Advanced Boron-Catalyzed Synthesis of Vildagliptin for Commercial Scale-up
The pharmaceutical landscape for Type 2 diabetes treatment continues to evolve, with Vildagliptin remaining a cornerstone therapeutic agent due to its efficacy as a DPP-IV inhibitor. As global demand for this active pharmaceutical ingredient (API) surges, the industry faces increasing pressure to optimize synthetic routes that balance high purity with economic viability. Patent CN108658826B introduces a transformative methodology that addresses long-standing challenges in vildagliptin synthesis, specifically targeting the elimination of complex purification steps and hazardous reagents. This technical insight report analyzes the proprietary boron-catalyzed pathway disclosed in the patent, offering a strategic roadmap for R&D directors and procurement specialists seeking a reliable vildagliptin intermediate supplier. By shifting away from traditional carbodiimide-mediated couplings, this innovation promises substantial cost reduction in API manufacturing while ensuring the stringent quality standards required for regulatory approval.
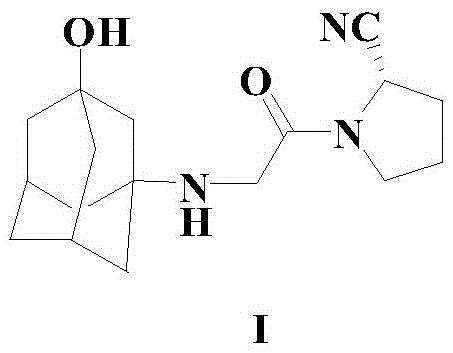
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vildagliptin has been plagued by low yields and difficult purification profiles associated with early-generation methodologies. As illustrated in prior art such as WO2010022690 and WO2013179300, conventional routes often rely on the condensation of L-proline derivatives followed by dehydration to generate key intermediates like (S)-2-cyano-1-(2-chloroacetyl)tetrahydropyrrole. The critical flaw in this approach lies in the subsequent reaction with 3-hydroxyadamantane, where the presence of multiple reactive sites on the adamantane scaffold leads to significant byproduct formation. Specifically, the nucleophilic attack can occur at the nitrogen or the oxygen atom, resulting in disubstituted byproducts (Compound IV) and O-substituted ethers (Compound V), which drastically lower the overall yield and complicate isolation.
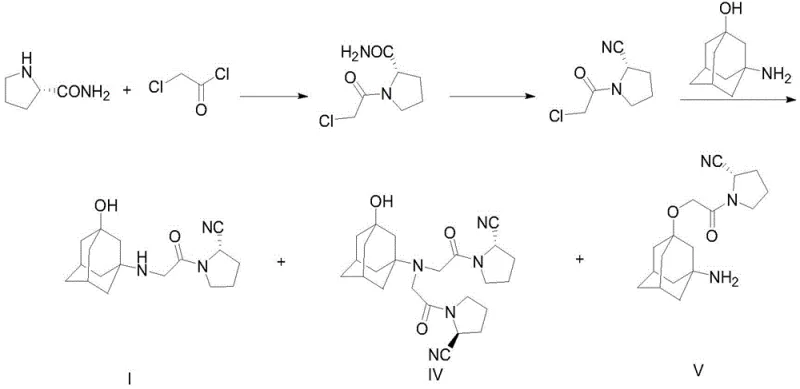
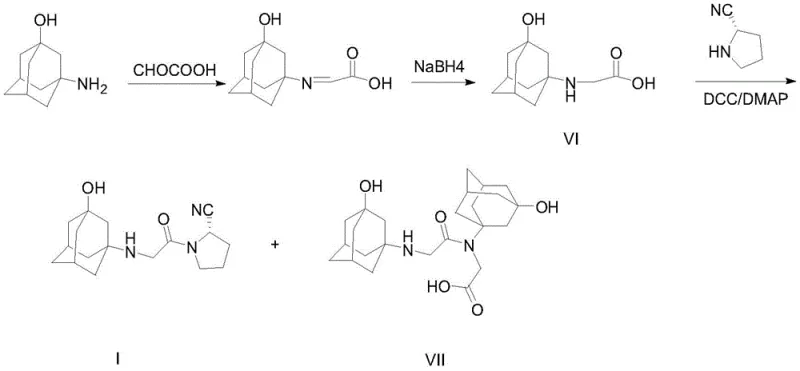
Furthermore, alternative strategies described in WO2011101861 attempt to utilize 3-hydroxyadamantane acetic acid derivatives (Compound VI) coupled with pyrrolidine nitriles. However, these methods frequently suffer from incomplete reactions and the generation of polypeptide condensation impurities (Compound VII). The reliance on harsh coupling conditions often necessitates the use of expensive reagents and generates substantial chemical waste, creating bottlenecks for supply chain heads concerned with environmental compliance and raw material availability. These legacy processes inherently lack the atom economy and selectivity required for modern, sustainable pharmaceutical manufacturing.
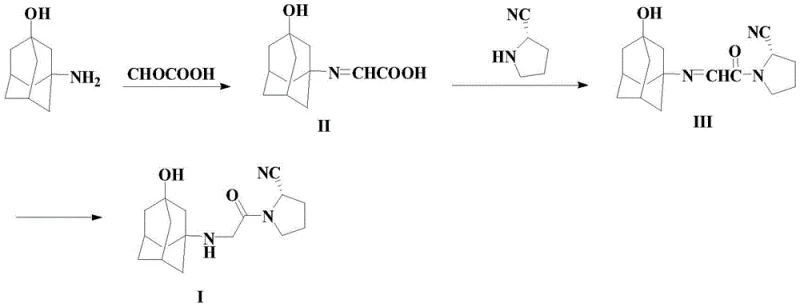
The Novel Approach
In stark contrast to these cumbersome legacy methods, the methodology outlined in Patent CN108658826B presents a streamlined, three-step sequence that fundamentally reimagines the bond construction strategy. The process initiates with the efficient imidization of 3-amino-1-adamantanol and glyoxylic acid to form a stable imino-acetic acid intermediate (Compound II). This intermediate then undergoes a highly selective acylation with (S)-pyrrolidine-2-carbonitrile, catalyzed by specialized boron species such as tris(2,2,2-trifluoroethyl) borate. This catalytic system effectively activates the carboxylic acid moiety without generating the stubborn urea byproducts typical of DCC or EDC coupling agents. The final step involves a mild reduction of the imine bond to yield the target vildagliptin (Compound I) with exceptional stereochemical integrity.
Mechanistic Insights into Boron-Catalyzed Amidation
The core innovation of this patent lies in the sophisticated application of Lewis acidic boron catalysts to drive the amidation reaction. Unlike traditional peptide coupling reagents that activate the acid through unstable O-acylisourea intermediates, the boron catalyst—specifically electron-withdrawing variants like 2,4-bis(trifluoromethyl)phenylboronic acid or tris(2,2,2-trifluoroethyl) borate—coordinates with the carbonyl oxygen of the imino-acetic acid. This coordination increases the electrophilicity of the carbonyl carbon, facilitating a nucleophilic attack by the amine of the pyrrolidine nitrile. The electron-withdrawing nature of the trifluoromethyl groups on the catalyst is crucial, as it enhances the Lewis acidity, thereby accelerating the reaction rate and ensuring high conversion even under reflux conditions in solvents like toluene or cyclopentyl methyl ether.
From an impurity control perspective, this mechanism offers a distinct advantage by suppressing side reactions. The mild activation provided by the boron catalyst prevents the over-activation that often leads to racemization or self-condensation of the amino acid components. Furthermore, the subsequent reduction step, whether performed using a palladium carbon/ammonium formate system or hydrazine hydrate, is highly chemoselective for the C=N double bond. This selectivity ensures that the sensitive nitrile group and the adamantane cage remain intact, resulting in a crude product profile that is remarkably clean. For R&D teams, this translates to a simplified purification workflow, often requiring only crystallization rather than extensive chromatographic separation, which is a critical factor in maintaining high-purity vildagliptin specifications.
How to Synthesize Vildagliptin Efficiently
The synthesis protocol detailed in the patent provides a robust framework for producing vildagliptin with high efficiency and minimal environmental impact. The process begins with the condensation of 3-amino-1-adamantanol and glyoxylic acid monohydrate in a solvent such as tetrahydrofuran or dichloromethane, utilizing acetic anhydride as a dehydrating agent to form the imino-intermediate. Following isolation, this intermediate is subjected to the key boron-catalyzed amidation with (S)-pyrrolidine-2-carbonitrile. The reaction is typically conducted under reflux with a water separator to drive the equilibrium forward. Finally, the resulting imine-amide is reduced to the secondary amine using a transfer hydrogenation system. For a comprehensive, step-by-step technical guide including precise molar ratios and temperature profiles, please refer to the standardized synthesis instructions below.
- Condense 3-amino-1-adamantanol with glyoxylic acid using acetic anhydride to form the imino-acetic acid intermediate.
- Perform acylation with (S)-pyrrolidine-2-carbonitrile using tris(2,2,2-trifluoroethyl) borate or electron-withdrawing phenylboronic acid catalysts.
- Reduce the resulting imine-amide intermediate using palladium carbon/ammonium formate or hydrazine hydrate to yield high-purity vildagliptin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this boron-catalyzed route represents a significant opportunity to optimize the cost structure of vildagliptin production. By eliminating the need for expensive and hazardous coupling reagents like EDC, HOBt, or sodium pyrosulfite, the process inherently reduces the raw material cost per kilogram. Moreover, the avoidance of chloroacetyl intermediates removes the requirement for handling corrosive acid chlorides, thereby lowering safety compliance costs and reducing the need for specialized containment equipment. The simplified workup procedures, which rely on crystallization rather than complex extractions or column chromatography, further contribute to substantial cost savings in labor and solvent consumption.
- Cost Reduction in Manufacturing: The transition to a boron-catalyzed system eliminates the generation of stoichiometric amounts of urea waste associated with carbodiimide couplings. This reduction in waste volume directly lowers disposal costs and improves the overall mass balance of the process. Additionally, the use of commercially available and inexpensive catalysts like tris(2,2,2-trifluoroethyl) borate ensures that the catalytic cycle remains economically viable even at large scales, driving down the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials such as 3-amino-1-adamantanol and glyoxylic acid mitigates the risk of supply disruptions often associated with specialized, custom-synthesized intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, ensures consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The one-pot nature of certain steps and the use of green reduction methods (such as ammonium formate transfer hydrogenation) align with modern green chemistry principles. This not only simplifies the scale-up from pilot plant to commercial manufacturing but also ensures compliance with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical advantages of the boron-catalyzed method.
Q: How does the boron-catalyzed method improve purity compared to traditional EDC/HOBt coupling?
A: The boron-catalyzed method avoids the use of carbodiimide coupling agents like EDC or DCC, which often generate difficult-to-remove urea byproducts. By utilizing electron-withdrawing boron catalysts, the reaction achieves high selectivity for the amide bond without self-condensation impurities, significantly simplifying downstream purification.
Q: What are the primary impurities avoided in this new synthetic route?
A: Traditional routes involving chloroacetyl intermediates often suffer from disubstitution on the adamantane nitrogen and O-substitution on the hydroxyl group. This new route bypasses the reactive chloroacetyl species entirely, preventing the formation of disubstituted byproducts and ether-linked impurities.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes readily available raw materials like 3-amino-1-adamantanol and glyoxylic acid, operates under relatively mild conditions, and employs a one-pot strategy that reduces solvent usage and operational complexity, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vildagliptin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of vildagliptin intermediate meets the exacting standards required for downstream API synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your competitive position in the global diabetes care market.
