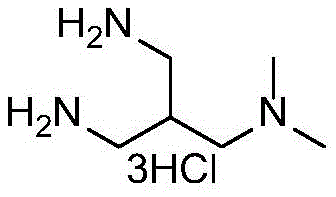
Scalable Manufacturing of 2-(Aminomethyl)-N1,N1-dimethylpropane-1,3-diamine Trihydrochloride for Advanced Pharmaceutical Applications
The pharmaceutical industry's relentless pursuit of efficient pathways for complex biological modulators has highlighted the critical importance of robust intermediate synthesis. Patent CN111548275A introduces a groundbreaking methodology for producing 2-(aminomethyl)-N1,N1-dimethylpropane-1,3-diamine trihydrochloride, a pivotal building block for the synthesis of AP20187. This molecule serves as a cell-permeable dimerizer for FK506 binding proteins (FKBPs), playing an essential role in initiating biological signaling cascades and gene expression studies. The disclosed technology represents a significant leap forward, transitioning from failed laboratory curiosities to a viable, high-yield industrial process that addresses long-standing purity and scalability challenges faced by R&D teams globally.
Understanding the limitations of historical approaches is vital for appreciating the technical merit of this new invention. Previous attempts to synthesize this scaffold relied heavily on malononitrile condensation followed by hydrogenation. However, this legacy pathway was plagued by catastrophic inefficiencies; the hydrogenation step frequently resulted in yields below 5%, often producing no detectable product at all. Furthermore, the free base generated in these failed attempts possessed extremely high water solubility, rendering standard purification techniques ineffective and necessitating cumbersome protecting group manipulations that added cost and complexity without guaranteeing success.
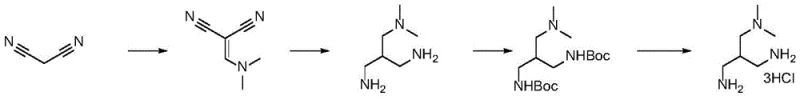
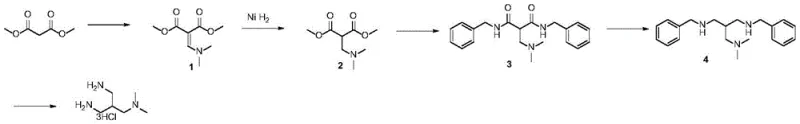
In stark contrast, the novel approach detailed in the patent utilizes dimethyl malonate as a starting material, engaging it in a condensation reaction with DMF-DMA to form a stable enamine intermediate. This strategic shift allows for a controlled, stepwise construction of the carbon backbone. The subsequent nickel-catalyzed hydrogenation proceeds with high efficiency, avoiding the pitfalls of the nitrile reduction. By employing benzylamine for aminolysis, the process introduces temporary protecting groups that facilitate purification, which are later cleanly removed via hydrogenolysis. This logical sequence of functional group transformations ensures high purity and reproducibility, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into the Catalytic Hydrogenation and Reductive Sequence
The core of this synthesis lies in the precise control of reduction potentials and nucleophilic substitutions. The initial formation of the enamine via DMF-DMA activation creates an electron-rich system that is highly susceptible to catalytic hydrogenation. When subjected to Raney Nickel under hydrogen pressure at 60-80°C, the double bond is selectively reduced while preserving the ester functionality, yielding the crucial amino-ester intermediate. This step is mechanistically superior to nitrile reduction because it avoids the formation of secondary amine byproducts and over-reduction issues that typically plague nitrile chemistry, thereby ensuring a cleaner reaction profile.
Following the establishment of the amine backbone, the process employs a powerful reduction strategy using Lithium Aluminum Hydride (LiAlH4). This reagent effectively reduces the amide bonds formed during the benzylamine exchange step directly to amines. The mechanism involves the nucleophilic attack of hydride ions on the carbonyl carbon, followed by the elimination of the oxygen species to form the C-N bond. This transformation is critical for generating the triamine core. Finally, the removal of the benzyl protecting groups via Pd/C catalyzed hydrogenolysis in an acidic medium not only deprotects the amines but simultaneously forms the stable trihydrochloride salt, streamlining the isolation process and enhancing the final product's stability for storage and transport.
How to Synthesize 2-(Aminomethyl)-N1,N1-dimethylpropane-1,3-diamine Trihydrochloride Efficiently
The synthesis protocol outlined in the patent offers a clear, reproducible roadmap for manufacturing this complex intermediate. It begins with the activation of dimethyl malonate and proceeds through a series of well-defined chemical transformations including hydrogenation, aminolysis, and hydride reduction. Each step has been optimized for temperature and solvent conditions to maximize yield and minimize impurity formation. For R&D teams looking to implement this process, the detailed standardized synthetic steps provided in the guide below offer a comprehensive framework for immediate adoption and scale-up.
- Condense dimethyl malonate with DMF-DMA at 40-55°C to form the enamine intermediate.
- Perform catalytic hydrogenation using Raney Nickel at 60-80°C to reduce the double bond and generate the amino ester.
- Execute aminolysis with benzylamine under reflux to introduce the benzyl protecting groups and form the diamide intermediate.
- Reduce the diamide intermediate using Lithium Aluminum Hydride (LiAlH4) in THF to obtain the protected triamine.
- Conduct final hydrogenolysis with Pd/C in acidic methanol to remove benzyl groups and form the trihydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to this dimethyl malonate-based route offers substantial strategic benefits. The reliance on commodity chemicals such as dimethyl malonate, DMF-DMA, and benzylamine eliminates the dependency on specialized, high-cost precursors that often bottleneck supply chains. This accessibility of raw materials ensures that production can be ramped up quickly without facing sourcing delays, providing a level of supply chain resilience that is critical for maintaining continuous manufacturing schedules in the fast-paced pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of the failed malononitrile route removes the massive waste associated with low-yield processes. By achieving high yields in each step, particularly the critical hydrogenation and reduction phases, the overall material throughput is significantly improved. Furthermore, the use of standard catalysts like Raney Nickel and Palladium on Carbon, which can often be recovered and recycled, drastically lowers the cost of goods sold compared to routes requiring exotic or single-use reagents.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality or environmental factors. This stability translates to fewer failed batches and a more predictable production timeline. For supply chain managers, this reliability is paramount, as it reduces the need for excessive safety stock and allows for leaner inventory management strategies while still meeting the rigorous demands of downstream API synthesis.
- Scalability and Environmental Compliance: The reactions described, such as hydrogenation and reflux, are unit operations that are well-understood and easily scaled from kilogram to tonne levels in standard chemical reactors. Additionally, the process avoids the generation of difficult-to-treat waste streams associated with nitrile hydrolysis or complex protection/deprotection cycles. This alignment with green chemistry principles simplifies waste treatment protocols and ensures compliance with increasingly stringent environmental regulations, facilitating smoother regulatory approvals for commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this intermediate. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on why this specific route is preferred for industrial applications over legacy methods.
Q: Why is the conventional malononitrile route considered unsuitable for industrial production?
A: The conventional route utilizing malononitrile suffers from critical failures during the hydrogenation step, where yields drop below 5% or fail completely. Additionally, the resulting free base exhibits excessive water solubility, making purification impossible without complex and costly protecting group strategies that ultimately lower overall efficiency.
Q: What are the key advantages of the dimethyl malonate pathway described in CN111548275A?
A: This novel pathway utilizes inexpensive, commodity-grade raw materials like dimethyl malonate and employs standard, stable reaction types such as catalytic hydrogenation and amide reduction. The process avoids the purification nightmares of the old route, offering significantly higher yields and a straightforward workflow suitable for large-scale manufacturing.
Q: How does this synthesis method impact supply chain reliability for AP20187 production?
A: By relying on common reagents and avoiding exotic catalysts or difficult-to-source precursors, this method ensures a stable supply chain. The robustness of the reaction conditions minimizes batch-to-batch variability, reducing the risk of production delays and ensuring consistent availability of this critical pharmaceutical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Aminomethyl)-N1,N1-dimethylpropane-1,3-diamine Trihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced therapeutics like AP20187 depends on the availability of high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 2-(aminomethyl)-N1,N1-dimethylpropane-1,3-diamine trihydrochloride meets the exacting standards required for clinical and research applications.
We invite you to collaborate with us to optimize your supply chain for this critical building block. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can drive efficiency and reliability in your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
