Advanced Synthesis of 6-Amino-7-Hydroxyl-3,4-Dihydroquinazoline-4-Ketone for Global TKI Production
The pharmaceutical industry's relentless pursuit of more efficient oncology treatments has placed a premium on the availability of high-quality tyrosine kinase inhibitor (TKI) intermediates. Patent CN103288757A introduces a groundbreaking preparation method for 6-amino-7-hydroxyl-3,4-dihydroquinazoline-4-ketone, a pivotal building block for next-generation anticancer drugs such as Afatinib, Canertinib, and Dacomitinib. This innovation addresses long-standing challenges in organic synthesis by replacing harsh, non-selective nitration conditions with a sophisticated solid-acid catalyzed system. By fundamentally altering the reaction pathway, this technology not only enhances the chemical purity of the final intermediate but also drastically simplifies the manufacturing workflow, offering a compelling value proposition for global supply chains seeking reliability and cost-efficiency in the production of complex heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazoline derivatives bearing both amino and hydroxyl substituents has been plagued by significant chemoselectivity issues. Traditional methodologies typically rely on mixed acid nitration systems, comprising concentrated nitric and sulfuric acids, which are notoriously aggressive oxidizing environments. In the presence of such harsh reagents, the electron-rich amino and phenolic groups on the starting aromatic ring are highly susceptible to unwanted oxidation, leading to the formation of tars, quinones, and other intractable byproducts. To circumvent these destructive side reactions, conventional routes are forced to incorporate multiple protection and deprotection steps, wherein sensitive functional groups are temporarily masked. This multi-step detour not only consumes additional expensive reagents and solvents but also inevitably depresses the overall yield due to material losses at each isolation stage, while simultaneously generating substantial volumes of hazardous chemical waste that complicate environmental compliance.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN103288757A achieves a paradigm shift by enabling direct nitration without the need for functional group protection. As illustrated in the reaction scheme below, the process utilizes specific solid acid catalysts, such as trichloro-triazine (TCT) or aluminum dihydrogen phosphate, in conjunction with nitrating agents like zinc nitrate or concentrated nitric acid. This catalytic system modulates the electrophilicity of the nitrating species, ensuring that substitution occurs selectively at the desired position on the aromatic ring while leaving the sensitive amino and hydroxyl moieties intact. By bypassing the protection-deprotection cycle entirely, the synthetic route is condensed into a streamlined sequence of nitration, reduction, and cyclization. This direct approach not only maximizes atom economy but also significantly shortens the production timeline, providing a robust and economically superior alternative for the industrial manufacture of TKI intermediates.
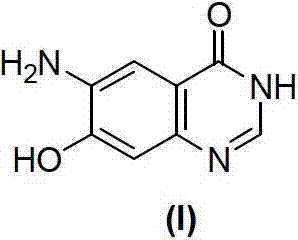
Mechanistic Insights into Solid-Acid Catalyzed Selective Nitration
The core mechanistic advantage of this patented process lies in the unique interaction between the solid acid catalyst and the nitrating agent within the reaction medium. When catalysts like trichloro-triazine (TCT) are employed, they act as mild dehydrating agents and Lewis acids that facilitate the generation of the active nitronium ion or its equivalent in a controlled manner. Unlike the chaotic protonation environment created by sulfuric acid, the solid acid surface or dissolved species provides a structured microenvironment that stabilizes the transition state for electrophilic aromatic substitution. This stabilization lowers the activation energy for the desired nitration while raising the barrier for oxidative degradation of the phenol and aniline groups. Consequently, the reaction proceeds with high regioselectivity at the position ortho to the hydroxyl group, driven by the directing effects of the existing substituents, but without the over-oxidation that typically plagues free phenol nitration. This precise control over reaction kinetics is what allows the process to tolerate unprotected functional groups that would otherwise be destroyed.
Furthermore, the choice of solvent plays a critical role in maintaining the integrity of the intermediates throughout the transformation. The patent specifies the use of polar aprotic solvents like acetonitrile or even water, which are compatible with the solid acid catalysts and help to dissipate the heat of reaction effectively. In traditional mixed acid processes, the exotherm is often difficult to control, leading to thermal runaways that exacerbate side reactions. Here, the milder conditions, often conducted at room temperature (20-30°C), ensure that the intermediate nitro-compounds remain stable prior to the subsequent reduction step. The reduction itself, converting the nitro group to an amino group, and the final cyclization to form the quinazolinone ring are standard transformations, but their efficiency is vastly improved because the starting material for these steps is of significantly higher purity. This cascade of high-fidelity reactions ensures that the final product, 6-amino-7-hydroxyl-3,4-dihydroquinazoline-4-ketone, meets the stringent purity specifications required for downstream API synthesis.
How to Synthesize 6-Amino-7-Hydroxyl-3,4-Dihydroquinazoline-4-Ketone Efficiently
The implementation of this synthesis strategy requires careful attention to the stoichiometry of the catalyst and the choice of the nitrating source to maximize yield and minimize impurities. The process begins with the suspension of the starting material, such as 3-amino-4-hydroxybenzoic acid or its esters, in a suitable solvent like acetonitrile. A catalytic amount of trichloro-triazine (typically 1-5% molar equivalent) is added, followed by the slow introduction of the nitrating agent, such as zinc nitrate or concentrated nitric acid, while maintaining the temperature within the ambient range to prevent thermal stress on the molecule. Following the completion of the nitration, indicated by TLC or HPLC monitoring, the reaction mixture is worked up to isolate the nitro-intermediate, which is then subjected to reduction conditions to generate the diamine precursor. Finally, the cyclization step closes the heterocyclic ring to afford the target quinazolinone. For the detailed standardized operating procedures and specific workup protocols, please refer to the guide below.
- Perform selective nitration of 3-amino-4-hydroxybenzoic acid derivatives using zinc nitrate or concentrated nitric acid with a solid acid catalyst like trichloro-triazine (TCT) in acetonitrile at room temperature.
- Reduce the resulting nitro-intermediate to the corresponding diamine species using standard reduction protocols suitable for sensitive phenolic compounds.
- Execute the final ring-closing cyclization reaction to form the quinazolinone core structure, yielding the target 6-amino-7-hydroxyl-3,4-dihydroquinazoline-4-ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthetic route translates into tangible operational improvements and risk mitigation strategies. The most immediate impact is observed in the reduction of raw material complexity; by eliminating the need for protecting group reagents such as silyl chlorides, anhydrides, or bulky esters, the bill of materials is significantly simplified. This reduction in the number of distinct chemical inputs decreases the administrative burden of sourcing and qualifying multiple vendors, thereby streamlining the procurement process. Moreover, the removal of protection and deprotection steps inherently reduces the total volume of solvents and reagents consumed per kilogram of product, leading to a leaner manufacturing process that is less susceptible to supply chain disruptions caused by shortages of niche specialty chemicals. The overall result is a more resilient supply chain capable of sustaining continuous production schedules with fewer bottlenecks.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the drastic simplification of the synthetic sequence. In traditional routes, the cost of goods sold is heavily inflated by the expenses associated with protection and deprotection chemistry, which often account for a substantial portion of the total processing time and material usage. By obviating these steps, the new method effectively cuts out entire stages of production, reducing labor costs, utility consumption, and equipment occupancy time. Furthermore, the higher selectivity of the nitration reaction minimizes the formation of difficult-to-remove impurities, which reduces the load on purification units and increases the recovery rate of the final product. This cumulative effect results in a significantly lower cost base for the intermediate, providing a competitive edge in pricing negotiations for the final API.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the robustness of this process offers superior predictability. Conventional methods involving multiple protection steps are prone to variability; slight deviations in moisture content or temperature during protection can lead to incomplete reactions or difficult purifications, causing batch failures and delays. The direct nitration method described in the patent is more forgiving and operates under milder, easier-to-control conditions, such as room temperature stirring in common solvents. This operational stability ensures consistent batch-to-batch quality and reliable delivery timelines. Additionally, the use of widely available commodity chemicals like zinc nitrate and acetonitrile, rather than exotic protecting agents, ensures that raw material availability remains high, safeguarding against supply interruptions that could halt production lines.
- Scalability and Environmental Compliance: As regulatory pressures regarding chemical waste and emissions intensify globally, the environmental profile of a manufacturing process has become a critical factor in vendor selection. This novel route aligns perfectly with green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product). The elimination of protection groups means fewer byproducts are generated, and the avoidance of strong mineral acids like sulfuric acid reduces the corrosive load on equipment and the neutralization burden on wastewater treatment facilities. The process is inherently safer to scale up due to the absence of highly exothermic protection reactions and the use of less hazardous reagents. This makes the technology ideal for large-scale commercial production, allowing manufacturers to expand capacity without incurring prohibitive costs for waste management infrastructure or facing regulatory hurdles related to hazardous effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in patent CN103288757A, providing a clear understanding of how this method compares to legacy processes. Understanding these nuances is essential for R&D teams evaluating process transfer and for commercial teams assessing the long-term viability of this supply source. The answers below clarify the specific advantages related to catalyst selection, reaction conditions, and product quality assurance.
Q: Why is the new nitration method superior to traditional mixed acid nitration for this intermediate?
A: Traditional mixed acid nitration often causes oxidation of the sensitive amino and hydroxyl groups on the benzene ring, necessitating complex protection and deprotection steps. The patented method utilizes solid acid catalysts like trichloro-triazine (TCT) or aluminum dihydrogen phosphate, which enhance selectivity and prevent side reactions, allowing the reaction to proceed directly without protecting groups.
Q: What are the primary cost drivers eliminated by this synthetic route?
A: The most significant cost reduction comes from eliminating the reagents and solvents required for protecting and deprotecting the amino and hydroxyl functionalities. Additionally, the use of milder reaction conditions (room temperature) reduces energy consumption, and the higher selectivity minimizes waste generation and purification costs associated with removing oxidation byproducts.
Q: Is this process scalable for industrial production of Afatinib and Dacomitinib intermediates?
A: Yes, the process is highly amenable to scale-up. By avoiding hazardous mixed acids and exothermic protection steps, the safety profile is improved. The use of common solvents like acetonitrile and water, along with stable solid catalysts, ensures that the reaction can be safely transferred from laboratory glassware to large-scale stainless steel reactors for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Amino-7-Hydroxyl-3,4-Dihydroquinazoline-4-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving oncology therapies depends on the unwavering quality and availability of key intermediates. Our technical team has thoroughly analyzed the potential of the solid-acid catalyzed nitration route described in CN103288757A and is fully prepared to implement this state-of-the-art technology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch of 6-amino-7-hydroxyl-3,4-dihydroquinazoline-4-ketone meets stringent purity specifications, free from the genotoxic impurities often associated with traditional nitration methods.
We invite pharmaceutical partners and contract manufacturers to engage with us to explore how this optimized synthetic route can enhance your project economics. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation, ensuring that your supply chain for next-generation TKI APIs is built on a foundation of scientific excellence and operational reliability.
