Advanced Manufacturing of 1-Iodo-3-Perfluoroalkyl Olefins for Global Pharmaceutical Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to introduce fluorine atoms into organic scaffolds, given the profound impact fluorination has on metabolic stability and bioavailability. Patent CN112047839A discloses a groundbreaking approach for the preparation of 1-iodo-3-perfluoroalkyl olefin compounds, which serve as critical building blocks for complex drug candidates and advanced materials. These compounds possess high chemical activity and rich reactivity, making them indispensable for coupling reactions such as Kumada, Negishi, and Sonogashira protocols, as well as radical cycloaddition processes. The structural versatility of these molecules allows for the incorporation of diverse perfluoroalkyl groups, ranging from trifluoromethyl to longer perfluoroalkyl chains, thereby enabling the fine-tuning of physicochemical properties for specific therapeutic applications. 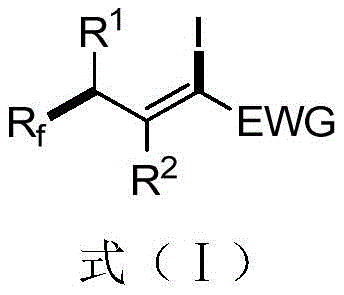
The significance of this technology lies in its ability to overcome the limitations of traditional fluorination methods, which often suffer from harsh reaction conditions and poor selectivity. By leveraging visible-light promotion, the process operates under mild conditions that are insensitive to air and moisture, a critical factor for reducing operational costs in large-scale manufacturing environments. The resulting 1-iodo-3-perfluoroalkyl olefins exhibit high cis-trans selectivity, particularly favoring the Z-isomer, which is often the thermodynamically preferred configuration for subsequent biological activity. This level of stereocontrol is achieved without the need for complex chiral auxiliaries, streamlining the synthetic route and enhancing overall process efficiency for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for alkenyl iodides frequently rely on the use of expensive and scarce transition metal photocatalysts, such as Ruthenium or Iridium complexes, which significantly inflate the cost of goods sold for high-volume intermediates. Furthermore, conventional methods often necessitate the addition of specific initiators and additives in multiple stages, complicating the reaction workflow and increasing the risk of impurity formation that requires rigorous purification steps. For instance, prior art methods may involve the stepwise addition of initiators to control radical generation, which introduces variability and reduces the reproducibility of the process across different batch sizes. The reliance on sensitive reagents also demands stringent exclusion of air and moisture, requiring specialized equipment and inert atmosphere conditions that are not always feasible in standard multipurpose chemical plants.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a visible-light promoted atom transfer radical addition (ATRA) reaction that proceeds efficiently without the need for external noble metal photocatalysts. This method employs inexpensive and readily available organic solvents and operates under mild temperatures ranging from 0°C to 80°C, drastically reducing energy consumption and safety hazards associated with high-temperature processes. The reaction system is robust against air and moisture, allowing for simpler reactor setups and reducing the downtime associated with vessel drying and purging. Additionally, the process generates nitrogen gas as the sole byproduct during the decomposition of the diazo precursor, aligning perfectly with green chemistry principles and minimizing the environmental footprint of the manufacturing operation.
Mechanistic Insights into Visible-Light Promoted ATRA and Isomerization
The core of this technological advancement lies in the synergistic combination of visible-light promotion and manganese-catalyzed isomerization to achieve high stereocontrol. Initially, the alkenyl diazo compound reacts with the perfluoroalkyl iodide under visible light irradiation to form an electron donor-acceptor (EDA) complex, which facilitates the generation of perfluoroalkyl radicals without external photosensitizers. These radicals add across the double bond of the diazo compound, resulting in a mixture of Z and E isomers of the 1-iodo-3-perfluoroalkyl olefin. The use of visible light sources such as CFL, LED white, or blue light ensures that the reaction can be driven by energy-efficient lighting systems, further contributing to the sustainability of the process. 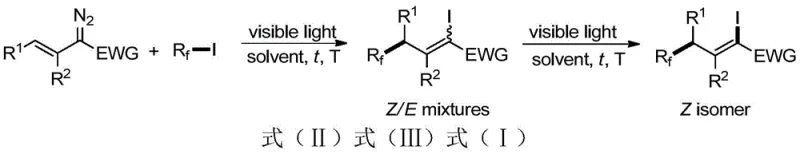
Following the initial ATRA step, the reaction mixture undergoes a crucial isomerization phase catalyzed by manganese complexes, such as dimanganese decacarbonyl. This step is pivotal for converting the thermodynamic mixture of isomers into the desired Z-isomer with high selectivity, often achieving Z/E ratios greater than 30:1. The manganese catalyst facilitates the equilibration of the double bond geometry under continued light irradiation, ensuring that the final product is enriched in the stereochemically pure form required for downstream coupling reactions. This dual-step mechanism not only enhances the purity of the final intermediate but also simplifies the purification process, as the high selectivity reduces the burden on chromatographic separation techniques.
How to Synthesize 1-Iodo-3-Perfluoroalkyl Olefin Efficiently
The synthesis of these high-value intermediates follows a streamlined two-step protocol that is amenable to both laboratory-scale optimization and industrial-scale production. The process begins with the mixing of the alkenyl diazoacetate and perfluoroalkyl iodide in a suitable organic solvent, followed by irradiation to initiate the radical addition. After the initial reaction is complete, excess perfluoroalkyl iodide is removed via vacuum distillation, and the manganese catalyst is introduced to drive the isomerization to completion. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and quality control across different manufacturing sites.
- Conduct visible-light promoted ATRA reaction between alkenyl diazo compound and perfluoroalkyl iodide in organic solvent to obtain Z/E mixture.
- Remove excess perfluoroalkyl iodide via vacuum distillation from the reaction mixture.
- Add manganese catalyst and continue reaction under light to isomerize mixture to cis-1-iodo-3-perfluoroalkyl olefin compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this manufacturing route offers substantial cost reductions by eliminating the dependency on precious metal catalysts that are subject to volatile market pricing and supply constraints. The use of earth-abundant manganese catalysts and common organic solvents ensures a stable and predictable cost structure, allowing for more accurate budgeting and long-term supply agreements. Furthermore, the simplicity of the reaction workup, which involves basic concentration and chromatography, reduces the consumption of auxiliary materials and solvents, contributing to a lower overall cost of production per kilogram of the final intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive Ruthenium or Iridium photocatalysts significantly lowers the raw material costs, while the mild reaction conditions reduce energy expenditures associated with heating and cooling. The high yield and selectivity of the process minimize the loss of valuable starting materials, ensuring that the maximum amount of input is converted into saleable product. Additionally, the simplified purification process reduces the labor and time required for downstream processing, further enhancing the economic viability of the route for commercial scale-up.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including alkenyl diazo compounds and perfluoroalkyl iodides, are commercially available from multiple suppliers, reducing the risk of single-source dependency. The robustness of the reaction to air and moisture means that storage and handling requirements are less stringent, lowering the risk of spoilage during transit and warehousing. This reliability ensures consistent delivery schedules and reduces the likelihood of production delays caused by material degradation or supply shortages.
- Scalability and Environmental Compliance: The generation of nitrogen gas as the only byproduct simplifies waste management and reduces the environmental compliance burden associated with hazardous waste disposal. The process is inherently scalable, as the visible-light irradiation can be adapted to flow chemistry setups or large batch reactors without significant loss of efficiency. This scalability supports the transition from pilot-scale development to full commercial production, meeting the growing demand for fluorinated intermediates in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 1-iodo-3-perfluoroalkyl olefin compounds. These answers are derived from the detailed technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these aspects is crucial for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the advantages of this visible-light method over traditional photocatalysts?
A: This method eliminates the need for expensive noble metal photocatalysts like Ru or Ir complexes, utilizing inexpensive manganese catalysts and visible light, significantly reducing raw material costs and simplifying metal removal processes.
Q: How is high Z-selectivity achieved in the final product?
A: High Z-selectivity is achieved through a specific manganese-catalyzed isomerization step following the initial ATRA reaction, which converts the Z/E mixture into the desired cis-isomer with ratios often exceeding 30:1.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up due to its insensitivity to air and moisture, simple operation conditions, and the generation of nitrogen gas as the only byproduct, aligning with green manufacturing standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Iodo-3-Perfluoroalkyl Olefin Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the visible-light promoted ATRA reaction to deliver high-purity intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from development to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for drug substance production.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this innovative synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your project timelines and enhance your competitive advantage in the market. 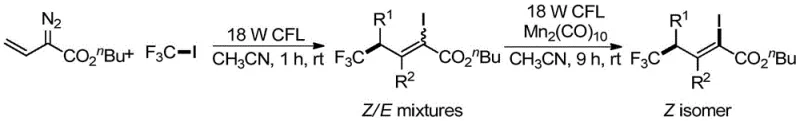
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
