Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics and functional materials. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These specific molecular architectures are of immense value due to the prevalence of the 1,2,4-triazole ring in bioactive molecules and its utility as a bidentate ligand in coordination chemistry for applications such as organic light-emitting diodes (OLEDs). The disclosed technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot oxidative cyclization strategy that dramatically improves operational simplicity and overall yield.
For R&D directors and process chemists evaluating new routes, the ability to introduce both quinolinyl and trifluoromethyl motifs simultaneously with high regioselectivity is a compelling advantage. This patent provides a comprehensive framework for synthesizing these diversified structures using readily available precursors, thereby reducing the barrier to entry for developing novel drug candidates or advanced electronic materials. By leveraging a metal-free catalytic system, the method addresses key pain points regarding heavy metal contamination and environmental compliance, positioning it as a superior alternative for modern green chemistry initiatives in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the methodology described in CN113307790B, the synthesis of quinolyl-substituted 1,2,4-triazoles was fraught with significant inefficiencies that hindered large-scale application. The traditional approach typically relied on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the desired molecular complexity. This multi-step pathway not only resulted in a dismal total yield of approximately 17%, representing a massive loss of valuable raw materials, but also required severe reaction conditions that demanded rigorous control over temperature and atmosphere. Furthermore, the reliance on multiple isolation and purification steps between each transformation increased the operational burden, extended production timelines, and generated substantial chemical waste, making the conventional route economically unviable for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to the arduous traditional pathways, the novel approach utilizes a direct oxidative cyclization strategy that merges 2-methylquinoline and trifluoroacetimidoyl hydrazide in a single pot. As illustrated in the reaction scheme below, this method employs a catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) promoted by diphenylphosphoric acid to drive the transformation efficiently.
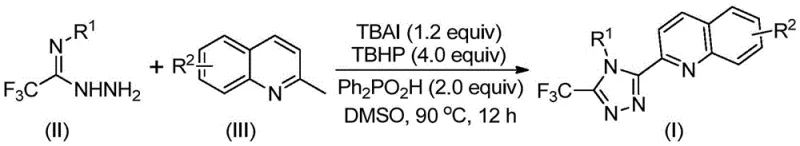
This innovative route operates under significantly milder conditions, typically requiring heating to 80-100°C in a solvent like DMSO for 8 to 14 hours, without the need for strict anhydrous or oxygen-free environments. The elimination of transition metal catalysts is a critical improvement, as it removes the necessity for expensive and difficult-to-remove heavy metal scavengers during workup. Experimental data from the patent indicates that this method can achieve isolated yields as high as 97% for certain substrates, such as the 4-fluorophenyl derivative, demonstrating exceptional efficiency and atom economy compared to the legacy 17% yield processes.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The mechanistic underpinning of this transformation involves a sophisticated cascade of oxidative and cyclization events initiated by the TBAI/TBHP system. Initially, the tetrabutylammonium iodide and tert-butyl peroxide promote the oxidation of the methyl group on the 2-methylquinoline substrate, effectively converting it in situ into a 2-quinolinecarbaldehyde intermediate. This reactive aldehyde species then undergoes a condensation reaction with the trifluoroacetimidoyl hydrazide to form a dehydrated hydrazone intermediate. Subsequent oxidative iodination facilitates an intramolecular electrophilic substitution reaction, which closes the triazole ring. The final step involves aromatization to yield the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole product. It is also plausible that the reaction proceeds partially through a free radical mechanism, given the nature of the peroxide oxidant, which contributes to the robustness of the reaction across various substrate electronic environments.
From an impurity control perspective, the use of diphenylphosphoric acid as an additive plays a crucial role in stabilizing intermediates and promoting the desired cyclization pathway over potential side reactions. The tolerance of this catalytic system towards diverse functional groups—such as halogens, alkyls, alkoxy groups, and nitro groups on both the hydrazide and the quinoline rings—suggests a highly selective mechanism that minimizes the formation of byproducts. This selectivity is paramount for pharmaceutical applications where impurity profiles must be strictly controlled to meet regulatory standards, ensuring that the final high-purity API intermediate requires minimal downstream purification effort beyond standard column chromatography or crystallization.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators should adhere to the optimized molar ratios and conditions established in the patent examples. The process begins by charging a reaction vessel with the requisite amounts of tetrabutylammonium iodide, 70% tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in an aprotic organic solvent, with DMSO being the preferred medium for maximizing conversion. The detailed standardized synthesis steps, including specific stoichiometric ratios for different substituents and precise workup procedures, are outlined in the guide below.
- Combine tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for a duration of 8 to 14 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the final 3-quinolyl-5-trifluoromethyl substituted product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers profound strategic advantages that extend beyond mere chemical efficiency. The shift away from precious metal catalysts directly impacts the cost structure of manufacturing by eliminating the procurement of expensive palladium or copper salts and the associated costs of specialized ligands. Furthermore, the removal of heavy metals from the process flow significantly reduces the complexity and cost of wastewater treatment and final product purification, leading to substantial cost savings in the overall production lifecycle. The use of cheap, commodity-grade starting materials like 2-methylquinoline and aromatic amines ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts creates a direct pathway for cost reduction in pharmaceutical intermediate manufacturing by removing the need for expensive catalyst loading and the subsequent rigorous purification steps required to meet residual metal specifications. The high yields achieved, often exceeding 80-90% for optimized substrates, mean that less raw material is wasted per kilogram of finished product, drastically improving the material cost basis. Additionally, the ability to run the reaction without strict inert atmosphere conditions reduces the capital expenditure on specialized reactor equipment and lowers the operational energy costs associated with maintaining nitrogen or argon blankets.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity chemicals such as tetrabutylammonium iodide and tert-butyl peroxide enhances supply chain reliability by reducing dependency on single-source suppliers of exotic reagents. Since the reaction tolerates a wide range of functional groups and does not require ultra-dry solvents, the logistics of raw material storage and handling are simplified, reducing the risk of production delays caused by material degradation or stringent handling requirements. This robustness allows for more flexible scheduling and inventory management, ensuring consistent delivery of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The simplicity of the post-treatment process, which involves filtration and standard chromatography, facilitates easy scale-up from gram-scale laboratory synthesis to multi-ton commercial production without encountering the heat transfer or mixing issues common in complex multi-step sequences. The absence of toxic heavy metals aligns perfectly with increasingly stringent global environmental regulations, minimizing the generation of hazardous waste and simplifying the permitting process for manufacturing facilities. This environmental compliance not only protects the company from regulatory fines but also enhances the brand reputation as a sustainable and responsible chemical supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process (CN113307790B) utilizes a metal-free catalytic system based on tetrabutylammonium iodide and tert-butyl peroxide, eliminating the need for costly palladium or copper catalysts and simplifying downstream purification.
Q: What are the typical reaction conditions for this oxidative cyclization?
A: The reaction operates under relatively mild conditions, requiring heating to 80-100°C in an aprotic solvent like DMSO for 8-14 hours, without the need for strict anhydrous or oxygen-free environments.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method uses cheap and commercially available starting materials, avoids sensitive reaction conditions, and has been demonstrated to be easily expandable to gram-scale reactions, making it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative cyclization technology described in CN113307790B for producing high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole delivered meets the exacting standards required by the global pharmaceutical and electronic materials industries.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthetic route for their specific project needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable, high-quality chemical solutions.
