Scalable One-Pot Synthesis of Indolyl-Substituted Indol-3-One Intermediates for Global Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex nitrogen-containing heterocycles, particularly those found in bioactive natural products. Patent CN111925312B introduces a significant breakthrough in the synthesis of indolyl-substituted indol-3-one derivatives, a core scaffold prevalent in potent alkaloids such as Halichrome A, LipidGreen, and Cephalinone. These molecules are not merely academic curiosities; they represent critical building blocks for next-generation therapeutic agents targeting various oncological and neurological pathways. The disclosed technology leverages a novel reductive cyclization strategy that bypasses the multi-step, low-yielding sequences traditionally associated with constructing the 2,2-disubstituted indol-3-one core. By utilizing triphenylphosphine (PPh3) as a mediating reducing agent under an inert argon atmosphere, this method achieves high conversion rates while maintaining exceptional structural integrity of the sensitive indole moiety.
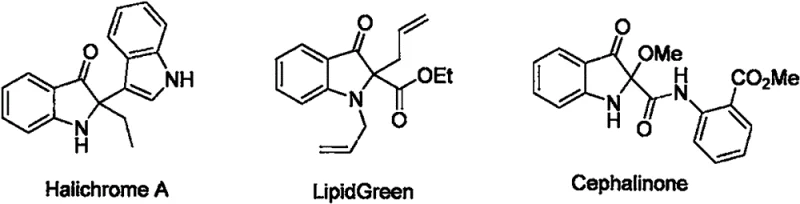
For R&D directors evaluating new synthetic routes, the stability and purity of the final intermediate are paramount. The invention details a general formula (Formula I) where diverse substituents (R1 through R4) can be introduced without compromising the reaction efficiency. This flexibility allows medicinal chemists to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. The ability to tolerate functional groups such as halogens, cyano, nitro, esters, and aldehydes on the aromatic rings expands the chemical space available for drug discovery teams. Furthermore, the reaction proceeds with high regioselectivity, minimizing the formation of difficult-to-remove isomers that often plague indole functionalization chemistry. This level of control is essential for ensuring that downstream processing remains efficient and that the final active pharmaceutical ingredient (API) meets stringent regulatory purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indol-3-one skeleton has been a formidable challenge in organic synthesis, often requiring harsh conditions that limit substrate scope. Traditional approaches frequently rely on strong oxidizing agents or precious metal catalysts like palladium or rhodium, which introduce significant cost burdens and supply chain vulnerabilities. These conventional methods often necessitate multiple protection and deprotection steps to prevent side reactions at the indole nitrogen or the reactive C3 position, leading to atom inefficiency and substantial waste generation. Moreover, the use of transition metals creates a critical bottleneck in purification; removing trace metal residues to meet ppm-level specifications for pharmaceutical use requires additional scavenging steps, chromatography, or recrystallization, all of which erode overall yield and extend production timelines. The complexity of these legacy routes makes them ill-suited for the rapid, cost-sensitive demands of modern commercial manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN111925312B offers a streamlined, one-pot solution that elegantly merges indole compounds with o-nitroalkyne compounds. The core innovation lies in the use of triphenylphosphine (PPh3) acting as an oxygen acceptor to facilitate the reductive cyclization, effectively converting the nitro group into the necessary functionality for ring closure without external hydrogen sources. This reaction operates under mild thermal conditions, typically between 50°C and 120°C, with a preferred operating temperature of 90°C, which significantly reduces energy consumption compared to high-temperature pyrolysis methods. The process is conducted in o-dichlorobenzene, a high-boiling solvent that ensures good solubility for the diverse range of substrates while remaining stable under the reaction conditions. By eliminating the need for transition metals and simplifying the workup to a direct purification step, this approach drastically reduces the operational complexity and equipment footprint required for production.
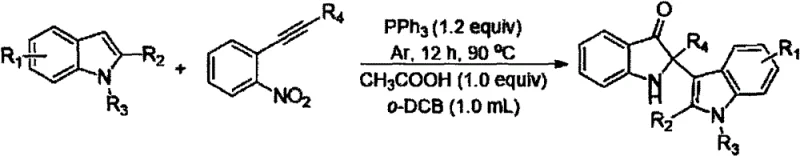
Mechanistic Insights into PPh3-Mediated Reductive Cyclization
The mechanistic pathway of this transformation is a testament to the power of organophosphorus chemistry in modern synthesis. The reaction initiates with the interaction between the o-nitroalkyne and triphenylphosphine, where the phosphine acts as a nucleophile towards the nitro group, eventually leading to the formation of a nitrene or similar reactive intermediate after oxygen transfer to form triphenylphosphine oxide. This activated species then undergoes an intramolecular or intermolecular cyclization with the electron-rich indole nucleus. The presence of an acid additive, such as acetic acid, trifluoroacetic acid, or phosphoric acid, plays a crucial catalytic role by protonating intermediates and facilitating the elimination of water or other small molecules to aromatize the final system. The acid also helps in activating the carbonyl or imine precursors formed during the cascade, ensuring the reaction proceeds to completion with high fidelity. Understanding this mechanism allows process chemists to fine-tune the acid strength and stoichiometry to optimize rates for specific substrate combinations.
From an impurity control perspective, the mechanism inherently favors the formation of the thermodynamically stable 2,2-disubstituted indol-3-one product. The use of PPh3 avoids the radical pathways often associated with metal-catalyzed reductions, which can lead to polymerization or indiscriminate reduction of other sensitive functional groups on the molecule. The patent data indicates that even with sterically demanding groups like cyclopropyl or bulky aryl substituents on the alkyne, the reaction maintains high conversion efficiency, often exceeding 80-90% in optimized examples. This suggests that the transition state is accessible and that steric hindrance is effectively managed by the reaction conditions. For quality assurance teams, this predictability translates to a consistent impurity profile, making validation and regulatory filing significantly more straightforward compared to stochastic metal-catalyzed processes.
How to Synthesize Indolyl-Substituted Indol-3-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The protocol involves a straightforward mixing of reagents followed by a controlled heating phase under an inert atmosphere. It is critical to ensure that the argon purge is thorough to prevent oxidation of the phosphine reagent prior to the reaction start. The molar ratios are flexible but generally favor a slight excess of the o-nitroalkyne and the reducing agent to drive the equilibrium forward. Detailed standard operating procedures regarding exact stoichiometry, addition rates, and purification techniques are essential for reproducibility.
- Mix indole compounds, acid (e.g., acetic acid), triphenylphosphine (PPh3), and o-dichlorobenzene solvent in a reaction vessel.
- Purge the vessel with argon three times, add o-nitroalkyne compounds, and heat the mixture to 50-120°C for 8-16 hours.
- Upon completion, purify the reaction mixture to isolate the target indolyl-substituted indol-3-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from metal-catalyzed to organophosphorus-mediated chemistry represents a strategic opportunity to de-risk the supply of critical intermediates. The reliance on triphenylphosphine, a commodity chemical with a stable global supply chain, eliminates exposure to the volatile pricing and geopolitical constraints often associated with precious metals like palladium or platinum. Furthermore, the simplification of the downstream processing—specifically the removal of metal scavenging steps—translates directly into reduced manufacturing cycle times and lower operational expenditures. The robustness of the reaction across a wide range of substrates means that a single production line can be utilized for multiple campaigns with minimal changeover cleaning, enhancing asset utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a significant line item from the bill of materials. Additionally, the byproduct of the reduction, triphenylphosphine oxide, is often easier to separate than metal complexes, potentially allowing for solvent recovery and recycling strategies that further lower the cost of goods sold. The one-pot nature of the synthesis consolidates what were previously multiple unit operations into a single reactor charge, reducing labor costs and energy consumption associated with heating, cooling, and transferring materials between vessels.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as substituted indoles and o-nitroalkynes, manufacturers can source raw materials from multiple qualified vendors, preventing single-source bottlenecks. The mild reaction conditions reduce the stress on reactor equipment, lowering maintenance frequency and extending the lifespan of capital assets. This reliability ensures consistent delivery schedules to downstream API manufacturers, which is critical for maintaining uninterrupted drug production lines.
- Scalability and Environmental Compliance: The process generates less hazardous waste compared to traditional methods that utilize heavy metals or strong oxidizers. The absence of metal residues simplifies wastewater treatment protocols and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly stringent environmental regulations, future-proofing the production facility against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What is the primary advantage of this synthesis method over traditional routes?
A: The primary advantage is the one-pot nature of the reaction which utilizes cheap, non-toxic reagents like triphenylphosphine instead of expensive transition metal catalysts, significantly simplifying purification and reducing environmental impact.
Q: What represents the substrate scope for this indol-3-one synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents on the indole ring (such as halogens, esters, and aldehydes) and diverse groups on the alkyne moiety (including cyclopropyl, aryl, and heteroaryl groups).
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses mild reaction conditions (50-120°C), common solvents like o-dichlorobenzene, and avoids sensitive metal catalysts, making it highly robust and easily scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolyl-Substituted Indol-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this PPh3-mediated synthesis route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of indolyl-substituted indol-3-one meets the exacting standards required for clinical and commercial applications. We understand that consistency is key in the pharma supply chain, and our process engineering team is ready to optimize this specific chemistry for your unique volume requirements.
We invite you to collaborate with us to leverage this cost-effective and robust technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how we can accelerate your timeline to market while optimizing your overall production budget.
