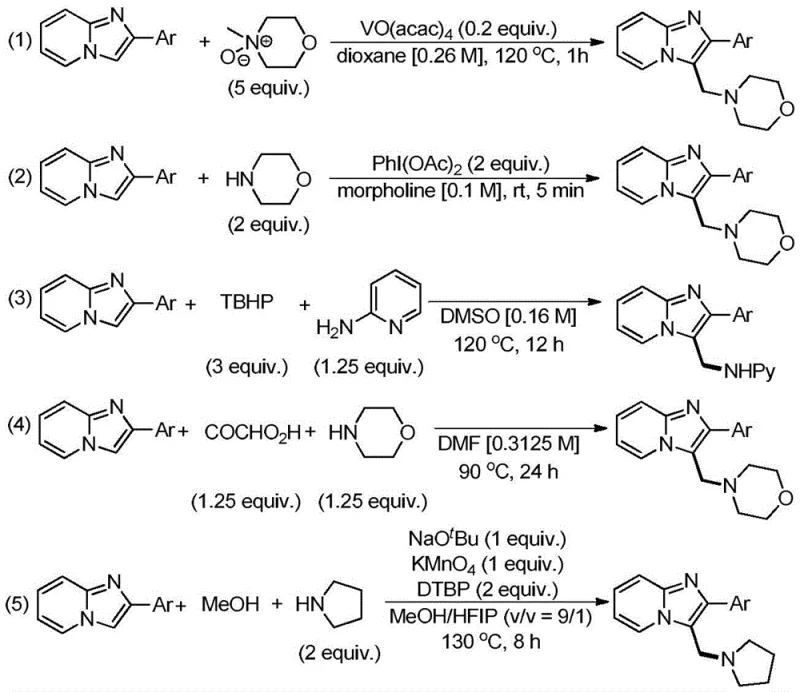
Furthermore, the reliance on pre-functionalized imidazo[1,2-a]pyridine substrates in many traditional approaches restricts the structural diversity accessible to researchers and increases the step count in the overall synthesis. The need for rigorous purification to remove trace metal contaminants is another critical bottleneck, especially for pharmaceutical applications where strict limits on heavy metals are enforced. These cumulative factors result in prolonged lead times, higher production costs, and a larger environmental footprint, making conventional methods less attractive for high-volume manufacturing. The industry urgently requires a paradigm shift towards more direct, efficient, and cleaner synthetic technologies that can overcome these inherent inefficiencies without compromising on yield or product quality.
The Novel Approach
The methodology disclosed in CN113372346B fundamentally redefines the synthetic landscape by employing a direct four-component condensation strategy that operates under exceptionally mild conditions. Instead of relying on pre-formed heterocycles or external oxidants, this novel approach utilizes readily available 2-aminopyridine and alpha-bromoaryl ethyl ketones to generate the imidazo[1,2-a]pyridine core in situ. The reaction proceeds efficiently at room temperature, typically completing within one hour, which drastically reduces energy consumption and thermal stress on sensitive functional groups. By eliminating the need for metal catalysts and strong bases, the process simplifies the workup procedure, allowing for easier isolation of the target product and reducing the risk of metal contamination in the final API.
This streamlined protocol demonstrates excellent functional group tolerance, accommodating various substituents on the aryl ring including electron-donating methyl and methoxy groups as well as electron-withdrawing halogens and trifluoromethyl groups. The use of ethanol as a preferred solvent further enhances the green profile of the reaction, replacing toxic chlorinated or polar aprotic solvents commonly found in legacy processes. The high yields observed across a broad substrate scope, often exceeding 80-90%, indicate a robust and reliable process suitable for industrial adoption. This novel approach not only lowers the barrier to entry for synthesizing these valuable scaffolds but also provides a sustainable pathway for the commercial production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Multicomponent One-Pot Cyclization
Understanding the underlying reaction mechanism is crucial for optimizing process parameters and ensuring consistent quality during scale-up. The proposed mechanism involves a cascade of transformations initiated by the nucleophilic attack of 2-aminopyridine on the alpha-bromoaryl ethyl ketone, leading to the formation of a 2-phenylimidazo[1,2-a]pyridine intermediate (Intermediate A). Concurrently, the secondary amine reacts with dichloromethane to generate a chloromethyldiethylamine species (Intermediate B), which serves as the aminomethylating agent. This dual activation strategy allows for the simultaneous construction of the heterocyclic core and the installation of the aminomethyl side chain in a single operational step, maximizing efficiency and minimizing intermediate isolation.
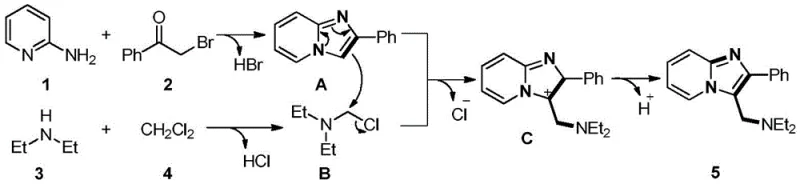
The subsequent nucleophilic substitution between Intermediate A and Intermediate B forms the C-N bond, followed by a dehydroaromatization step that yields the thermodynamically stable final product. This mechanistic pathway explains the high atom economy observed, as the only byproducts are typically inorganic salts or small molecules that are easily removed. From an impurity control perspective, the stoichiometric balance of the four components is critical; the patent data suggests that equimolar ratios are sufficient to drive the reaction to completion without the need for excess reagents. This precise control minimizes the formation of side products related to over-alkylation or polymerization, ensuring a clean reaction profile that simplifies downstream purification. The absence of radical initiators or strong oxidants further reduces the potential for oxidative degradation of the product, contributing to the high purity specifications achievable with this method.
How to Synthesize 3-Aliphatic Aminomethyl Imidazo[1,2-a]pyridine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent quality and mixing efficiency to replicate the high yields reported in the patent examples. The general procedure involves combining the four key components in a suitable reaction vessel, with ethanol identified as the optimal solvent for balancing solubility and environmental safety. Maintaining the reaction temperature at ambient conditions is sufficient, though slight variations between 20°C and 60°C do not significantly impact the outcome, providing operational flexibility. The reaction progress can be conveniently monitored using thin-layer chromatography (TLC), with typical completion times around one hour, allowing for rapid turnover in process development workflows.
- Combine 2-aminopyridine, alpha-bromoaryl ethyl ketone, secondary amine compound, and dichloromethane in an equimolar ratio within a reaction vessel.
- Add ethanol as the preferred green solvent to achieve a substrate concentration of approximately 1 mol/L.
- Stir the mixture at room temperature for 1 hour, monitor via TLC, and purify the residue by column chromatography after solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible benefits that extend beyond mere chemical efficiency. The elimination of expensive transition metal catalysts and specialized oxidants directly translates to substantial cost savings in raw material procurement, reducing the overall cost of goods sold (COGS) for the final intermediate. Since the starting materials such as 2-aminopyridine and alpha-bromoacetophenone are commodity chemicals available from multiple global suppliers, the risk of supply chain disruption is significantly minimized compared to routes relying on proprietary or scarce reagents. This diversification of the supply base enhances supply chain reliability and provides leverage in price negotiations with vendors.
- Cost Reduction in Manufacturing: The removal of metal catalysts eliminates the need for expensive scavenging resins or complex filtration steps required to meet strict residual metal limits in pharmaceutical products. This simplification of the downstream processing workflow reduces both the time and consumables associated with purification, leading to lower operational expenditures. Furthermore, the high atom utilization rate means less waste is generated per kilogram of product, lowering waste disposal costs and improving the overall sustainability metrics of the manufacturing site.
- Enhanced Supply Chain Reliability: By utilizing widely available commodity chemicals and avoiding reagents with long lead times or geopolitical supply risks, manufacturers can maintain consistent production schedules. The robustness of the reaction conditions, which tolerate minor fluctuations in temperature and stoichiometry, ensures high batch-to-batch consistency, reducing the rate of failed batches and associated losses. This reliability is critical for meeting the just-in-time delivery requirements of major pharmaceutical clients and maintaining long-term contractual agreements.
- Scalability and Environmental Compliance: The use of ethanol as a primary solvent aligns with green chemistry principles and facilitates easier regulatory approval for manufacturing processes in regions with strict environmental regulations. The exothermic nature of the reaction is manageable at room temperature, reducing the cooling load and energy infrastructure requirements for large-scale reactors. This ease of scale-up allows for seamless transition from kilogram-level development to multi-ton commercial production without the need for significant process re-engineering or capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details helps stakeholders make informed decisions regarding technology transfer and process integration.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive transition metal catalysts, oxidants, and high-temperature conditions, operating efficiently at room temperature with high atom economy.
Q: Is the process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of common solvents like ethanol, mild reaction conditions, and simple workup procedures makes this route highly suitable for large-scale commercial manufacturing.
Q: What is the substrate compatibility for the aryl group in this reaction?
A: The reaction tolerates a wide range of substituents including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups, maintaining yields generally above 70%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazo[1,2-a]pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into viable manufacturing processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement metal-free, room-temperature syntheses like the one described in CN113372346B demonstrates our dedication to innovation and sustainability in chemical manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
