Transforming Pharmaceutical Intermediate Manufacturing: High-Yield Iridium-Catalyzed Synthesis of Allyl Alcohol Derivatives for Commercial Scale-Up
The patent CN111072450A introduces a groundbreaking synthetic methodology for allyl alcohol derivatives that addresses critical limitations in traditional production routes. This innovation leverages a novel two-step process starting from readily available organic acyl chlorides, which undergo ethylene addition to form stable 3-chloropropyl intermediates followed by iridium-catalyzed reduction. The methodology achieves exceptional yields exceeding ninety percent per step while eliminating the need for expensive and unstable α-carbonyl alkene precursors that plague conventional approaches. Crucially, the incorporation of alkali metal bicarbonate as a reaction auxiliary enables selective reduction that was previously unattainable with standard reducing agents, representing a significant advancement in synthetic organic chemistry. This patent provides a robust foundation for commercial manufacturing of these versatile intermediates essential for liquid crystal materials and pharmaceutical applications, with particular relevance to trans-configured alkene synthesis required in complex drug molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of allyl alcohol derivatives primarily relies on two problematic approaches that create substantial barriers for industrial implementation. The first method employs aldehydes reacting with vinyl chloride Grignard reagents, but this requires aldehydes that are rarely commercially available and must be synthesized through multi-step processes involving acids, acyl chlorides, or alcohols—each step introducing yield losses and environmental burdens from expensive reagents or hazardous byproducts. The second conventional route utilizes selective reduction of α-carbonyl alkenes (compound 1B), yet these precursors are both more expensive to produce than aldehydes and chemically unstable, prone to self-polymerization during storage or heating, which severely compromises process reliability and product consistency. Both methods suffer from low overall yields due to intermediate instability and complex purification requirements, making them economically unviable for large-scale pharmaceutical intermediate production where consistent quality and cost efficiency are paramount for supply chain operations.
The Novel Approach
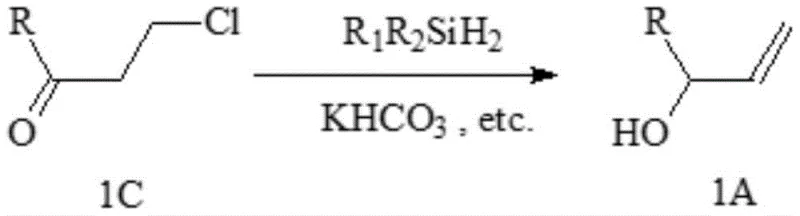
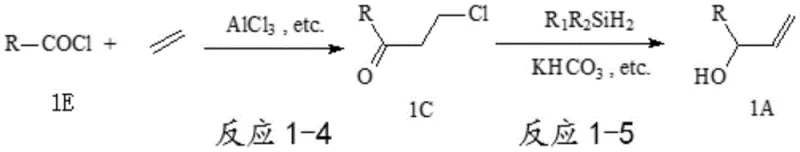
The patented methodology overcomes these limitations through an innovative two-step sequence that begins with stable organic acyl chlorides reacting with ethylene under Lewis acid catalysis to form robust 3-chloropropyl derivatives (compound 1C), which then undergo iridium-catalyzed reduction with dialkyldihydrosilanes in the presence of alkali metal bicarbonate. This approach eliminates the need for unstable α-carbonyl alkenes while leveraging commercially accessible starting materials that significantly reduce raw material costs. The critical innovation lies in the bicarbonate promoter that enables selective reduction to allyl alcohol derivatives without forming undesired chlorohydrin byproducts—a breakthrough that achieves yields exceeding ninety percent per step with minimal purification requirements. This process demonstrates exceptional scalability from laboratory to commercial production while maintaining stringent purity specifications essential for pharmaceutical applications, directly addressing the supply chain vulnerabilities inherent in prior art methods.
Mechanistic Insights into Iridium-Catalyzed Bicarbonate-Promoted Reduction
The core innovation resides in the unique mechanistic pathway enabled by the synergistic interaction between iridium complexes and alkali metal bicarbonate during the reduction step. Conventional reduction methods using dialkyldihydrosilanes alone on 3-chloropropyl derivatives produce undesired chlorohydrin byproducts (compound 1D) due to competitive hydride transfer pathways, but the addition of bicarbonate creates a controlled basic environment that directs selectivity toward allylic alcohol formation. The iridium catalyst facilitates hydride transfer from silicon to carbonyl carbon while bicarbonate acts as a proton shuttle, preventing chloride displacement and enabling β-hydride elimination that forms the critical allylic double bond. This mechanism operates efficiently at moderate temperatures (20–60°C) without requiring expensive transition metal removal steps, which is particularly advantageous for pharmaceutical intermediate production where trace metal contamination must be minimized through rigorous quality control protocols.
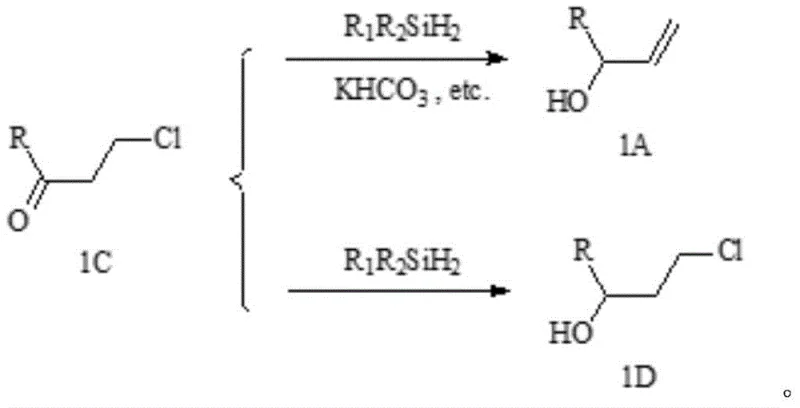
Impurity control is achieved through multiple design elements inherent in this methodology. The stability of the 3-chloropropyl intermediate under non-alkaline conditions prevents premature decomposition during storage or handling, while the bicarbonate promoter suppresses side reactions that typically generate regioisomers or over-reduction products. The reaction’s tolerance for various solvents—including alcohols and ethers—allows optimization for impurity dissolution without compromising yield, and the straightforward aqueous workup effectively removes catalyst residues and inorganic byproducts. This results in crude products with gas chromatography purity exceeding ninety-eight percent that require only minimal crystallization for pharmaceutical-grade specifications, significantly reducing quality control burdens compared to conventional routes that generate complex impurity profiles requiring extensive purification.
How to Synthesize Allyl Alcohol Derivatives Efficiently
This patented process represents a significant advancement in synthetic methodology for producing high-value allyl alcohol derivatives essential in pharmaceutical manufacturing. The two-step sequence begins with commercially available acyl chlorides that undergo ethylene addition under mild Lewis acid catalysis, followed by an iridium-catalyzed reduction step that achieves exceptional selectivity through innovative use of bicarbonate promoters. Detailed standardized synthesis procedures have been developed to ensure consistent quality and yield across production scales. The following section provides a comprehensive step-by-step guide for implementing this methodology in industrial settings, covering critical parameters from reagent selection to purification protocols.
- Prepare the reaction mixture by adding alkali metal bicarbonate, dialkyldihydrosilane, iridium complex catalyst, and alcohol/ether solvent to a nitrogen-purged reactor, maintaining temperature at 20-60°C while slowly introducing the 3-chloropropyl derivative solution to control exothermic gas evolution.
- After complete addition, sustain the reaction temperature for 3-6 hours, then filter the mixture and hydrolyze the filtrate with water at controlled temperature (20-40°C), adjusting pH to 3-6 with acid to isolate the crude product.
- Perform liquid separation and extract the aqueous layer with the reaction solvent, combine organic phases, and concentrate under reduced pressure to obtain high-purity allyl alcohol derivative without additional purification steps.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route delivers substantial value across procurement and supply chain functions by addressing fundamental pain points in traditional manufacturing approaches. The methodology eliminates dependency on unstable intermediates that create supply vulnerabilities while leveraging cost-effective starting materials that enhance sourcing flexibility. By streamlining the production sequence into two high-yielding steps with minimal purification requirements, the process significantly reduces manufacturing complexity and associated operational risks. These advantages translate directly into improved supply chain resilience and cost efficiency—critical factors for procurement teams managing global sourcing strategies for pharmaceutical intermediates where consistent quality and reliable delivery are non-negotiable requirements.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts avoids expensive heavy metal removal processes required in conventional routes, while the use of readily available organic acyl chlorides as starting materials creates substantial cost savings compared to alternative pathways requiring complex aldehyde preparations. The high-yielding two-step sequence minimizes solvent consumption and waste generation through simplified workup procedures, significantly reducing overall manufacturing costs without compromising product quality or purity specifications.
- Enhanced Supply Chain Reliability: The stability of key intermediates under standard storage conditions eliminates supply chain disruptions caused by precursor decomposition during transit or warehousing. Sourcing flexibility is enhanced through multiple viable pathways for acyl chloride procurement from established global suppliers, reducing single-source dependencies that create vulnerability in traditional manufacturing approaches. The robust process design maintains consistent output quality across production scales, ensuring reliable delivery timelines even during peak demand periods without requiring specialized handling or storage conditions.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory to commercial production volumes while maintaining consistent yield and purity profiles, enabling seamless transition from development to full-scale manufacturing without revalidation. The elimination of hazardous reagents and simplified waste streams significantly reduce environmental impact compared to conventional routes involving toxic catalysts or complex purification steps. This environmentally favorable profile aligns with increasing regulatory requirements for sustainable manufacturing practices while supporting corporate ESG initiatives through reduced energy consumption and waste generation.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations regarding implementation of this patented synthesis methodology. These insights are derived directly from patent documentation and experimental validation data, providing evidence-based responses to common concerns raised by technical procurement teams evaluating new manufacturing routes for pharmaceutical intermediates.
Q: How does this method overcome instability issues of α-carbonyl alkene compounds in conventional synthesis?
A: The patented process replaces unstable α-carbonyl alkene compounds with stable 3-chloropropyl derivatives (1C) that maintain integrity under non-alkaline conditions, eliminating self-polymerization risks while achieving >90% yield through the iridium-catalyzed reduction pathway.
Q: What enables the high yield (>90%) in both reaction steps compared to prior art?
A: The synergistic effect of alkali metal bicarbonate as a reaction auxiliary with iridium catalysts enables selective reduction without byproduct formation, while the two-step route from readily available acyl chlorides avoids low-yield aldehyde preparation stages inherent in conventional methods.
Q: How does the use of organic acyl chlorides as starting materials reduce manufacturing costs?
A: Organic acyl chlorides are significantly more cost-effective than corresponding aldehydes or α-carbonyl alkenes, and the elimination of transition metal catalysts avoids expensive heavy metal removal processes, creating substantial cost savings in commercial-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Alcohol Derivatives Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex pharmaceutical intermediates like allyl alcohol derivatives. With stringent purity specifications consistently achieved through rigorous QC labs and process validation protocols, we ensure product quality meets the most demanding regulatory requirements across global markets. Our technical team has successfully implemented this patented methodology across multiple client projects, demonstrating consistent yields exceeding ninety percent while maintaining exceptional impurity profiles suitable for high-value pharmaceutical applications where structural precision is critical for final drug efficacy.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific supply chain requirements. Contact us today to obtain detailed COA data and route feasibility assessments tailored to your production scale and quality specifications.
