Scalable Production of Dabigatran Etexilate Mesylate: A Safer, High-Purity Synthetic Route
Introduction to Advanced Anticoagulant Manufacturing
The global demand for novel oral anticoagulants continues to surge, driven by an aging population and the increasing prevalence of atrial fibrillation. At the forefront of this therapeutic class is Dabigatran Etexilate Mesylate, a direct thrombin inhibitor that has revolutionized stroke prevention. However, the commercial viability of this critical medication relies heavily on the robustness and safety of its manufacturing process. Patent CN108727334B discloses a groundbreaking production process that addresses historical bottlenecks in synthesis, offering a pathway to higher purity and improved supply chain stability. This innovative methodology replaces hazardous high-pressure hydrogenation steps with mild, selective acylation and sophisticated crystallization techniques.
The chemical architecture of Dabigatran Etexilate Mesylate is complex, featuring multiple functional groups including benzimidazole, amidine, and carbamate moieties that require precise handling to prevent degradation. As illustrated in the structural diagram below, the molecule demands a synthesis strategy that maintains integrity across these sensitive regions while ensuring the final salt form meets stringent pharmacopeial standards for moisture and impurity profiles.
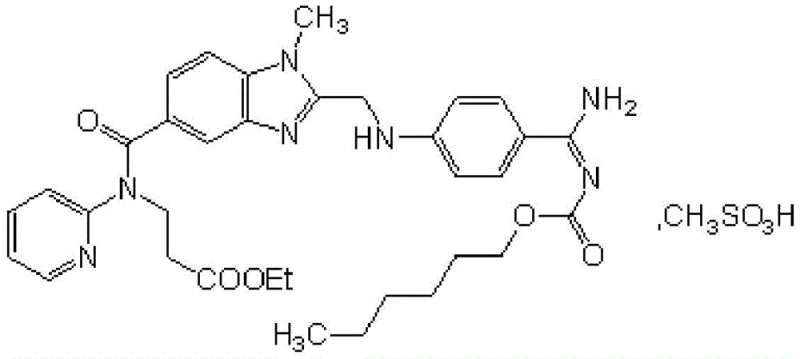
For procurement leaders and R&D directors, the shift towards this refined process represents a significant opportunity. By mitigating the risks associated with heavy metal catalysts and corrosive gases, manufacturers can achieve a more consistent product quality. This report analyzes the technical merits of this patent, demonstrating how it serves as a blueprint for becoming a reliable pharmaceutical intermediate supplier capable of delivering high-purity anticoagulants at a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Dabigatran Etexilate has been plagued by operational hazards and scalability issues that hinder cost-effective manufacturing. Early routes, such as those reported by Boehringer Ingelheim, often relied on palladium-carbon catalyzed high-pressure hydrogenation. While effective in a laboratory setting, this step introduces significant risks in a plant environment, including the potential for catalyst leakage, expensive metal residue removal processes, and the inherent dangers of high-pressure hydrogen gas. Furthermore, alternative literature methods frequently utilized thionyl chloride and required the introduction of dry hydrogen chloride gas for amidine formation.
These conventional approaches create severe pain points for supply chain heads. The use of thionyl chloride and gaseous HCl leads to serious equipment corrosion, necessitating specialized, costly reactor linings and frequent maintenance downtime. Additionally, many legacy processes depend on column chromatography for purification. In the context of industrial chemistry, column chromatography is a major bottleneck; it is solvent-intensive, difficult to automate, and practically impossible to scale to multi-ton quantities without exorbitant costs. These factors combined result in a fragile supply chain with high environmental waste discharge and inconsistent batch-to-batch quality.
The Novel Approach
The process detailed in patent CN108727334B offers a transformative solution by fundamentally redesigning the synthetic trajectory to prioritize safety and scalability. Instead of relying on reduction reactions, this novel approach utilizes a strategic sequence of activation, cyclization, and controlled acylation. A key differentiator is the use of CDI (N,N'-carbonyldiimidazole) for activating the starting materials, which allows for mild reaction conditions that preserve the integrity of the molecular scaffold. The elimination of the palladium-carbon hydrogenation step not only removes a major safety hazard but also simplifies the downstream purification workflow significantly.
Moreover, the new route introduces a sophisticated purification protocol that completely bypasses the need for column chromatography. By leveraging specific solvent systems and salification techniques—such as using an organic solution containing oxalic acid dihydrate—the process achieves high selectivity through crystallization alone. This shift from chromatographic separation to crystallization-based purification is a hallmark of mature industrial chemistry. It ensures that the production of Dabigatran Etexilate Mesylate can be scaled up efficiently, reducing the environmental footprint through lower solvent consumption and minimizing the generation of hazardous waste streams associated with traditional methods.
Mechanistic Insights into Controlled Acylation and Crystallization
The core technical breakthrough of this synthesis lies in the meticulous control of reaction kinetics and thermodynamic equilibrium during the formation of the carbamate linkage and the subsequent purification. In Step 3, the conversion of intermediate PR-II to the crude Dabigatran Etexilate (PR-III) involves reacting with n-hexyl chloroformate. The patent specifies a critical operational parameter: the batched addition of the chloroformate reagent. Unlike a one-shot addition which can lead to localized exotherms and the formation of bis-acylated by-products or hydrolysis products, the batched addition method maintains a steady concentration of the electrophile. This kinetic control ensures that the nucleophilic attack by the amidine nitrogen occurs selectively, maximizing yield and minimizing the generation of structurally related impurities that are difficult to remove later.
Furthermore, the choice of solvent system plays a pivotal role in the reaction's success. The use of a mixed system, such as tetrahydrofuran/potassium carbonate/water or dichloromethane/triethylamine, creates a microenvironment that facilitates the reaction while stabilizing the intermediates. The presence of a weak base like potassium carbonate helps to scavenge the HCl generated during acylation without being strong enough to degrade the sensitive amidine group. This delicate balance is essential for maintaining the high purity required for pharmaceutical applications.
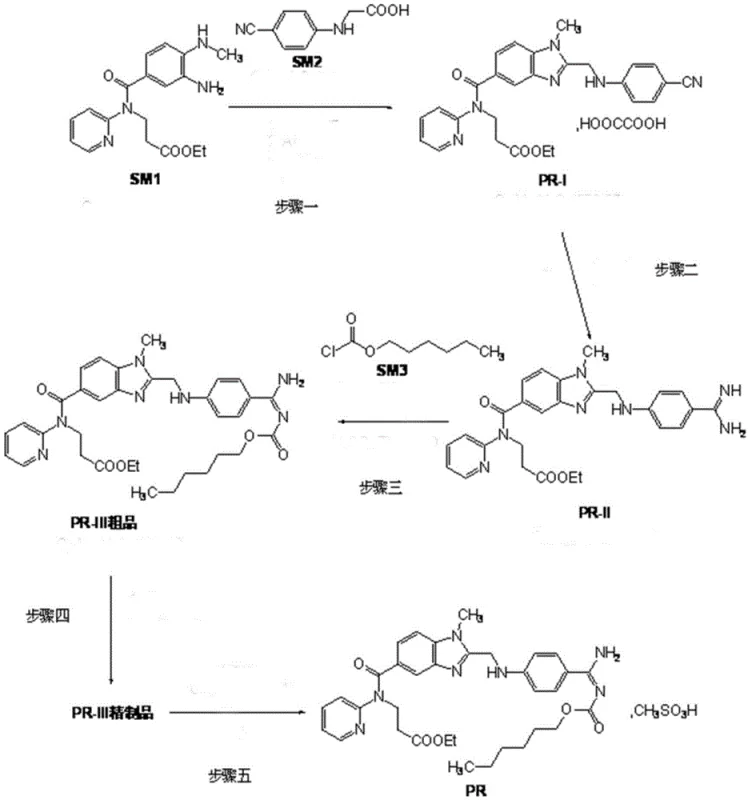
Equally important is the refining mechanism described in Step 4, which directly addresses the issue of moisture content—a critical quality attribute for this hygroscopic API. The process employs a dual-solvent recrystallization strategy. First, the crude product is treated with acetone and water. This step is designed to remove small polar impurities and initial moisture. However, since the final product has poor stability in aqueous solvents, a second recrystallization using ethyl acetate is performed. Ethyl acetate acts as a superior solvent for removing non-polar impurities while allowing the product to crystallize in a lattice structure that traps minimal water. This synergistic purification ensures that the maximum single impurity is controlled to below 0.06% and moisture content remains under 0.2%, meeting the rigorous specifications demanded by regulatory bodies.
How to Synthesize Dabigatran Etexilate Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding temperature, solvent ratios, and addition rates to ensure reproducibility at scale. The process is divided into five distinct stages, beginning with the activation of raw materials and concluding with the final salt formation. Each step has been engineered to maximize throughput while minimizing the formation of side products. For R&D teams looking to replicate or license this technology, understanding the specific mass ratios and thermal profiles is essential for successful technology transfer.
- Activation and Cyclization: React SM1 and SM2 using CDI activation followed by acetic acid cyclization and oxalic acid salification to obtain intermediate PR-I.
- Amidination: Convert PR-I to intermediate PR-II via Pinner reaction with HCl/ethanol followed by ammoniation, avoiding hazardous gaseous HCl handling.
- Controlled Acylation & Refining: React PR-II with n-hexyl chloroformate using batched addition in a mixed solvent system, followed by sequential acetone/water and ethyl acetate recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages beyond mere chemical yield. The primary value driver is the drastic simplification of the manufacturing infrastructure. By eliminating the requirement for high-pressure hydrogenation reactors and specialized corrosion-resistant equipment for handling gaseous HCl, the capital expenditure (CAPEX) required for production facilities is significantly reduced. This allows for more flexible manufacturing arrangements and lowers the barrier to entry for secondary suppliers, thereby enhancing market competition and potentially lowering costs for buyers.
- Cost Reduction in Manufacturing: The removal of palladium-carbon catalysts eliminates the need for expensive heavy metal scavenging resins and the associated validation testing for residual metals. Furthermore, replacing column chromatography with crystallization drastically reduces solvent consumption and waste disposal costs. The batched addition of reagents improves atom economy by preventing over-reaction, ensuring that raw materials are converted into saleable product rather than waste, leading to substantial cost savings in raw material procurement.
- Enhanced Supply Chain Reliability: Traditional routes often suffer from long lead times due to complex purification steps and equipment maintenance caused by corrosion. This new process utilizes mild conditions and standard stainless steel equipment, which reduces unplanned downtime and maintenance intervals. The robustness of the crystallization-based purification ensures consistent batch quality, reducing the risk of failed batches that could disrupt the supply of this critical anticoagulant to the global market.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The absence of chromatographic purification means the process can be scaled from kilograms to tons without a linear increase in operational complexity. Additionally, the reduction in hazardous waste discharge and the avoidance of toxic reagents align with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising output.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Dabigatran Etexilate Mesylate using this advanced methodology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on quality control and process safety.
Q: How does this process improve safety compared to traditional Dabigatran synthesis?
A: This process eliminates the need for palladium-carbon high-pressure hydrogenation and avoids the use of large amounts of corrosive hydrogen chloride gas, significantly reducing equipment corrosion risks and operational hazards.
Q: What is the key to controlling moisture content in the final API?
A: The process utilizes a specific dual-step refining strategy for intermediate PR-III, employing acetone/water crystallization followed by ethyl acetate recrystallization, which effectively controls moisture to below 0.2%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route is designed for industrial scalability by removing the need for column chromatography purification and utilizing mild reaction conditions that are easily managed in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Mesylate Supplier
The technical superiority of this synthesis route underscores the importance of partnering with a manufacturer who possesses both the intellectual property understanding and the engineering capability to execute it flawlessly. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary infrastructure to handle the specific solvent systems and crystallization protocols required to maintain the stringent purity specifications and low moisture content defined in this patent.
We invite global pharmaceutical partners to collaborate with us to secure a stable supply of high-quality anticoagulant intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can optimize your supply chain for Dabigatran Etexilate Mesylate.
