Advanced Chiral Carbene Precursors for High-Selectivity Asymmetric Hydroboration and Commercial Scale-Up
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel chiral ligand systems described in patent CN109776422B. This intellectual property details the development of chiral 1,3-diarylimidazolium salt carbene precursors that serve as highly effective ligands for copper-catalyzed reactions. Specifically, these precursors enable the asymmetric hydroboration of non-activated terminal alkenes to produce chiral Markovnikov borides with exceptional regioselectivity and enantioselectivity. For R&D directors and procurement specialists in the pharmaceutical and fine chemical sectors, this represents a critical advancement in accessing complex chiral building blocks that were previously difficult to synthesize efficiently. The technology addresses the longstanding challenge of achieving high stereocontrol in the functionalization of simple olefins without relying on scarce and costly precious metal catalysts.
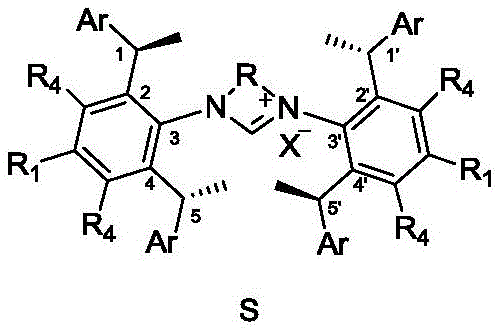
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct hydroboration of non-activated terminal alkenes to form chiral Markovnikov borides has been a formidable challenge in organic synthesis. Traditional methods often relied on rhodium-based catalytic systems, which, while effective to a degree, suffered from significant limitations in terms of enantioselective excess values, typically ranging between 72% and 90% ee. Furthermore, the reliance on rhodium introduces substantial cost volatility and supply chain risks due to the scarcity of this precious metal. The purification processes required to remove trace rhodium residues from pharmaceutical intermediates are also stringent and expensive, adding layers of complexity to the manufacturing workflow. Additionally, many conventional ligands lacked the necessary steric bulk and electronic tunability to enforce the high degree of stereocontrol required for demanding synthetic applications, often resulting in mixtures of regioisomers that complicate downstream processing.
The Novel Approach
The innovative approach outlined in the patent utilizes chiral 1,3-diarylimidazolium salts which, upon complexation with copper, form robust catalytic species capable of overcoming these historical barriers. By leveraging the unique steric environment provided by the chiral benzyl substituents on the imidazolium backbone, these catalysts achieve regioselectivity ratios favoring the Markovnikov product as high as 90:10 and enantioselectivity values reaching up to 97% ee. This shift from rhodium to copper not only drastically reduces the raw material costs associated with the catalyst but also simplifies the regulatory pathway for drug substance manufacturing by eliminating heavy metal concerns. The modular nature of the imidazolium salt synthesis allows for fine-tuning of the ligand properties to suit specific substrate profiles, offering a versatile platform for diverse chemical transformations beyond just hydroboration.
Mechanistic Insights into Copper-NHC Catalyzed Asymmetric Hydroboration
The catalytic cycle begins with the generation of the active copper-N-heterocyclic carbene (NHC) species from the imidazolium salt precursor in the presence of a base and a copper source. This active complex coordinates with the diboron reagent to form a copper-boryl species, which is the key intermediate responsible for the bond-forming event. The chiral pocket created by the bulky aryl groups on the NHC ligand dictates the approach of the alkene substrate, ensuring that the boryl group adds to the internal carbon of the terminal alkene with high fidelity. This mechanistic pathway effectively overrides the natural electronic preference for anti-Markovnikov addition, directing the reaction towards the desired branched product. The stability of the copper-NHC bond ensures that the catalyst remains active throughout the reaction cycle, minimizing decomposition and allowing for lower catalyst loadings which is crucial for industrial scalability.
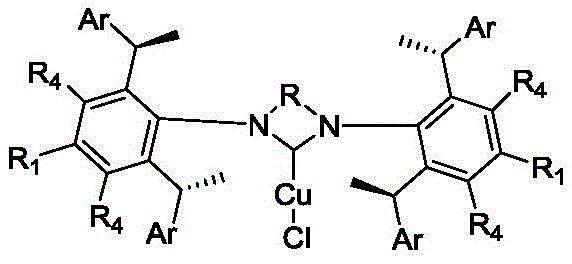
Impurity control is inherently built into this system through the high specificity of the catalyst-substrate interaction. The rigid chiral framework prevents the formation of unwanted side products such as anti-Markovnikov isomers or homocoupling byproducts that are common in less selective systems. This high level of chemoselectivity reduces the burden on purification teams, as the crude reaction mixtures are significantly cleaner compared to those generated by traditional methods. For quality assurance teams, this means fewer chromatographic steps are required to meet stringent purity specifications, directly translating to higher overall yields and reduced solvent consumption. The ability to predictably control the stereochemical outcome also ensures batch-to-batch consistency, a critical factor for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Chiral 1,3-Diarylimidazolium Salts Efficiently
The synthesis of these valuable precursors follows a logical and scalable multi-step sequence that begins with readily available chiral amines and dicarbonyl compounds. The process involves condensation reactions to form diimine intermediates, followed by reduction to diamines, and finally cyclization and quaternization to yield the target imidazolium salts. Each step has been optimized to maximize yield and minimize waste, utilizing common solvents and reagents that are easily sourced on a global scale. The detailed standardized synthesis steps see the guide below.
- Condense chiral benzylamine derivatives with dicarbonyl compounds like glyoxal or diacetyl in alcoholic solvents with acid catalysis to form diimine intermediates.
- Reduce the diimine intermediates using reducing agents such as lithium aluminum hydride or sodium borohydride to obtain the corresponding diamine precursors.
- Cyclize the diamines with orthoformates or dihaloalkanes under heating conditions, followed by quaternization with alkyl halides to yield the final imidazolium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling economic and logistical benefits that extend beyond simple performance metrics. The transition from precious metal catalysts to base metal copper systems fundamentally alters the cost structure of producing chiral organoboranes, removing the exposure to volatile rhodium markets. This stability allows for more accurate long-term budgeting and pricing strategies for downstream customers. Furthermore, the robustness of the imidazolium salts simplifies storage and transportation requirements, as they do not require the specialized handling conditions often mandated for air-sensitive or unstable catalytic species. This ease of handling reduces the risk of supply disruptions and ensures a reliable flow of materials to production facilities.
- Cost Reduction in Manufacturing: The substitution of expensive rhodium catalysts with abundant copper salts results in a substantial decrease in raw material costs per kilogram of product. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side reactions, improving the overall atom economy of the process. The simplified purification protocols required due to high crude purity further reduce operational expenditures related to solvent usage and waste disposal. These cumulative efficiencies drive down the cost of goods sold, making high-value chiral intermediates more accessible for large-scale commercial applications.
- Enhanced Supply Chain Reliability: Copper is a globally abundant commodity with a stable supply chain, unlike rhodium which is subject to geopolitical constraints and mining bottlenecks. By decoupling the catalytic process from scarce precious metals, manufacturers can secure a more resilient supply chain that is less susceptible to external shocks. The synthetic routes for the ligands themselves utilize commodity chemicals, ensuring that the production of the catalyst precursor can be scaled up rapidly to meet surging demand without waiting for specialized feedstock. This reliability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for key clients.
- Scalability and Environmental Compliance: The processes described are amenable to scale-up from laboratory to multi-ton production without significant re-engineering, facilitating a smooth technology transfer. The use of copper also aligns with green chemistry principles by reducing the environmental footprint associated with precious metal mining and refining. Lower metal residues in the final product simplify wastewater treatment and reduce the regulatory burden associated with heavy metal discharge limits. This environmental compatibility supports corporate sustainability goals and ensures compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral carbene technology in industrial settings. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation to ensure accuracy and relevance for decision-makers.
Q: What is the primary advantage of these chiral imidazolium salts over traditional rhodium catalysts?
A: These copper-based NHC precursors offer superior enantioselectivity (up to 97% ee) and regioselectivity for Markovnikov hydroboration compared to previous rhodium methods which often yielded lower ee values around 72-90%.
Q: Are these carbene precursors stable for long-term storage and transport?
A: Yes, the 1,3-diarylimidazolium salt structure provides exceptional thermal and chemical stability, making them easy to store and handle compared to free carbene species, which facilitates global logistics.
Q: How does this technology impact the cost of producing chiral organoboranes?
A: By utilizing abundant copper salts instead of expensive precious metals like rhodium and employing robust synthetic routes, the overall manufacturing cost is significantly reduced while maintaining high purity standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,3-Diarylimidazolium Salt Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable chemical solutions. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to manufacture these complex chiral ligands and their copper complexes with consistent quality. Our rigorous QC labs and stringent purity specifications ensure that every batch meets the exacting standards required for pharmaceutical and fine chemical applications, providing our partners with the confidence to integrate these materials into their critical synthesis routes.
We invite you to contact our technical procurement team to discuss how this technology can optimize your specific manufacturing processes. Request a Customized Cost-Saving Analysis to quantify the potential economic benefits for your operation, and ask for specific COA data and route feasibility assessments tailored to your target molecules. Let us help you leverage this cutting-edge catalytic system to achieve superior selectivity and efficiency in your next project.
