Advanced Synthesis of 2,2,6,6-Tetraethyl-3,5-Heptanedione for High-Performance Metal Complexes
Introduction to Novel Synthetic Pathways for Specialty Beta-Diketones
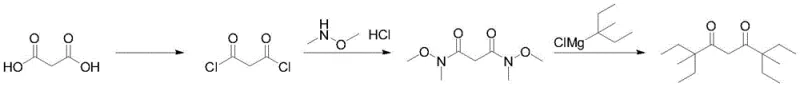
The chemical industry is constantly seeking more efficient routes to produce high-value intermediates that serve critical roles in advanced material science and analytical chemistry. A significant breakthrough in this domain is documented in patent CN112174788A, which details a robust preparation method for 2,2,6,6-tetraethyl-3,5-heptanedione. This specific beta-diketone is not merely a commodity chemical; it is a sophisticated ligand capable of forming stable bidentate complexes with metal ions such as europium and praseodymium, making it indispensable as an NMR shift reagent. Furthermore, its application extends to the analysis of aluminum, beryllium, and chromium in gas chromatography, as well as serving as a key component in organic electroluminescent devices. The traditional synthesis of such sterically hindered diketones has long been plagued by inefficiencies, but this new methodology offers a transformative solution by leveraging a three-step sequence starting from inexpensive malonic acid.
The core innovation lies in the strategic use of a Weinreb amide intermediate coupled with a Lewis acid-catalyzed Grignard reaction. This approach effectively bypasses the thermodynamic and kinetic barriers associated with bulky substituents. By converting malonic acid first into malonyl chloride and subsequently into N,N'-dimethoxy-N,N'-dimethylmalonamide, the process sets the stage for a highly controlled nucleophilic addition. The final step utilizes 3-methylpentyl magnesium chloride in the presence of a boron-based catalyst, ensuring high conversion rates and exceptional product purity. This route represents a paradigm shift for manufacturers looking to secure a reliable supply of high-purity pharmaceutical intermediates and electronic chemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-diketone compounds, particularly those with significant steric bulk like 2,2,6,6-tetraethyl-3,5-heptanedione, has relied heavily on the acylation reaction between ketones and esters under strong alkaline conditions. This classical Claisen condensation or related acylation pathways face severe challenges when applied to substrates with large ethyl groups at the alpha positions. The immense steric hindrance created by these four ethyl groups drastically impedes the approach of nucleophiles to the carbonyl carbon, leading to sluggish reaction kinetics and incomplete conversions. Consequently, these conventional methods typically suffer from disappointingly low synthesis yields, often necessitating complex and costly purification procedures to isolate the desired product from a mixture of unreacted starting materials and side products. The difficulty in purification not only drives up the cost of goods sold but also introduces variability in the quality of the final material, which is unacceptable for high-end applications like OLED manufacturing or precision analytical reagents.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data introduces a streamlined and chemically elegant solution that circumvents these steric issues entirely. By employing malonic acid as the foundational building block, the process initiates with a straightforward acyl chlorination to generate malonyl chloride, a highly reactive electrophile. This intermediate is then transformed into a Weinreb amide, a specialized functional group known for its ability to react with organometallic reagents to stop cleanly at the ketone stage, preventing the formation of tertiary alcohols which is a common pitfall in Grignard reactions. This strategic choice of intermediate ensures that the subsequent addition of the bulky 3-methylpentyl Grignard reagent proceeds with high fidelity. The result is a synthesis route that is not only operationally simple and safe but also delivers a product with a content of more than 99 percent, demonstrating a clear superiority over legacy technologies in terms of both efficiency and quality control.

Mechanistic Insights into Lewis Acid-Catalyzed Grignard Addition
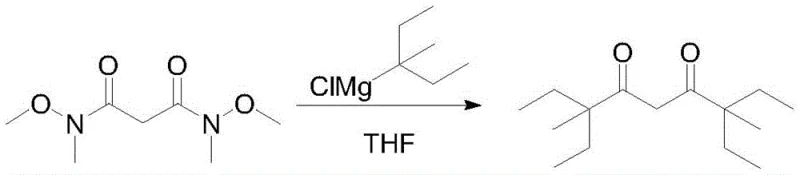
The crown jewel of this synthetic strategy is the final transformation step, where the N,N'-dimethoxy-N,N'-dimethylmalonamide reacts with the Grignard reagent. Under standard conditions, the reaction between a sterically hindered amide and a bulky Grignard reagent like 3-methylpentyl magnesium chloride would be exceptionally slow and inefficient. However, the introduction of a catalytic amount of tris(pentafluorophenyl)borane, B(C6F5)3, fundamentally alters the reaction landscape. This powerful Lewis acid interacts with the carbonyl oxygen of the Weinreb amide, increasing the electrophilicity of the carbonyl carbon and making it more susceptible to nucleophilic attack. Simultaneously, it is hypothesized that the boron species may coordinate with the Grignard reagent or the intermediate chelate, stabilizing the transition state and lowering the activation energy barrier. This dual activation mechanism allows the reaction to proceed rapidly even at low temperatures, typically between -20°C and -15°C, which helps in suppressing potential side reactions and maintaining the integrity of the sensitive intermediates.
The impact of this catalytic system on impurity control and yield is profound. Experimental data within the patent reveals that in the absence of the boron catalyst, the reaction requires a prolonged duration of 10 hours and still only achieves a mediocre yield of 57.2%. Conversely, with the inclusion of 0.05 to 0.3 equivalents of B(C6F5)3, the reaction time is slashed to merely 1 hour, and the yield surges to over 83%. This dramatic improvement is not just a matter of speed; it signifies a much cleaner reaction profile with fewer by-products. The high selectivity ensures that the final distillation yields a product with GC purity exceeding 99%, which is critical for its end-use as a shift reagent where trace impurities could interfere with NMR spectral resolution. This mechanistic understanding underscores the value of the process for producing high-purity electronic chemical precursors where material consistency is paramount.
How to Synthesize 2,2,6,6-Tetraethyl-3,5-Heptanedione Efficiently
Implementing this synthesis in a production environment requires careful attention to reaction conditions and reagent stoichiometry to maximize the benefits of the catalytic system. The process begins with the activation of malonic acid, followed by the formation of the Weinreb amide under strictly anhydrous conditions to prevent hydrolysis of the acid chloride. The final Grignard step demands precise temperature control and the accurate dosing of the boron catalyst to ensure the rapid and complete conversion of the starting material. For R&D teams looking to replicate or scale this chemistry, the following standardized steps outline the critical operational parameters derived from the patent examples, ensuring a robust transfer from laboratory bench to pilot plant.
- React malonic acid with thionyl chloride or oxalyl chloride in the presence of a catalyst like DMF or pyridine to generate malonyl chloride.
- Convert malonyl chloride into N,N'-dimethoxy-N,N'-dimethylmalonamide using dimethylhydroxylamine hydrochloride and an acid-binding agent such as triethylamine.
- Perform a Grignard reaction with 3-methylpentyl magnesium chloride in the presence of a boron catalyst B(C6F5)3 to yield the final diketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical advantages that extend far beyond simple yield improvements. The reliance on malonic acid as a starting material is a strategic masterstroke, as it is a widely available, commodity-grade chemical with a stable global supply chain, mitigating the risks associated with sourcing exotic or scarce precursors. Furthermore, the reagents used throughout the three steps, such as thionyl chloride, oxalyl chloride, and standard amines, are staple chemicals in the fine chemical industry, ensuring that raw material costs remain predictable and competitive. The elimination of complex transition metal catalysts, often based on precious metals like palladium or platinum which require expensive removal steps to meet residual metal specifications, further simplifies the downstream processing. This reduction in processing complexity translates directly into significant cost reduction in pharmaceutical intermediates manufacturing, as it lowers both the capital expenditure on specialized equipment and the operational expenditure on waste treatment and purification media.
- Cost Reduction in Manufacturing: The implementation of the B(C6F5)3 catalytic system drastically reduces reaction times from 10 hours to just 1 hour, which significantly increases the throughput capacity of existing reactor assets without the need for new capital investment. By avoiding the use of expensive transition metals and utilizing a highly efficient catalytic cycle, the process minimizes the consumption of high-cost reagents. Additionally, the high crude purity achieved (>99% by GC) implies that less solvent and energy are required for final purification via distillation, leading to substantial cost savings in utility consumption and waste disposal fees.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions, which operate at mild temperatures ranging from -30°C to 75°C depending on the step, reduces the risk of thermal runaways and safety incidents that could disrupt production schedules. The use of common solvents like dichloromethane, toluene, and tetrahydrofuran ensures that solvent supply is never a bottleneck, unlike processes requiring specialized or regulated solvents. This operational stability guarantees a consistent output of high-purity 2,2,6,6-tetraethyl-3,5-heptanedione, allowing downstream customers to maintain their own production schedules for NMR reagents or OLED materials without fear of supply interruptions.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been designed with industrial amplification in mind, moving seamlessly from gram-scale laboratory experiments to multi-kilogram production batches. The short reaction sequence (only three steps) minimizes the cumulative loss of material at each stage, maximizing the overall atom economy of the process. Moreover, the avoidance of heavy metal contaminants simplifies the environmental compliance landscape, as the effluent streams do not require rigorous treatment for toxic metal removal, thereby reducing the environmental footprint and aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this material into their supply chains, we have compiled answers to common inquiries based on the detailed technical disclosures in the patent literature. These responses address critical aspects regarding the chemical stability, the specific role of the catalyst, and the purity profiles achievable through this method. Understanding these nuances is essential for R&D directors who need to validate the material for sensitive applications such as gas chromatography analysis or as a dopant in optoelectronic devices.
Q: Why is the Weinreb amide route preferred for synthesizing bulky beta-diketones?
A: Traditional acylation methods often suffer from low yields due to severe steric hindrance. The Weinreb amide strategy prevents over-addition of the Grignard reagent, ensuring the formation of the ketone rather than a tertiary alcohol, resulting in purities exceeding 99%.
Q: What is the function of B(C6F5)3 in this synthesis?
A: B(C6F5)3 acts as a potent Lewis acid catalyst. It activates the carbonyl group of the Weinreb amide and coordinates with the Grignard reagent, significantly accelerating the reaction rate from 10 hours to 1 hour and improving yield from 57.2% to over 83%.
Q: What are the primary applications of 2,2,6,6-tetraethyl-3,5-heptanedione?
A: This compound serves as a bidentate ligand forming stable complexes with metals like Europium and Praseodymium for use as NMR shift reagents. It is also utilized in organic electroluminescent devices (OLEDs) and for analyzing specific metals in gas chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2,6,6-Tetraethyl-3,5-Heptanedione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-performance ligands in advancing analytical and electronic technologies. Our team of expert chemists has thoroughly analyzed the synthetic route described in CN112174788A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to the market. We are committed to delivering 2,2,6,6-tetraethyl-3,5-heptanedione with stringent purity specifications, ensuring that every batch meets the rigorous demands of NMR spectroscopy and OLED fabrication. Our state-of-the-art rigorous QC labs are equipped to verify the absence of trace impurities and residual metals, providing our partners with the confidence they need to rely on our materials for their most sensitive applications.
We invite global pharmaceutical and electronic material companies to collaborate with us to optimize their supply chains for this valuable intermediate. By leveraging our expertise in process chemistry and scale-up engineering, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project timelines are met with the highest quality standards and commercial reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
