Revolutionizing C2-Oxygenated Indole Synthesis: A Green, Metal-Free Protocol for Commercial Scale-Up
Introduction to Advanced C-O Bond Construction Technology
The landscape of organic synthesis is continuously evolving towards greener, more atom-economical processes, particularly in the realm of pharmaceutical intermediate manufacturing. Patent CN110922350B, published in September 2021, introduces a groundbreaking methodology for the construction of C-O bonds at the C2 position of N-acetyl-3-oxyindole compounds. This technology addresses a critical gap in the functionalization of indole derivatives, which are ubiquitous scaffolds in bioactive molecules. As illustrated in the structural diversity of natural products below, the C2-oxygenated indole motif is a privileged structure found in potent antitumor antibiotics such as Mitomycin C, Mitomycin K, Cephalinone B, and Mersicarpine.
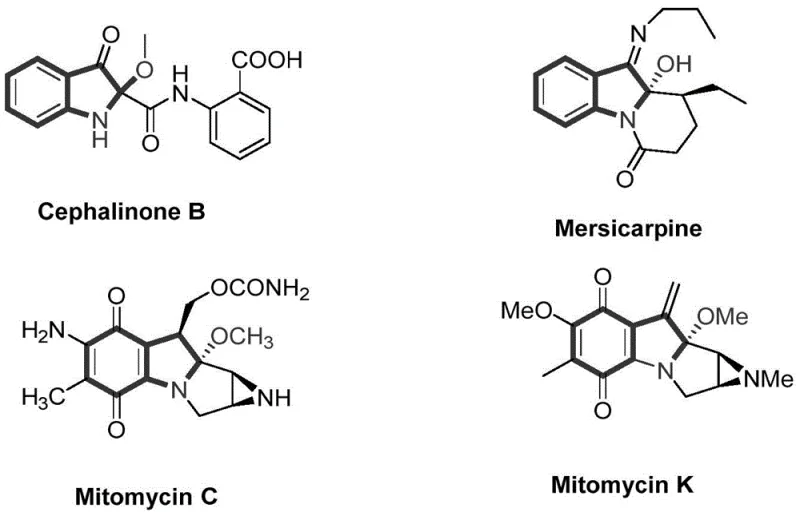
The significance of this patent lies in its departure from traditional transition-metal catalysis, offering a metal-free alternative that aligns perfectly with modern green chemistry principles. By utilizing a simple iodide/oxidant system, this invention enables the direct acyloxylation of the C2 position under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift away from complex ligand design and expensive metal removal steps, paving the way for more streamlined supply chains and reduced environmental footprints in the production of high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of indole C-H bonds into C-O bonds has relied heavily on transition metal catalysis, which presents significant challenges for industrial scalability and cost efficiency. Previous literature, such as the work by Koley et al. in 2018, utilized copper(I) catalysts coupled with benzoic acid, yet suffered from low yields ranging between 35% and 55%, alongside the inherent toxicity and removal difficulties associated with copper residues. Similarly, Wang's development of Rh(II)-catalyzed selective C7-hydroxylation required stoichiometric amounts of PhI(OAc)2 and expensive rhodium metals, creating a substantial economic barrier for large-scale application.
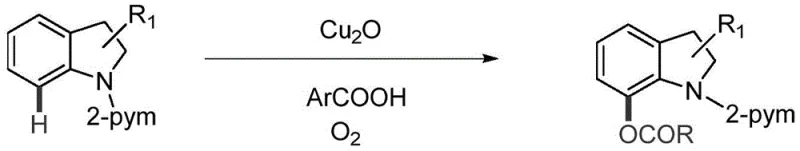
Furthermore, other reported methods involving palladium catalysts, such as those by Lei, Kwong, and Suna, or cobalt-catalyzed systems reported by Liu, invariably introduce heavy metal contaminants that necessitate rigorous purification protocols to meet stringent pharmaceutical purity specifications. These conventional routes often demand harsh reaction conditions, elevated temperatures, or inert atmospheres, which increase energy consumption and operational complexity. The reliance on precious metals not only inflates raw material costs but also introduces supply chain vulnerabilities related to the availability of these critical elements, making them less ideal for the robust manufacturing of reliable pharmaceutical intermediate supplier networks.
The Novel Approach
In stark contrast to these metal-dependent methodologies, the novel approach disclosed in CN110922350B employs a catalytic system based on potassium iodide (KI) and hydrogen peroxide (H2O2). This metal-free protocol achieves the direct acyloxylation at the C2 position of N-acetyl-3-oxindoles with exceptional efficiency and selectivity. The general reaction scheme, depicted below, highlights the simplicity of transforming Formula (1) and Formula (2) into the desired Formula (3) without the need for complex ligands or noble metals.
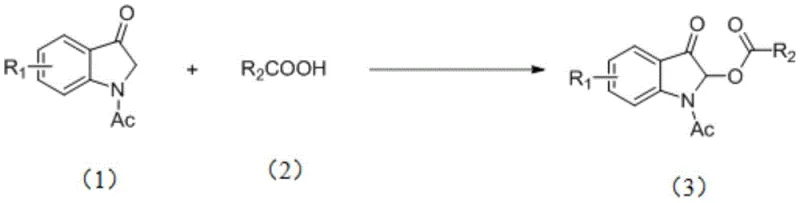
This innovative strategy operates at room temperature, drastically reducing energy requirements compared to thermal activation methods. The use of ethyl acetate as a preferred solvent further enhances the green profile of the process, replacing hazardous chlorinated solvents often used in traditional oxidations. By avoiding transition metals entirely, this method eliminates the risk of metal leaching into the final product, thereby simplifying the downstream purification process and ensuring compliance with strict regulatory limits on heavy metal impurities in active pharmaceutical ingredients (APIs).
Mechanistic Insights into Iodide-Catalyzed Acyloxylation
The mechanistic pathway of this transformation is believed to proceed through a radical-mediated oxidation cycle initiated by the interaction between the iodide catalyst and the peroxide oxidant. In the presence of hydrogen peroxide, the iodide species is oxidized to generate reactive iodine intermediates, which subsequently activate the carboxylic acid to form an acyloxy radical or a hypervalent iodine species capable of attacking the electron-rich C2 position of the oxindole ring. This radical mechanism allows for the cleavage of the C-H bond and the formation of the new C-O bond with high regioselectivity, specifically targeting the C2 position adjacent to the carbonyl group.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining a clean reaction profile. Unlike strong acidic or basic conditions that might lead to hydrolysis of the N-acetyl group or ring-opening of the oxindole scaffold, this neutral to slightly acidic environment preserves the integrity of the sensitive heterocyclic core. The absence of transition metals also precludes the formation of metal-coordinated side products or oligomerization pathways often observed in Pd or Cu-catalyzed reactions. Consequently, the crude reaction mixture typically contains fewer byproducts, facilitating easier isolation of the target compound through standard silica gel chromatography or crystallization techniques.
How to Synthesize N-Acetyl-3-Oxyindole Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The process involves dissolving the N-acetylindole-3-one substrate and the chosen carboxylic acid in ethyl acetate, followed by the sequential addition of the KI catalyst and H2O2 oxidant. The reaction proceeds smoothly at ambient temperature, requiring only simple stirring to reach completion. Detailed standardized synthetic steps, including specific molar ratios and workup procedures derived from the patent examples, are provided in the guide below to assist process engineers in replicating this high-yielding transformation.
- Dissolve N-acetylindole-3-one (Formula 1) and the corresponding carboxylic acid (Formula 2) in ethyl acetate solvent within a reaction vessel.
- Add potassium iodide (KI) as the catalyst (5 mol%) and hydrogen peroxide (H2O2, 1.5 equivalents) as the oxidant to the mixture at room temperature.
- Stir the reaction mixture for approximately 5 hours at room temperature, then perform standard workup including extraction with ethyl acetate and silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free technology offers tangible strategic advantages that extend beyond mere chemical novelty. The primary value proposition lies in the drastic simplification of the raw material portfolio and the elimination of cost drivers associated with precious metal catalysis. By shifting from palladium or rhodium-based systems to a potassium iodide framework, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing without compromising on yield or purity standards.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with commodity-grade potassium iodide results in a substantial decrease in raw material expenditure. Furthermore, the elimination of metal scavenging resins and specialized filtration equipment required to remove trace metals from the final product reduces both capital expenditure and operating costs. The high atom economy of using hydrogen peroxide, which generates water as the only byproduct, minimizes waste disposal fees and aligns with sustainability goals, leading to overall lower production costs per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Relying on globally available and stable reagents like KI and H2O2 mitigates the supply risks associated with geopolitically sensitive precious metals. This ensures a more robust and continuous supply chain, reducing the likelihood of production delays due to catalyst shortages. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for specialized corrosion-resistant alloys, enhancing equipment utilization rates and flexibility across different production campaigns.
- Scalability and Environmental Compliance: The protocol's operation at room temperature and atmospheric pressure significantly lowers the energy intensity of the process, contributing to a reduced carbon footprint. The use of ethyl acetate, a solvent with favorable toxicological and environmental profiles, simplifies solvent recovery and recycling operations. This green chemistry approach not only facilitates easier regulatory approval but also positions the manufacturing site as a leader in sustainable chemical production, appealing to environmentally conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation of this novel synthesis route, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent documentation. These insights cover critical aspects such as substrate compatibility, reaction optimization, and product characterization, serving as a quick reference for technical teams evaluating the feasibility of this process for their specific projects.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes potassium iodide (KI) as a cheap, non-toxic catalyst and hydrogen peroxide as a green oxidant, completely eliminating the need for costly palladium, rhodium, or copper catalysts often found in conventional methods.
Q: What is the substrate scope for the carboxylic acid component in this reaction?
A: The method demonstrates excellent functional group tolerance, successfully reacting with aromatic carboxylic acids (including substituted benzoic acids), heterocyclic acids (pyrrole, thiophene, furan), aliphatic acids, and unsaturated acids like cinnamic acid.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction proceeds at room temperature without the need for large-scale instruments or harsh conditions, and the use of common solvents like ethyl acetate facilitates easy scale-up and downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetyl-3-Oxyindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free C-H functionalization technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory protocols like the one described in CN110922350B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acetyl-3-oxyindole derivatives delivered meets the highest quality standards required by global regulatory agencies.
We invite you to collaborate with our technical procurement team to explore how this cost-effective and environmentally friendly synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the efficient and sustainable manufacturing of complex pharmaceutical intermediates.
