Scalable Manufacturing of High-Purity 2-Amino-3-Cyano Benzopyran Derivatives for Global Pharma
Scalable Manufacturing of High-Purity 2-Amino-3-Cyano Benzopyran Derivatives for Global Pharma
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for safer, more efficient synthetic routes for critical pharmaceutical intermediates. Patent CN102424675B represents a significant technological leap in the preparation of 2-amino-3-cyano benzopyran derivatives, a structural motif frequently encountered in bioactive molecules targeting various therapeutic areas. This proprietary methodology addresses long-standing challenges in heterocyclic synthesis by replacing hazardous cyanating agents with benign alternatives while maintaining exceptional yield profiles. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial for optimizing supply chains and ensuring regulatory compliance in the production of complex fine chemicals. The innovation lies not just in the final product quality, but in the holistic design of the reaction pathway which prioritizes operational simplicity and environmental stewardship.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of cyano groups into organic frameworks has relied heavily on the use of stoichiometric amounts of toxic inorganic cyanides such as potassium cyanide (KCN) or sodium cyanide. These traditional protocols often necessitate rigorous safety measures, specialized containment infrastructure, and complex waste treatment procedures to neutralize residual cyanide, thereby inflating the operational expenditure significantly. Furthermore, alternative methods involving the dehydration of oximes or the conversion of carboxylic acids often suffer from harsh reaction conditions, requiring high temperatures or strong dehydrating agents that can compromise the integrity of sensitive functional groups elsewhere in the molecule. Such limitations create bottlenecks in commercial scale-up, where the handling of bulk toxic materials becomes a major liability for supply chain continuity and worker safety. Additionally, multi-step sequences with low atom economy result in substantial material waste, contradicting modern principles of green chemistry and sustainable manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN102424675B utilizes a strategic three-step cascade that circumvents the need for free cyanide salts entirely. The process initiates with a Knoevenagel-type condensation between an aromatic aldehyde and ethyl cyanoacetate, utilizing a dual catalyst system of potassium fluoride and proline in alcoholic solvents. This is followed by a cyclization with 1,3-dimedone derivatives to construct the benzopyran core, and finally, a transformative exchange reaction with malononitrile to install the critical 3-cyano functionality. 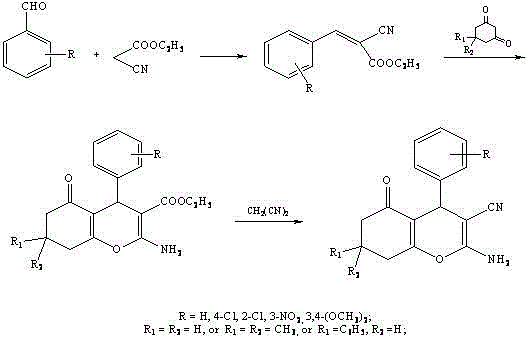 This novel route operates under remarkably mild thermal conditions, typically between 40°C and 80°C, which drastically reduces energy consumption compared to conventional high-temperature dehydrations. By leveraging malononitrile as a safe cyanide source, the process eliminates the regulatory burden associated with剧毒 materials, offering a streamlined path to high-purity intermediates suitable for sensitive pharmaceutical applications.
This novel route operates under remarkably mild thermal conditions, typically between 40°C and 80°C, which drastically reduces energy consumption compared to conventional high-temperature dehydrations. By leveraging malononitrile as a safe cyanide source, the process eliminates the regulatory burden associated with剧毒 materials, offering a streamlined path to high-purity intermediates suitable for sensitive pharmaceutical applications.
Mechanistic Insights into the Catalytic Cascade
The success of this synthesis hinges on the precise orchestration of base-catalyzed transformations that maximize selectivity while minimizing byproduct formation. In the initial step, the combination of potassium fluoride and proline acts as a highly efficient organocatalytic system. The fluoride ion serves to activate the carbonyl species or stabilize transition states, while proline likely facilitates the enolization of the cyanoacetate through hydrogen bonding networks, promoting the nucleophilic attack on the aldehyde. This synergistic effect ensures rapid conversion to the 2-amino-3-aryl acrylic acid ester intermediate with high stereochemical control, preventing polymerization or side reactions that often plague aldol-type condensations. The subsequent cyclization with dimedone derivatives proceeds via a Michael addition followed by intramolecular transesterification or lactonization, driven by weak organic bases like piperidine or pyridine which are sufficiently nucleophilic to initiate the reaction without causing decomposition of the sensitive cyano group.
The final and most critical mechanistic step involves the conversion of the ethyl ester moiety into the nitrile group using malononitrile. This transformation is essentially a nucleophilic acyl substitution where the active methylene of malononitrile attacks the ester carbonyl, followed by the elimination of ethanol and decarboxylation or fragmentation to yield the stable aromatic nitrile system. The use of malononitrile is particularly ingenious as it serves both as the carbon source for the nitrile and as a driving force for the equilibrium due to the stability of the resulting conjugated system. Impurity control is inherently built into this mechanism; the mild basic conditions prevent the hydrolysis of the newly formed nitrile group, a common issue in acidic or strongly basic traditional cyanation methods. Consequently, the crude product profile is exceptionally clean, reducing the burden on downstream purification processes such as column chromatography or recrystallization, which directly translates to higher recovery rates and lower solvent usage in a commercial setting.
How to Synthesize 2-Amino-3-Cyano Benzopyran Derivatives Efficiently
Implementing this synthesis requires careful attention to solvent selection and catalyst loading to replicate the high yields reported in the patent literature. The process is designed to be robust, tolerating a variety of substituents on the aromatic ring, including electron-withdrawing groups like chloro and nitro, as well as electron-donating methoxy groups. Operators should note that the reaction temperature window of 40°C to 80°C is critical; exceeding this range may lead to the degradation of the malononitrile or the formation of polymeric tars, while lower temperatures may result in incomplete conversion of the sterically hindered intermediates. The detailed standardized synthesis steps below outline the precise molar ratios and workup procedures required to achieve the benchmark 70-80% overall yield.
- Perform addition reaction between aromatic aldehyde and ethyl cyanoacetate using KF/Proline catalyst in alcohol solvent at 40-80°C.
- React the resulting 2-amino-3-aryl acrylic acid ester with 1,3-dimedone derivative using a weak organic base catalyst to form the chromene ester intermediate.
- Convert the ester intermediate to the final 2-amino-3-cyano derivative by reacting with malononitrile in solvent under catalytic conditions, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic benefits beyond mere technical feasibility. The elimination of hazardous cyanide salts from the bill of materials fundamentally alters the risk profile of the manufacturing process, removing the need for expensive hazmat shipping, specialized storage facilities, and complex emergency response protocols. This simplification of the safety infrastructure leads to a drastic reduction in overhead costs associated with regulatory compliance and insurance. Furthermore, the use of commodity solvents such as ethanol, methanol, and DMF ensures that raw material sourcing remains stable and cost-effective, insulating the supply chain from volatility associated with exotic or controlled reagents. The mild reaction conditions also imply lower energy intensity, contributing to a reduced carbon footprint and aligning with corporate sustainability goals which are increasingly important for multinational clients.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the high atom economy and the avoidance of expensive heavy metal catalysts or toxic reagents that require costly disposal. By utilizing inexpensive organic bases like piperidine and readily available malononitrile, the direct material cost per kilogram of product is significantly optimized. Additionally, the high purity of the crude product minimizes the number of recrystallization cycles needed, leading to substantial savings in solvent recovery and waste treatment expenses. The overall yield of 70-80% ensures that raw material utilization is maximized, reducing the effective cost of goods sold and improving margin potential for high-volume contracts.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions, which do not require anhydrous environments or inert gas protection to the same extent as organometallic couplings. This tolerance to ambient conditions simplifies the manufacturing workflow, reducing the likelihood of batch failures due to moisture ingress or equipment malfunction. The availability of starting materials like aromatic aldehydes and dimedone derivatives from multiple global suppliers prevents single-source bottlenecks, ensuring continuous production capability even during market disruptions. Shorter reaction times and simplified workups further accelerate the production cycle, allowing for faster turnaround times on customer orders and improved inventory turnover rates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot plant is straightforward due to the absence of exothermic hazards typically associated with cyanide additions. The liquid-phase homogeneous catalysis allows for efficient heat transfer and mixing in standard stainless steel reactors, facilitating seamless technology transfer. From an environmental perspective, the process generates significantly less hazardous waste, as the byproducts are primarily organic salts and alcohols that are easier to treat than heavy metal sludge or cyanide-containing effluents. This alignment with green chemistry principles simplifies the permitting process for new manufacturing lines and enhances the company's reputation as a responsible supplier in the global fine chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their broader drug substance manufacturing workflows.
Q: What are the primary safety advantages of this synthesis method compared to traditional cyanation?
A: Unlike traditional methods that utilize highly toxic potassium cyanide (KCN) or hydroxyl acetonitrile, this patented process employs malononitrile as the cyanating source under mild conditions, significantly reducing operator risk and hazardous waste generation.
Q: What is the overall yield efficiency of this benzopyran derivative synthesis?
A: The patent data indicates an impressive overall yield ranging from 70% to 80%, achieved through a short three-step sequence that minimizes material loss during intermediate isolation and purification stages.
Q: Can this process be adapted for large-scale commercial production?
A: Yes, the reaction conditions are mild (40-80°C) and utilize common solvents like ethanol and DMF, making the process highly amenable to scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Cyano Benzopyran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced on an industrial scale. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and potency of every batch of 2-amino-3-cyano benzopyran derivatives we produce. Our facility is equipped to handle the specific solvent systems and thermal requirements of this process safely and efficiently, guaranteeing a consistent supply of high-quality intermediates for your critical projects.
We invite you to collaborate with us to leverage this innovative synthetic route for your next development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down your overall production costs.
