Advanced Catalyst-Free Synthesis of 3-Difluoromethyl Coumarins for Commercial Scale-Up
Advanced Catalyst-Free Synthesis of 3-Difluoromethyl Coumarins for Commercial Scale-Up
The introduction of fluorine atoms or fluorine-containing groups into small organic molecules is a cornerstone strategy in modern medicinal chemistry, significantly enhancing lipophilicity, metabolic stability, and cell permeability. Patent CN112321553A discloses a groundbreaking methodology for synthesizing 3-position difluoromethyl substituted coumarin derivatives, a scaffold of immense value in the development of high-purity pharmaceutical intermediates and agrochemical agents. Unlike conventional approaches that rely on costly transition metals or complex multi-step reagent preparations, this invention utilizes [bis(difluoroacetoxy)iodo]benzene as a versatile difluoromethyl source under visible light catalysis. The process generates difluoromethyl radicals that undergo addition, oxidation, and rearrangement with aryl propiolates, yielding the target coumarin structures with high efficiency. This technological leap represents a paradigm shift for manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing, offering a streamlined pathway that bypasses the economic and environmental burdens associated with precious metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-position difluoromethyl substituted coumarins has been plagued by significant operational and economic inefficiencies that hinder large-scale adoption. Traditional protocols often necessitate the use of expensive transition metal photocatalysts, such as fac-Ir(ppy)3, which not only inflate raw material costs but also introduce stringent requirements for heavy metal removal to meet regulatory purity standards. Furthermore, alternative methods utilizing reagents like difluoromethyl benzothiazole sulfone require tedious multi-step syntheses to generate the difluoromethyl source itself, compounding the overall process complexity and waste generation. Other reported strategies involving sodium difluoromethanesulfinate and Eosin Y still depend on precursors that are not atom-economical and require additional synthetic steps. These legacy processes create substantial bottlenecks in supply chain continuity and escalate the cost of goods sold, making them less attractive for the commercial scale-up of complex pharmaceutical intermediates where margin compression is a constant pressure.
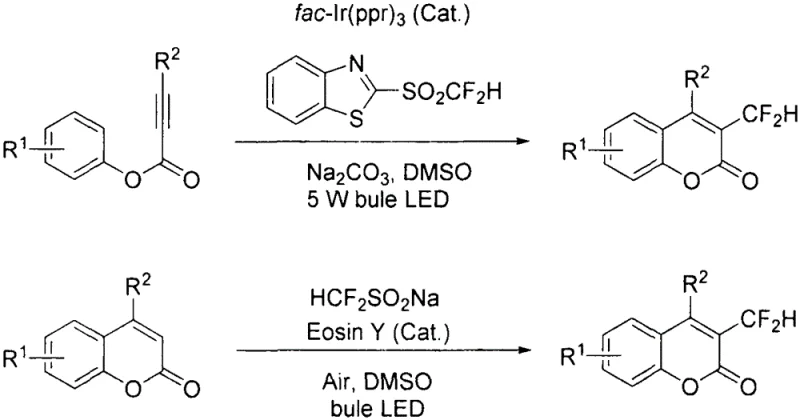
The Novel Approach
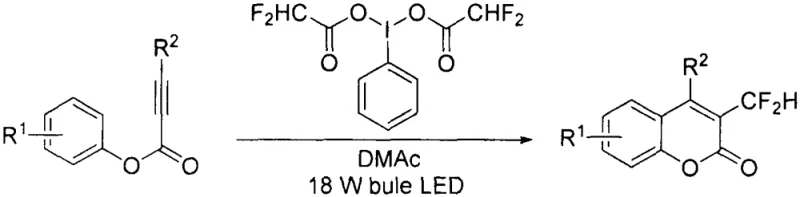
The methodology outlined in patent CN112321553A offers a transformative solution by employing [bis(difluoroacetoxy)iodo]benzene as a dual-function reagent that acts as both the difluoromethyl radical source and the terminal oxidant. This innovation eliminates the absolute necessity for external photocatalysts, whether they are expensive iridium complexes or organic dyes, thereby drastically simplifying the reaction mixture and downstream purification processes. The reaction proceeds smoothly at room temperature under irradiation from a standard blue 18W LED light, utilizing DMAc as the solvent, which indicates a mild and energy-efficient profile suitable for industrial translation. By removing the dependency on multi-step reagent synthesis and precious metal catalysts, this novel approach directly addresses the pain points of procurement managers focused on cost reduction in fine chemical manufacturing. The operational simplicity, combined with the use of readily available starting materials, positions this technology as a superior candidate for establishing a reliable supply chain for high-value fluorinated heterocycles.
Mechanistic Insights into Visible Light Mediated Radical Cyclization
The mechanistic pathway of this transformation is elegantly driven by the homolytic cleavage of the hypervalent iodine reagent under visible light irradiation, which generates the reactive difluoromethyl radical species in situ. Upon exposure to blue LED light, the [bis(difluoroacetoxy)iodo]benzene undergoes excitation and subsequent bond dissociation to release the difluoromethyl radical, which then adds regioselectively to the triple bond of the aryl propiolate substrate. This radical addition step is critical as it sets the stage for the subsequent cyclization, forming a vinyl radical intermediate that is poised for oxidation. The absence of an external photocatalyst suggests that the hypervalent iodine species itself or its decomposition products may facilitate the initial radical generation or participate in an electron transfer process, highlighting the unique redox properties of the reagent system. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters for specific substrate classes, ensuring that the radical flux is maintained at levels that favor productive cyclization over non-specific side reactions.
Following the radical addition, the intermediate undergoes a crucial oxidation step where the remaining iodine species acts as the oxidant, converting the vinyl radical into a cationic species that facilitates the final ring closure and aromatization to form the coumarin core. This intramolecular rearrangement and oxidation sequence is highly efficient, minimizing the formation of byproducts that typically arise from competing radical termination pathways. The dual role of the reagent ensures that the stoichiometry is balanced for both the carbon-fluorine bond formation and the oxidative cyclization, leading to a cleaner impurity profile compared to methods requiring separate oxidants. For quality control teams, this mechanistic feature translates to a more robust process with fewer unknown impurities, simplifying the validation of analytical methods and ensuring that the final high-purity pharmaceutical intermediate meets stringent specifications without extensive recrystallization or chromatographic polishing.
How to Synthesize 3-Difluoromethyl Coumarin Efficiently
The practical execution of this synthesis is designed for reproducibility and safety, leveraging standard laboratory equipment that can be easily scaled to pilot and production vessels. The protocol begins with the rigorous exclusion of moisture and oxygen, utilizing standard Schlenk line techniques to ensure the stability of the radical intermediates and the hypervalent iodine reagent. Reactants are dissolved in polar aprotic solvents like DMAc, which solubilize both the organic substrates and the iodine reagent effectively, creating a homogeneous reaction medium that maximizes photon absorption and radical diffusion. The detailed standardized synthesis steps provided below outline the precise molar ratios, lighting conditions, and workup procedures necessary to achieve optimal yields, serving as a foundational guide for process chemists looking to implement this technology. Adhering to these parameters ensures that the benefits of the catalyst-free system are fully realized, delivering consistent batch-to-batch quality essential for GMP manufacturing environments.
- Prepare an anhydrous and anaerobic environment in a 10mL Schlenk tube by heating and nitrogen purging to remove residual moisture and oxygen.
- Add aryl propiolate derivatives (0.30 mmol) and [bis(difluoroacetoxy)iodo]benzene (0.60 mmol) to the tube, followed by 1.00 mL of anhydrous DMAc solvent under nitrogen protection.
- Stir the mixture at room temperature under irradiation from a blue 18W LED light until TLC confirms completion, then proceed with aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patent offers compelling advantages that directly impact the bottom line and operational resilience of chemical manufacturing operations. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of rare earth metals like Iridium. Additionally, the simplified reagent profile reduces the number of SKUs required for production, streamlining inventory management and reducing the logistical burden on procurement teams. The mild reaction conditions, operating at room temperature with low-energy LED lighting, contribute to a lower carbon footprint and reduced utility costs, aligning with corporate sustainability goals while enhancing overall process economics. These factors collectively create a more agile and cost-effective supply chain capable of responding rapidly to market demands for fluorinated building blocks.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive photocatalysts such as Iridium complexes, which traditionally account for a disproportionate share of raw material costs in photochemical processes. By utilizing [bis(difluoroacetoxy)iodo]benzene, which can be synthesized in a single step from commodity chemicals, the process significantly lowers the direct material cost per kilogram of product. Furthermore, the dual functionality of the reagent eliminates the need for purchasing separate oxidants, reducing the total mass of reagents required and consequently lowering waste disposal costs. This lean reagent strategy allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy margins, providing a distinct advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as aryl propiolates and simple iodine precursors, ensures a robust and continuous supply of inputs, minimizing the risk of production stoppages due to raw material shortages. Unlike methods requiring specialized, multi-step synthesized reagents like difluoromethyl benzothiazole sulfone, the reagents for this process are accessible through standard chemical supply chains with short lead times. This accessibility empowers supply chain heads to negotiate better terms with vendors and maintain lower safety stock levels, freeing up working capital. The stability of the key reagent also simplifies storage and handling requirements, reducing the need for specialized containment infrastructure and further enhancing operational reliability.
- Scalability and Environmental Compliance: The operational simplicity of running reactions at room temperature under LED irradiation makes this process inherently scalable, avoiding the engineering challenges associated with high-pressure or high-temperature reactors. The absence of heavy metal catalysts significantly simplifies the wastewater treatment process, as there is no need for complex metal scavenging or recovery systems to meet environmental discharge limits. This reduction in environmental compliance burden accelerates the timeline for regulatory approvals and plant audits, facilitating faster time-to-market for new products. Moreover, the atom economy of the reaction is improved by the dual-role reagent, resulting in less chemical waste generation per unit of product, which aligns with green chemistry principles and reduces the overall environmental impact of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and process capabilities. These insights are intended to clarify the operational feasibility and strategic value of adopting this catalyst-free route for the production of fluorinated coumarins. By understanding the nuances of reagent stability, substrate scope, and purification requirements, stakeholders can make informed decisions about integrating this method into their existing manufacturing portfolios. The answers provided reflect the rigorous data and experimental results documented in the intellectual property, ensuring accuracy and reliability for technical evaluation.
Q: What are the primary advantages of this synthesis method over traditional transition metal catalysis?
A: This method eliminates the need for expensive transition metal photocatalysts like Iridium complexes or organic dyes like Eosin Y. Furthermore, the difluoromethylating reagent [bis(difluoroacetoxy)iodo]benzene serves a dual role as both the radical source and the oxidant, simplifying the reaction system and reducing waste generation significantly.
Q: Is the difluoromethylating reagent stable and easy to source for large-scale production?
A: Yes, [bis(difluoroacetoxy)iodo]benzene is chemically stable and can be conveniently prepared in a single step from iodobenzene diacetate and difluoroacetic acid. This accessibility ensures a reliable supply chain for commercial manufacturing without the multi-step synthesis required for precursors like difluoromethyl benzothiazole sulfone.
Q: What types of substituents are tolerated on the aryl alkynoate substrate?
A: The reaction demonstrates broad substrate scope, tolerating various functional groups including cyano, phenyl, methoxy, halogens (fluorine, chlorine, bromine), and tert-butyl groups at the R1 position, as well as cyclopropyl and substituted phenyl groups at the R2 position, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Difluoromethyl Coumarin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalyst-free visible light synthesis described in patent CN112321553A for producing high-value fluorinated intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this innovative chemistry are fully translated into industrial reality. Our state-of-the-art facilities are equipped with advanced photochemical reactors capable of precise wavelength control, allowing us to optimize the LED irradiation parameters for maximum efficiency and yield. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-difluoromethyl coumarin derivatives meets the exacting standards required by global pharmaceutical and agrochemical clients, delivering consistency and quality in every shipment.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can enhance your product portfolio and improve your supply chain economics. By engaging with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you navigate the complexities of fluorine chemistry and secure a reliable, scalable, and economically superior source for your critical intermediates, driving your projects forward with confidence and speed.
