Advanced Synthesis of Carbazole-Fluorene Hybrid Hole-Transporting Materials for Commercial OLED Manufacturing
Advanced Synthesis of Carbazole-Fluorene Hybrid Hole-Transporting Materials for Commercial OLED Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit high efficiency but also possess exceptional thermal and morphological stability to ensure long device lifespans. Patent CN102030702B introduces a breakthrough in this domain by disclosing a novel hole-transporting material, specifically N,N'-diphenyl-N-(9,9-dimethyl-2-fluorenyl)-N'-(9',9'-dimethyl-7'-(9''-phenyl-3''-carbazolyl)-2'-fluorenyl)-benzidine. This complex asymmetric molecule is engineered to overcome the inherent limitations of traditional triarylamine-based transporters, which often suffer from low glass transition temperatures and unwanted crystallization during device operation. The synthesis strategy outlined in the patent employs a modular approach, constructing the final architecture through three distinct fragments that are subsequently linked via robust catalytic coupling reactions. This structural innovation effectively suppresses crystallization while maintaining excellent hole mobility, addressing a critical pain point for display manufacturers seeking to enhance the reliability of next-generation flat-panel screens.
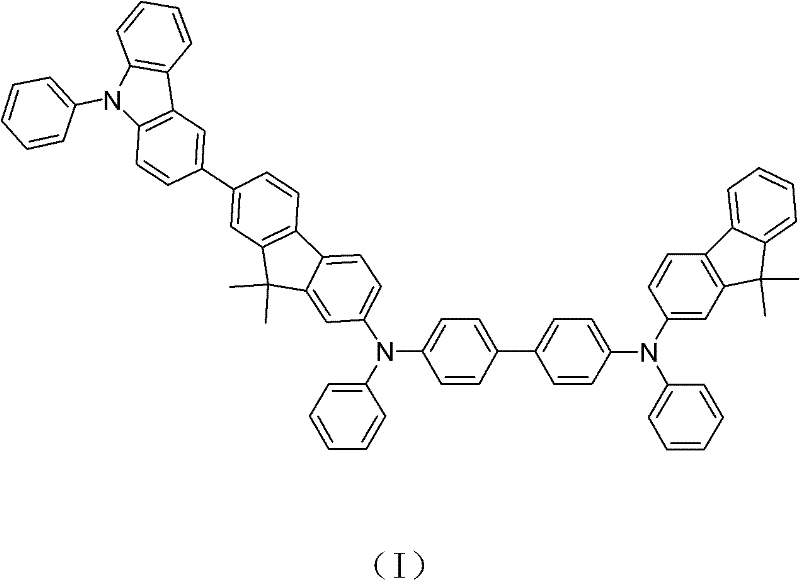
The molecular design integrates a carbazole unit with a fluorene backbone, creating significant steric hindrance that disrupts molecular packing. This disruption is key to achieving the low crystallinity required for stable amorphous films in OLED stacks. By preventing the formation of crystalline domains, the material ensures uniform charge transport and minimizes the risk of dark spot formation, a common failure mode in organic displays. Furthermore, the specific substitution pattern enhances the thermal decomposition temperature, allowing the material to withstand the rigorous thermal evaporation processes used in device fabrication without degrading. For R&D directors focused on material performance, this represents a significant step forward in balancing charge injection capabilities with physical durability, ultimately leading to displays with wider viewing angles and higher contrast ratios that remain stable over thousands of hours of operation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional hole-transporting materials, such as NPB or TPD, have long served as industry standards but are increasingly showing their age in high-performance applications. These conventional triarylamines often exhibit relatively low glass transition temperatures, making them susceptible to morphological changes when subjected to the heat generated during device operation or the deposition process. When these materials crystallize, they create grain boundaries that act as traps for charge carriers, leading to uneven current distribution and accelerated device degradation. Moreover, the synthesis of highly pure grades of these legacy materials often involves difficult purification steps to remove trace metal catalysts or unreacted amines, which can act as quenching centers in the emissive layer. The inability to effectively block excessive holes from entering the luminescent layer is another drawback, as this imbalance in charge carrier injection reduces quantum efficiency and shortens the operational lifetime of the OLED panel.
The Novel Approach
The synthetic route described in the patent offers a sophisticated solution by constructing a bulky, asymmetric molecule that inherently resists crystallization. Instead of relying on simple triarylamine cores, the process builds a hybrid structure where a carbazole moiety is attached to a fluorene-biphenyl-amine backbone. This architectural complexity introduces significant steric bulk that physically prevents the molecules from aligning into ordered crystalline lattices, thereby maintaining a stable amorphous state even under thermal stress. The use of sequential Ullmann condensations and a final Suzuki coupling allows for precise control over the molecular geometry, ensuring that the highest occupied molecular orbital (HOMO) levels are optimized for efficient hole injection from the anode. This approach not only solves the stability issue but also simplifies the device architecture by potentially eliminating the need for additional hole-blocking layers, thus streamlining the manufacturing process for electronic chemical producers.
Mechanistic Insights into Ullmann Condensation and Suzuki Coupling
The core of this synthesis relies on the strategic application of transition metal-catalyzed cross-coupling reactions to assemble the three distinct molecular fragments. The initial formation of the amine linkages is achieved through Ullmann condensation, utilizing a copper(I) iodide and phenanthroline catalytic system. This specific ligand environment is crucial for activating the aryl halides, facilitating the nucleophilic attack by the amine groups under reflux conditions in toluene. The mechanism involves the oxidative addition of the aryl iodide to the copper center, followed by coordination of the amine and subsequent reductive elimination to form the C-N bond. This method is particularly advantageous for scaling up as it avoids the use of more expensive palladium catalysts for these specific steps, offering a cost-effective pathway for constructing the nitrogen-containing backbone of the hole-transporting material.
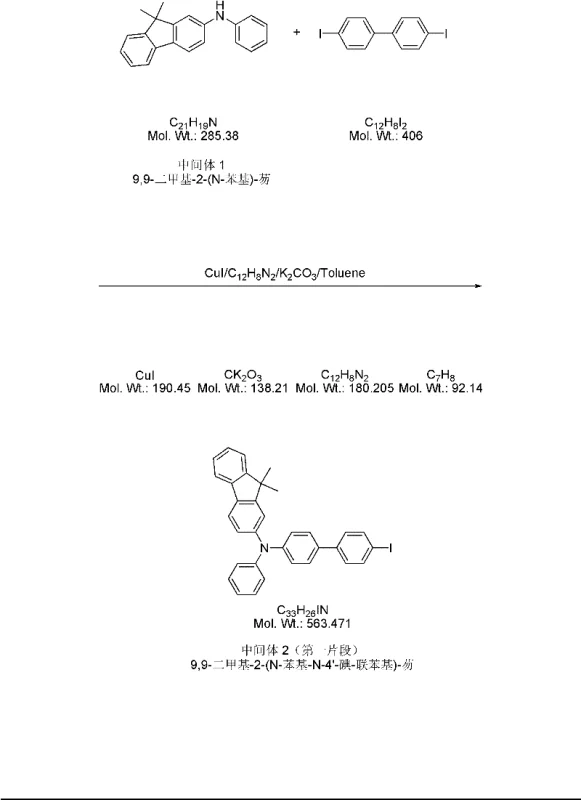
For the final assembly of the molecule, the patent employs a Suzuki-Miyaura coupling reaction, which is renowned for its tolerance to functional groups and high yields. This step connects the bromo-substituted fluorene intermediate (Fragment 7) with the carbazole boronic acid derivative (Fragment 6). The reaction proceeds via a palladium(0) catalytic cycle, typically using tetrakis(triphenylphosphine)palladium as the catalyst source. The mechanism involves the oxidative addition of the aryl bromide to the palladium center, transmetallation with the organoboron species activated by base, and finally reductive elimination to forge the critical C-C bond. This step is vital for introducing the carbazole unit without compromising the integrity of the rest of the molecule. The choice of solvents, such as a toluene/methanol mixture, and bases like potassium carbonate, ensures that the reaction proceeds smoothly at moderate temperatures, minimizing side reactions and preserving the high purity required for electronic grade materials.
How to Synthesize N,N'-diphenyl-N-(9,9-dimethyl-2-fluorenyl)-N'-(9',9'-dimethyl-7'-(9''-phenyl-3''-carbazolyl)-2'-fluorenyl)-benzidine Efficiently
The synthesis of this advanced hole-transporting material is a multi-stage process that requires precise control over reaction conditions to ensure high yields and purity. The protocol begins with the preparation of three separate intermediates, each synthesized through specific functional group transformations such as nitration, reduction, and lithiation. These fragments are then converged through sequential coupling reactions, culminating in a final purification stage that involves column chromatography and recrystallization. The detailed procedure outlines specific molar ratios, temperature ranges, and workup protocols designed to maximize the efficiency of each step while minimizing the formation of byproducts. For process chemists looking to implement this route, understanding the nuances of the catalyst loading and the purification of intermediates is essential for achieving the reported purity levels of greater than 99%.
- Synthesize Fragment 1 via Ullmann condensation of 9,9-dimethyl-2-aminofluorene with iodobenzene and 4,4'-diiodobiphenyl using CuI catalyst.
- Prepare Fragment 2 by nitrating and reducing 9,9-dimethyl-2-bromofluorene, followed by N-phenylation via Ullmann reaction.
- Generate Fragment 3 by lithiating 9-phenyl-3-bromocarbazole and reacting with borate ester to form the corresponding boronic acid.
- Couple Fragment 1 and Fragment 2 using Ullmann conditions to form the bromo-intermediate (Fragment 7).
- Perform final Suzuki coupling between Fragment 7 and Fragment 3 using Pd(PPh3)4 catalyst to yield the target hole-transporting material.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthesis method detailed in this patent offers several compelling advantages that translate directly into operational efficiency and cost optimization. The reliance on commodity chemicals such as fluorene, carbazole, and iodobenzene as starting materials ensures a stable and reliable supply chain, reducing the risk of bottlenecks associated with exotic or proprietary precursors. Furthermore, the modular nature of the synthesis allows for the parallel production of fragments, which can significantly reduce the overall lead time for manufacturing the final active pharmaceutical ingredient or electronic chemical. The high yields reported in the intermediate steps, particularly the reduction and coupling reactions, mean that less raw material is wasted, contributing to a more sustainable and economically viable production process that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The process utilizes copper-catalyzed Ullmann couplings for the formation of C-N bonds, which are significantly less expensive than palladium-catalyzed alternatives often used for similar transformations. By reserving the more costly palladium catalyst strictly for the final Suzuki coupling step, the overall catalyst cost per kilogram of product is drastically reduced. Additionally, the high conversion rates and the ability to purify intermediates through standard recrystallization techniques minimize the need for expensive preparative HPLC purification, leading to substantial cost savings in the downstream processing phases of electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The synthetic route is designed around robust chemical transformations that are well-understood and easily scalable, reducing the technical risk associated with technology transfer from lab to plant. The starting materials are widely available from multiple global suppliers, ensuring that procurement teams can negotiate competitive pricing and secure long-term contracts without fear of single-source dependency. This diversity in the supply base enhances the resilience of the supply chain against market fluctuations or geopolitical disruptions, guaranteeing a continuous flow of high-quality hole-transporting materials for OLED panel production lines.
- Scalability and Environmental Compliance: The reaction conditions employed, such as refluxing in toluene or THF, are compatible with standard stainless steel reactors found in most fine chemical manufacturing facilities, facilitating easy scale-up from pilot plant to commercial tonnage. The workup procedures primarily involve aqueous washes and filtration, which generate waste streams that are easier to treat and manage compared to processes requiring complex extractive workups or hazardous reagents. This alignment with environmental compliance standards reduces the burden on waste treatment facilities and supports the company's sustainability goals while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this novel hole-transporting material. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on performance metrics and process feasibility. Understanding these aspects is crucial for stakeholders evaluating the material for integration into their existing OLED manufacturing workflows or for R&D teams planning further derivatization studies.
Q: How does this new hole-transporting material improve OLED device lifespan?
A: The material features a bulky carbazole-fluorene hybrid structure that significantly lowers crystallinity and increases thermal stability compared to traditional triarylamines. This morphological stability prevents excessive holes from penetrating the emissive layer, thereby reducing degradation and extending the operational life of the OLED device.
Q: What is the purity level achievable with this synthesis method?
A: The patented process utilizes rigorous purification steps including silica gel column chromatography and recrystallization. Analytical data confirms that the final product achieves a purity greater than 99%, which is critical for preventing quenching sites and ensuring high efficiency in electronic applications.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method relies on robust catalytic systems like CuI/phenanthroline for Ullmann coupling and standard Suzuki conditions. The use of commercially available starting materials and high-yield intermediate steps makes the process economically viable and scalable for mass manufacturing of electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-diphenyl-N-(9,9-dimethyl-2-fluorenyl)-N'-(9',9'-dimethyl-7'-(9''-phenyl-3''-carbazolyl)-2'-fluorenyl)-benzidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance hole-transporting materials play in the advancement of OLED technology. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global display market. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the >99% purity threshold required for optimal device performance. Our state-of-the-art facilities are equipped to handle the complex catalytic reactions and purification steps necessary for synthesizing advanced electronic chemicals, guaranteeing a consistent supply of materials that drive innovation in the industry.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized synthesis routes can reduce your overall material costs without compromising quality. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will accelerate your product development timelines and strengthen your position in the competitive OLED marketplace.
