Advancing Chiral Amino Acid Production with Novel 2,4-Diaminopyridine Nitroxide Catalysts
Advancing Chiral Amino Acid Production with Novel 2,4-Diaminopyridine Nitroxide Catalysts
The landscape of asymmetric synthesis is continually evolving, driven by the demand for more efficient and selective catalytic systems. Patent CN111943929A introduces a groundbreaking class of chiral 2,4-diaminopyridine nitroxide catalysts that redefine the standards for acyl transfer reactions. This technology specifically targets the ring-opening of azlactones with alcohols, a critical transformation in the production of optically pure alpha-amino acid derivatives. By shifting the nucleophilic center from the pyridine nitrogen to the N-oxide oxygen, this innovation overcomes historical limitations associated with steric hindrance in C2-substituted pyridine catalysts. For R&D directors and process chemists, this represents a significant leap forward in accessing high-value chiral building blocks with exceptional enantiomeric excess.
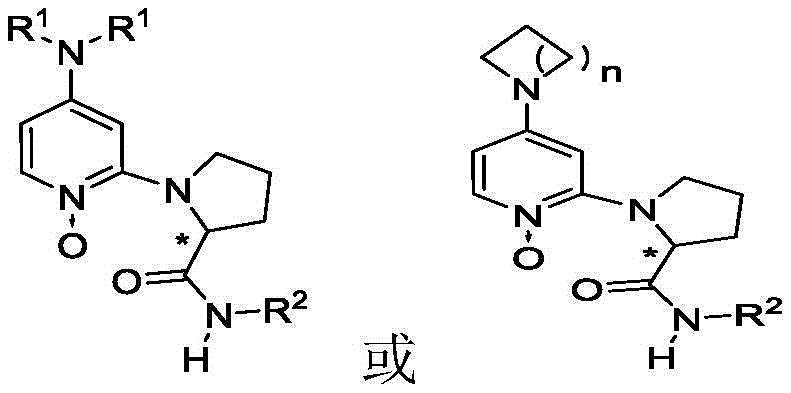
The core innovation lies in the unique structural architecture of the catalyst, which features a pyridine N-oxide core substituted at the 2 and 4 positions with amino groups. This configuration allows the oxygen atom of the N-oxide to act as the primary nucleophilic site, effectively distancing the bulky chiral auxiliary from the reactive center. Such a design minimizes the steric repulsion that typically plagues traditional 2-substituted DMAP catalysts, thereby enhancing catalytic turnover and enabling the use of smaller, more economical alcohol nucleophiles. This structural refinement is pivotal for achieving high yields and superior stereocontrol in the synthesis of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral acyl transfer catalysts has been dominated by DMAP derivatives where the chiral information is encoded at the C2, C3, or C4 positions of the pyridine ring. However, literature reports, including seminal work by Vedejs and Chen, have highlighted a critical flaw in 2-substituted DMAP catalysts: severe steric hindrance between the C2 chiral group and the N-acyl intermediate. This spatial conflict drastically reduces catalytic activity, often necessitating the use of stoichiometric amounts of the catalyst rather than sub-stoichiometric loading. Furthermore, existing technologies frequently require bulky, expensive alcohols like alpha-naphthalene-2-methanol to achieve acceptable selectivity, limiting their utility in cost-sensitive industrial applications.
The Novel Approach
The novel approach presented in this patent circumvents these steric barriers by converting the pyridine motif into a pyridine N-oxide. In this system, the nucleophilic attack is initiated by the oxygen atom of the N-oxide rather than the ring nitrogen. This subtle yet profound change relocates the acylation site away from the crowded C2 environment, restoring high catalytic activity even with small alcohol nucleophiles like methanol. The result is a versatile catalytic system capable of processing a wide range of azlactone substrates, including those with significant steric bulk at the C4 position, which were previously considered challenging or unreactive under standard conditions.

Mechanistic Insights into N-Oxide Catalyzed Acyl Transfer
The mechanistic pathway of this transformation relies on the enhanced nucleophilicity of the pyridine N-oxide oxygen. Upon interaction with the azlactone substrate, the catalyst forms a reactive acyl-pyridinium intermediate. Because the chiral environment is established by the 2,4-diamino substitution pattern, the subsequent nucleophilic attack by the alcohol occurs with high facial selectivity. This dynamic kinetic resolution process ensures that the racemic starting material is efficiently converted into a single enantiomer of the alpha-amino acid derivative. The presence of the N-oxide functionality also modulates the electronic properties of the pyridine ring, facilitating faster turnover rates compared to neutral pyridine analogues.
Impurity control is another critical aspect addressed by this mechanistic design. The high stereoselectivity inherent in the catalyst structure minimizes the formation of the undesired enantiomer, often achieving enantiomeric excess (ee) values exceeding 90% and up to 96% in optimized conditions. This level of purity is essential for pharmaceutical applications where regulatory standards demand strict control over chiral impurities. Additionally, the catalyst's stability under mild reaction conditions prevents degradation pathways that could lead to side products, ensuring a clean reaction profile that simplifies downstream purification processes.
How to Synthesize Chiral Amino Acid Derivatives Efficiently
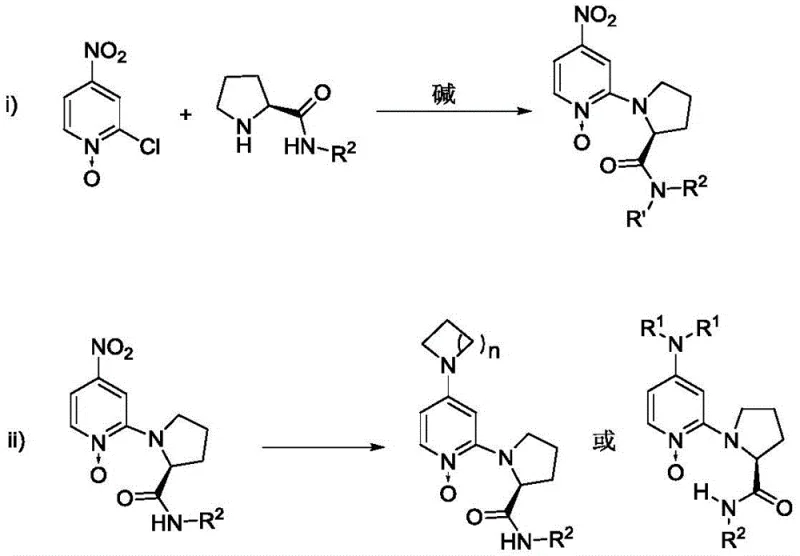
The synthesis of target chiral amino acid derivatives using this technology follows a streamlined protocol designed for operational simplicity and robustness. The process begins with the preparation of the catalyst itself, which involves a two-step substitution reaction starting from commercially available 2-chloro-4-nitropyridine N-oxide and chiral prolinamide derivatives. Once the catalyst is secured, the ring-opening reaction is conducted in common organic solvents such as dichloromethane or tetrahydrofuran. The detailed standardized synthesis steps for implementing this methodology in your laboratory are outlined below.
- Prepare the reaction mixture by combining racemic azlactone, the chiral 2,4-diaminopyridine nitroxide catalyst, and benzoic acid additive in an organic solvent such as dichloromethane.
- Introduce the alcohol nucleophile, typically methanol or other lower alcohols, to the stirred mixture at room temperature or slightly elevated temperatures ranging from -20°C to 35°C.
- Monitor the reaction progress until completion, then isolate the chiral alpha-amino acid derivative through vacuum concentration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible strategic benefits beyond mere chemical efficiency. The ability to utilize simple, low-cost alcohols like methanol instead of specialized, bulky nucleophiles significantly reduces raw material expenses. Furthermore, the catalyst's high activity allows for lower loading levels, decreasing the overall consumption of the chiral ligand. These factors combine to drive down the cost of goods sold (COGS) for the final amino acid intermediates, making the process economically viable for large-scale manufacturing without compromising on quality or selectivity.
- Cost Reduction in Manufacturing: The elimination of expensive, sterically hindered alcohols and the reduction in catalyst loading directly translate to substantial cost savings. By avoiding the need for stoichiometric amounts of catalyst, which was a limitation of previous 2-substituted DMAP systems, the process becomes far more resource-efficient. Additionally, the use of benign solvents and mild reaction conditions reduces energy consumption and waste disposal costs, contributing to a leaner and more sustainable manufacturing budget.
- Enhanced Supply Chain Reliability: The starting materials for both the catalyst and the reaction substrates are readily available commodity chemicals. 2-Chloro-4-nitropyridine N-oxide and prolinamide derivatives are produced by multiple global suppliers, mitigating the risk of supply chain disruptions. This abundance ensures consistent availability of key inputs, allowing for reliable production scheduling and reducing the lead time for high-purity pharmaceutical intermediates required by downstream API manufacturers.
- Scalability and Environmental Compliance: The reaction operates effectively at ambient or slightly elevated temperatures, removing the need for extreme cooling or heating infrastructure. This thermal flexibility simplifies the engineering requirements for scale-up from gram to tonnage quantities. Moreover, the high selectivity of the reaction minimizes the generation of hazardous by-products, aligning with increasingly stringent environmental regulations and facilitating easier waste stream management in commercial facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What distinguishes this nitroxide catalyst from traditional DMAP catalysts?
A: Unlike traditional DMAP catalysts that utilize the pyridine nitrogen as the nucleophilic site and suffer from steric hindrance at the C2 position, this novel catalyst utilizes the oxygen atom of the pyridine N-oxide as the nucleophile. This structural shift significantly reduces steric clashes, allowing for higher catalytic activity and the use of simpler, less bulky alcohol nucleophiles.
Q: Can this catalyst handle sterically hindered azlactone substrates?
A: Yes, the catalyst demonstrates robust performance even with challenging substrates such as C4-isopropyl and C4-phenyl substituted azlactones, which typically yield only trace products with conventional catalytic systems. It achieves high yields and excellent enantioselectivity for these difficult cases.
Q: Is the catalyst synthesis scalable for industrial production?
A: The synthesis involves a straightforward two-step substitution reaction using readily available raw materials like 2-chloro-4-nitropyridine N-oxide and chiral prolinamide. The process avoids complex transition metal chemistry, making it highly suitable for commercial scale-up and cost-effective manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Diaminopyridine Nitroxide Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of catalyst or intermediate meets the highest international standards, providing our partners with the confidence needed for regulatory filings.
We invite you to collaborate with us to explore the full capabilities of this novel catalytic system for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your production economics.
